APAC Ophthalmic Eye Dropper Market Overview
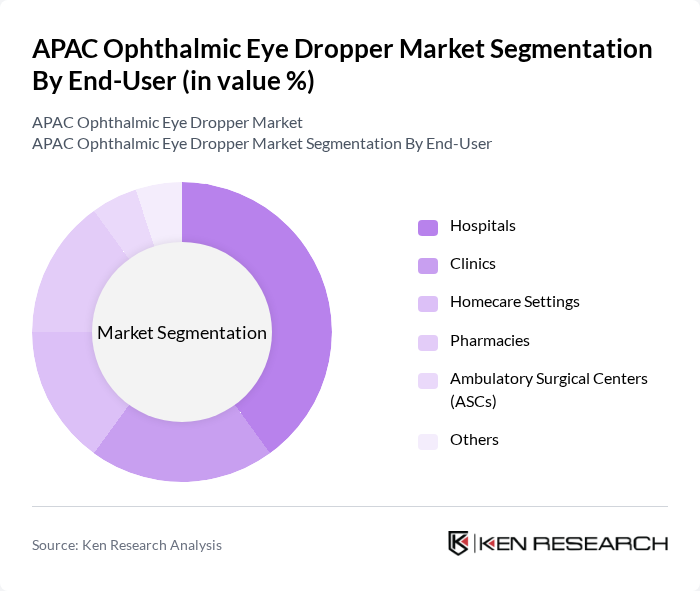
- The APAC Ophthalmic Eye Dropper Market is valued at USD 2.8 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of eye disorders, rising geriatric population, and advancements in drug delivery systems. The demand for innovative and user-friendly eye dropper designs has surged, reflecting a shift towards more efficient and effective treatment options for patients. Asia-Pacific is experiencing the fastest growth trajectory in the global ophthalmic eye dropper market, with expansion driven by rapidly aging populations, particularly in Japan, China, and South Korea, alongside rising prevalence of diabetes-related eye conditions leading to higher prescription drop usage.
- Key players in this market include China, Japan, and India, which dominate due to their large populations, increasing healthcare expenditure, and growing awareness of eye health. The presence of established pharmaceutical companies and a robust distribution network further enhance their market position, making these countries pivotal in the ophthalmic eye dropper landscape. India's government is investing significantly in advancing healthcare infrastructure, with the Union budget allocating substantial resources to transform its medical and pharmaceutical sectors, including ophthalmic eye droppers. Additionally, Japanese firms are beginning to pilot smart droppers integrated with smartphone apps to support aging citizens living alone, while China's pharmaceutical sector expansion is bolstering both domestic demand and global market positioning.
- The Indian government has implemented comprehensive regulations governing ophthalmic product safety and packaging standards to enhance consumer protection and prevent accidental misuse. These regulatory frameworks establish mandatory compliance requirements for product design, packaging specifications, and safety features, driving innovation in product development and packaging within the ophthalmic sector. Such regulations aim to protect vulnerable populations and ensure adherence to international safety standards.
APAC Ophthalmic Eye Dropper Market Segmentation
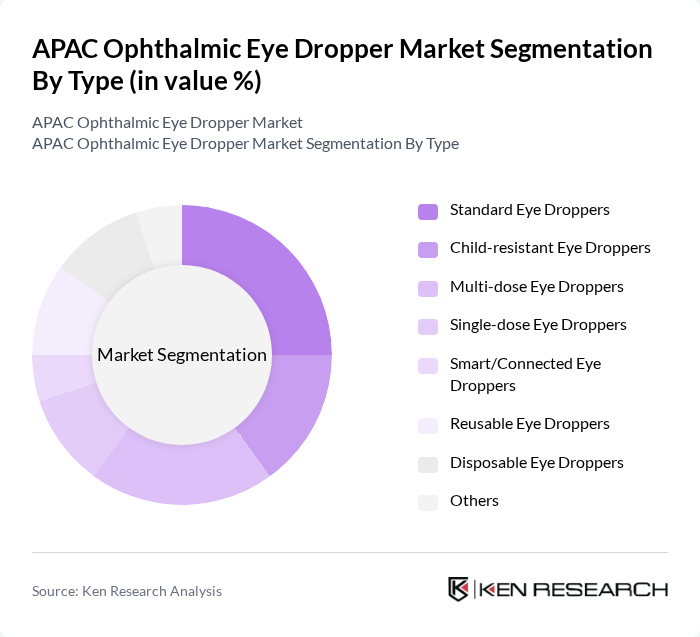
By Type:The market is segmented into various types of eye droppers, including Standard Eye Droppers, Child-resistant Eye Droppers, Multi-dose Eye Droppers, Single-dose Eye Droppers, Smart/Connected Eye Droppers, Reusable Eye Droppers, Disposable Eye Droppers, and Others. Each type serves specific consumer needs and preferences, influencing market dynamics. Multi-dose systems represent a significant segment, reflecting strong market demand for efficient, cost-effective delivery mechanisms across the region.
