About the Report
Base Year 2024Listen to the audio summary
Australia Point of Care POC Diagnostics Market Overview
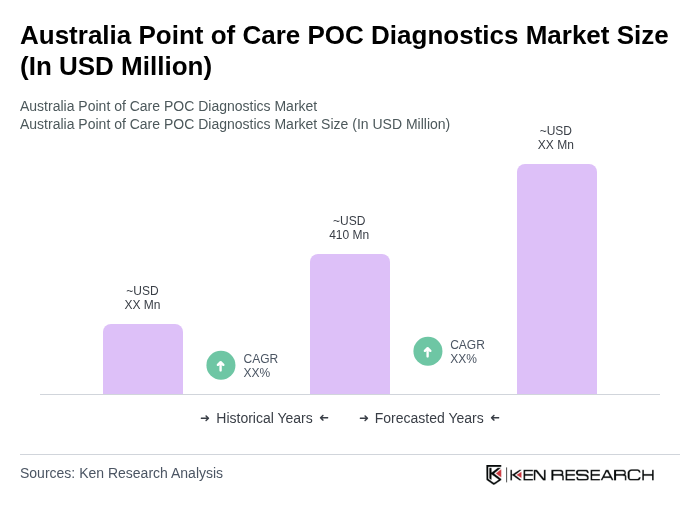
- The Australia Point of Care POC Diagnostics Market is valued at USD 410 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases such as diabetes, cancer, and cardiovascular diseases, the rising demand for rapid diagnostic tests, rising healthcare expenditure, increased awareness about early disease detection, and advancements in technology that enhance the accuracy and efficiency of diagnostic procedures.
- Key cities such as Sydney, Melbourne, and Brisbane dominate the market due to their advanced healthcare infrastructure, high population density, and significant investments in healthcare technology. These urban centers are also home to leading healthcare institutions and research facilities, fostering innovation and adoption of POC diagnostics.
- The Therapeutic Goods Administration (TGA) Medical Device Regulations, 2002 issued by the Australian Government Department of Health and Aged Care, govern the regulation of POC diagnostics as Class IIb or III medical devices depending on risk classification. Manufacturers must obtain TGA approval through conformity assessment procedures, including clinical evidence submission and post-market surveillance requirements for devices used in rapid testing across hospitals, clinics, and home settings.
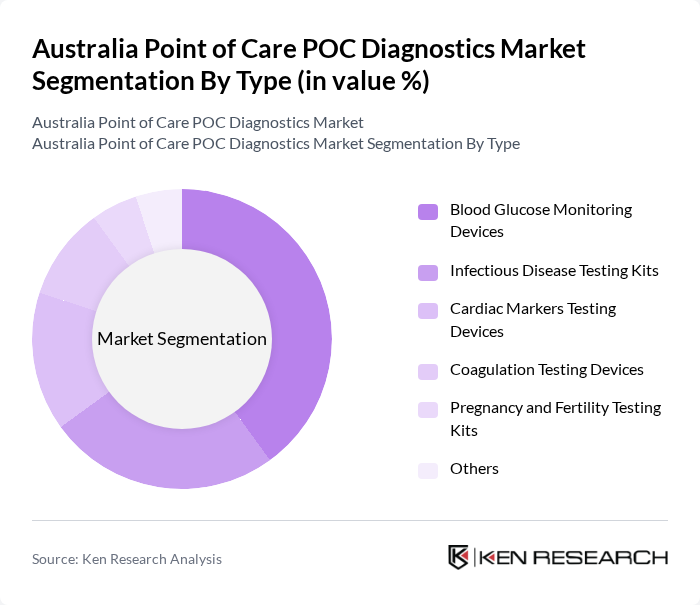
Australia Point of Care POC Diagnostics Market Segmentation
By Type:The market is segmented into various types of diagnostic devices, including Blood Glucose Monitoring Devices, Infectious Disease Testing Kits, Cardiac Markers Testing Devices, Coagulation Testing Devices, Pregnancy and Fertility Testing Kits, and Others. Among these, Blood Glucose Monitoring Devices are leading the market due to the rising prevalence of diabetes and the growing need for regular monitoring among patients. The convenience and ease of use of these devices have made them a preferred choice for both healthcare providers and patients.
