APAC T Cell Therapy Market Overview
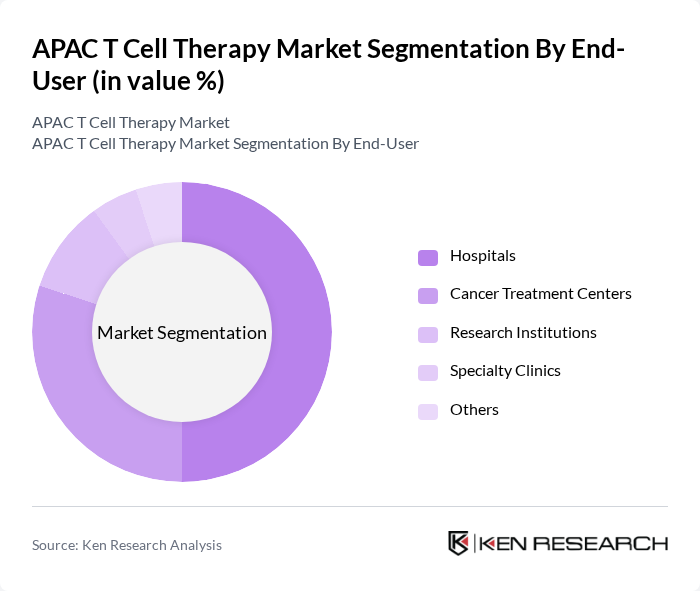
- The APAC T Cell Therapy Market is valued at USD 825 million, based on a five-year historical analysis. This growth is primarily driven by increasing investments in research and development, the rising prevalence of cancer, and advancements in cell engineering technologies that enhance the efficacy and safety of T cell therapies. The market is further supported by a surge in clinical trials, regulatory approvals for innovative therapies, and expanding access to advanced treatment options for patients across the region .
- Key players in this market include China, Japan, and India, which dominate due to robust healthcare infrastructure, significant investments in biotechnology, and large patient populations. China leads with rapid adoption of advanced therapies and extensive clinical research initiatives, while Japan benefits from strong regulatory frameworks and innovation in medical technologies. India is emerging as a hub for clinical trials and cost-effective treatment options, supported by improving financing programs and public-private partnerships .
- The National Guidelines for Stem Cell Research, 2017, issued by the Indian Council of Medical Research (ICMR) and the Department of Biotechnology, provide the regulatory framework for cellular therapies in India. These guidelines outline operational requirements for clinical trials, ethical review, and quality standards for manufacturing and administering T cell therapies, ensuring compliance and patient safety in research and clinical applications .
APAC T Cell Therapy Market Segmentation
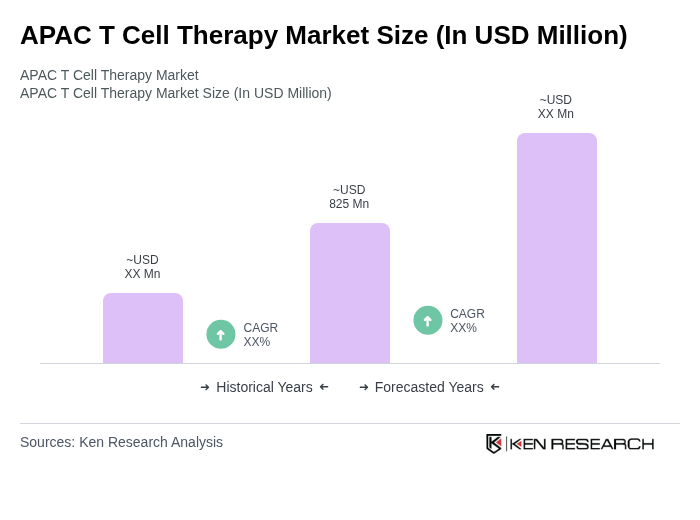
By Type:The T Cell Therapy market can be segmented into various types, including CAR-T Cell Therapy, TCR-T Cell Therapy, NK Cell Therapy, Dendritic Cell Therapy, and Others. Among these, CAR-T Cell Therapy is currently the most dominant segment due to its proven efficacy in treating hematological malignancies and its growing acceptance in clinical practice. The CD19-targeted CAR-T therapies, in particular, account for the largest share of the market, driven by their widespread application in blood cancers .