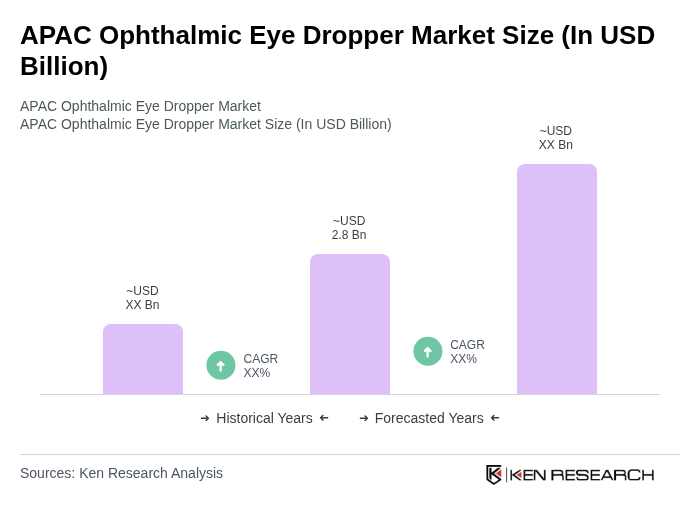
By End-User:The end-user segmentation includes Hospitals, Clinics, Homecare Settings, Pharmacies, Ambulatory Surgical Centers (ASCs), and Others. Each segment reflects the diverse applications of ophthalmic eye droppers across various healthcare settings. Hospitals maintain the largest share due to high-volume clinical usage and standardized procurement practices, while homecare settings are experiencing growth driven by increasing self-administration of ophthalmic medications among aging populations.
APAC Ophthalmic Eye Dropper Market Competitive Landscape
The APAC Ophthalmic Eye Dropper Market is characterized by a dynamic mix of regional and international players. Leading participants such as Alcon Laboratories, Inc., Bausch + Lomb, Johnson & Johnson Vision, Santen Pharmaceutical Co., Ltd., Novartis AG, Merck & Co., Inc., Pfizer Inc., AbbVie Inc., Ocular Therapeutix, Inc., Eyevance Pharmaceuticals, Sun Pharmaceutical Industries Ltd., Hikma Pharmaceuticals PLC, Aerie Pharmaceuticals, Inc., Bausch Health Companies Inc., Eyenovia, Inc., Aptar Pharma, Nemera, Gerresheimer AG, Nipro Corporation, Shandong Weigao Group Medical Polymer Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
APAC Ophthalmic Eye Dropper Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Eye Disorders:The APAC region is witnessing a significant rise in eye disorders, with an estimated 2.2 billion people affected by vision impairment. The World Health Organization reported that conditions like cataracts and glaucoma are prevalent, particularly in countries like India and China, where the aging population is projected to reach 400 million in future. This surge in eye health issues drives demand for effective ophthalmic treatments, including eye droppers.
- Rising Geriatric Population:The geriatric population in APAC is expected to exceed 630 million in future, according to the United Nations. This demographic shift is crucial as older adults are more susceptible to eye disorders, necessitating increased use of ophthalmic products. Countries like Japan and South Korea are leading in this trend, with over 29% of their populations aged 65 and above, thereby significantly boosting the demand for eye dropper solutions tailored for this age group.
- Advancements in Ophthalmic Drug Delivery Systems:Technological innovations in drug delivery systems are enhancing the efficacy of ophthalmic treatments. The introduction of precision eye droppers, which minimize wastage and improve dosage accuracy, is gaining traction. The market for these advanced devices is projected to grow, with investments in R&D expected to reach $1.5 billion in future, driven by the need for more effective and user-friendly ophthalmic solutions across the APAC region.
Market Challenges
- High Cost of Advanced Eye Dropper Technologies:The integration of advanced technologies in eye droppers often leads to higher production costs, which can limit accessibility. For instance, smart eye dropper devices can cost up to $200 each, making them less affordable for a significant portion of the population. This challenge is particularly pronounced in developing countries within the APAC region, where healthcare budgets are constrained, impacting overall market growth.
- Stringent Regulatory Requirements:The ophthalmic market in APAC faces rigorous regulatory scrutiny, which can delay product approvals. For example, the approval process for new ophthalmic products can take up to 24 months, as mandated by agencies like the Pharmaceuticals and Medical Devices Agency in Japan. These stringent regulations can hinder innovation and slow down the introduction of new eye dropper technologies, posing a challenge for manufacturers aiming to meet market demands swiftly.
APAC Ophthalmic Eye Dropper Market Future Outlook
The future of the APAC ophthalmic eye dropper market appears promising, driven by technological advancements and demographic shifts. As the prevalence of eye disorders continues to rise, the demand for innovative and effective drug delivery systems will likely increase. Additionally, the growing geriatric population will necessitate tailored solutions, fostering collaborations between manufacturers and healthcare providers. The market is expected to embrace digital health technologies, enhancing patient engagement and adherence to treatment regimens, ultimately improving health outcomes across the region.
Market Opportunities
- Expansion of E-commerce Platforms:The rise of e-commerce in APAC presents a significant opportunity for ophthalmic eye dropper manufacturers. With online sales projected to reach $500 billion in future, companies can leverage digital platforms to enhance product accessibility, particularly in rural areas where traditional distribution channels are limited. This shift can facilitate wider reach and increased sales for innovative eye dropper solutions.
- Development of Smart Eye Dropper Devices:The growing interest in smart healthcare solutions offers a lucrative opportunity for the ophthalmic market. Smart eye droppers equipped with IoT technology can provide real-time data on usage and adherence, appealing to tech-savvy consumers. With an estimated market value of $1 billion for smart medical devices in APAC in future, this segment is poised for rapid growth, attracting investments and fostering innovation.