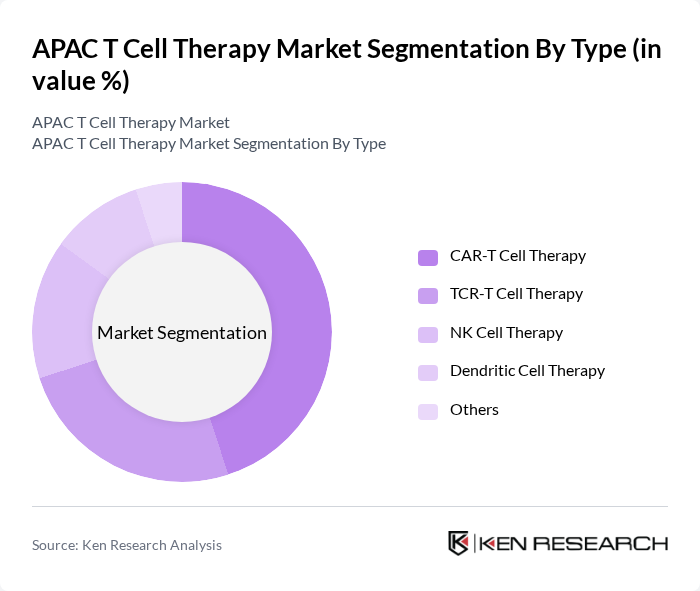
By End-User:The market can also be segmented based on end-users, which include Hospitals, Cancer Treatment Centers, Research Institutions, Specialty Clinics, and Others. Hospitals are the leading end-user segment, primarily due to their capacity to provide comprehensive care, multidisciplinary expertise, and access to advanced treatment technologies. Cancer Treatment Centers are also significant contributors, reflecting the concentration of expertise and resources for cellular immunotherapies in specialized settings .
APAC T Cell Therapy Market Competitive Landscape
The APAC T Cell Therapy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc., Bristol Myers Squibb Company, Kite Pharma, Inc., Amgen Inc., Celgene Corporation, Merck KGaA, Takeda Pharmaceutical Company Limited, Roche Holding AG, Pfizer, Inc., Johnson & Johnson, AbbVie, Inc., GenScript Biotech Corporation, APAC Biotech Pvt. Ltd., Immunocore Holdings plc contribute to innovation, geographic expansion, and service delivery in this space.
APAC T Cell Therapy Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer and Autoimmune Diseases:The APAC region is witnessing a significant rise in cancer cases, with an estimated4.57 million new cancer cases reportedaccording to the International Agency for Research on Cancer. Additionally, autoimmune diseases are affecting approximately5% of the populationin countries like Japan and South Korea. This growing patient population is driving demand for innovative T cell therapies, which are increasingly recognized for their potential in treating these conditions effectively.
- Advancements in CAR-T Cell Therapy Technology:The technological advancements in CAR-T cell therapies have been remarkable, withover 10 CAR-T products approved in the APAC region. These therapies have shown promising results, withcomplete response rates exceeding 50% in certain hematological malignancies. The ongoing research and development efforts are expected to enhance the efficacy and safety profiles of these therapies, further propelling their adoption across the region's healthcare systems.
- Rising Investment in R&D for T Cell Therapies:Investment in research and development for T cell therapies in the APAC region reached approximatelyUSD 1.38 billion, reflecting a15% increasefrom the previous period. This surge in funding is driven by both public and private sectors, aiming to accelerate the development of innovative therapies. The influx of capital is expected to foster collaborations and expedite clinical trials, ultimately enhancing the therapeutic landscape for patients in need.
Market Challenges
- High Cost of T Cell Therapies:The cost of T cell therapies remains a significant barrier, with treatments often exceedingUSD 400,000 per patient. This high price point limits accessibility, particularly in lower-income APAC countries where healthcare budgets are constrained. As a result, many patients may not receive timely treatment, leading to poorer health outcomes and increased mortality rates, which poses a challenge for market growth.
- Regulatory Hurdles and Approval Timelines:The regulatory landscape for T cell therapies in the APAC region is complex, with approval timelines averaging18-24 months. This lengthy process can delay patient access to potentially life-saving treatments. Additionally, varying regulatory standards across countries create inconsistencies that complicate the development and commercialization of these therapies, hindering market expansion and innovation.
APAC T Cell Therapy Market Future Outlook
The future of the APAC T cell therapy market appears promising, driven by ongoing advancements in technology and increasing collaboration among stakeholders. As healthcare systems evolve, the integration of personalized medicine and digital health technologies is expected to enhance treatment efficacy. Furthermore, the growing emphasis on patient-centric approaches will likely lead to improved outcomes, fostering greater acceptance and utilization of T cell therapies across the region.
Market Opportunities
- Growing Demand for Personalized Medicine:The shift towards personalized medicine is creating significant opportunities for T cell therapies, with an estimated market value ofUSD 3.5 billionin future. This trend is driven by the increasing recognition of the need for tailored treatments that address individual patient profiles, enhancing therapeutic effectiveness and patient satisfaction.
- Collaborations Between Biotech Firms and Research Institutions:Strategic partnerships are on the rise, withover 40 collaborations reported. These alliances are focused on leveraging combined expertise to accelerate the development of innovative T cell therapies. Such collaborations are expected to enhance research capabilities and streamline the path to market, ultimately benefiting patients and healthcare providers alike.