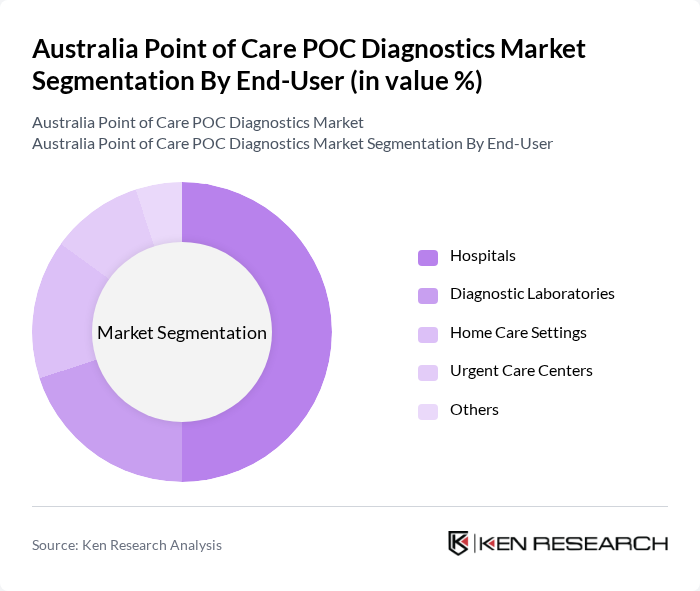
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Home Care Settings, Urgent Care Centers, and Others. Hospitals are the dominant end-user segment, driven by the increasing number of patients requiring immediate diagnostic results for effective treatment and the shift towards ambulatory and decentralized healthcare models. The integration of POC diagnostics in hospital settings enhances patient care by facilitating timely decision-making and improving overall healthcare delivery.
Australia Point of Care POC Diagnostics Market Competitive Landscape
The Australia Point of Care POC Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Cepheid, Alere Inc., Becton, Dickinson and Company, Thermo Fisher Scientific, Quidel Corporation, Hologic, Inc., Bio-Rad Laboratories, Ortho Clinical Diagnostics, Medtronic, Abbott Point of Care, Danaher Corporation, PerkinElmer, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Australia Point of Care POC Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the POC diagnostics market in Australia. In future, approximately 1.4 million Australians are projected to be diagnosed with diabetes, reflecting a 40% increase since 2010. This growing patient population necessitates rapid and accurate diagnostic solutions, propelling demand for POC testing devices that can deliver timely results, thereby improving patient management and outcomes.
- Rising Demand for Rapid Diagnostic Results:The urgency for quick diagnostic results is increasingly influencing the POC diagnostics market. In future, it is estimated that over 70% of healthcare providers in Australia will prioritize rapid testing capabilities to enhance patient care. This trend is driven by the need for immediate clinical decisions, particularly in emergency settings, where timely interventions can significantly impact patient survival rates and treatment efficacy, thus boosting the adoption of POC technologies.
- Technological Advancements in Diagnostic Devices:Continuous innovations in diagnostic technologies are transforming the POC diagnostics landscape. In future, the Australian market is expected to witness the introduction of over 20 new POC devices, incorporating advanced features such as AI integration and enhanced connectivity. These technological advancements not only improve accuracy and efficiency but also facilitate remote monitoring, aligning with the growing trend towards decentralized healthcare solutions and expanding market opportunities.
Market Challenges
- High Costs of Advanced Diagnostic Equipment:The financial burden associated with advanced POC diagnostic equipment poses a significant challenge. In future, the average cost of high-end POC devices is projected to exceed AUD 12,000, which can limit accessibility for smaller healthcare facilities. This high initial investment can deter adoption, particularly in rural areas where budget constraints are more pronounced, thereby hindering market growth and limiting patient access to essential diagnostic services.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape is a critical challenge for POC diagnostics in Australia. The Therapeutic Goods Administration (TGA) imposes stringent guidelines that can delay the approval process for new devices. In future, it is anticipated that the average time for regulatory approval will remain around 14-20 months, which can stifle innovation and market entry for new technologies, ultimately affecting the competitive landscape and availability of advanced diagnostic solutions.
Australia Point of Care POC Diagnostics Market Future Outlook
The future of the POC diagnostics market in Australia appears promising, driven by technological advancements and an increasing focus on personalized medicine. As healthcare providers continue to embrace telehealth solutions, the integration of POC testing into remote care models is expected to expand significantly. Additionally, the growing emphasis on preventive healthcare will likely lead to increased demand for portable diagnostic devices, enhancing patient engagement and facilitating early disease detection, ultimately improving health outcomes across the population.
Market Opportunities
- Expansion of Telehealth Services:The ongoing expansion of telehealth services presents a significant opportunity for POC diagnostics. In future, it is projected that telehealth consultations will increase by 50%, creating a demand for integrated POC testing solutions that can be utilized remotely, thereby enhancing patient access to timely diagnostics and improving overall healthcare delivery.
- Development of Portable Diagnostic Devices:The market for portable diagnostic devices is set to grow, driven by consumer demand for convenience and accessibility. In future, the introduction of at least 15 new portable POC devices is expected, catering to home healthcare needs. This trend will empower patients to manage their health proactively, reducing the burden on healthcare facilities and promoting a shift towards decentralized healthcare solutions.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Blood Glucose Monitoring Devices Infectious Disease Testing Kits Cardiac Markers Testing Devices Coagulation Testing Devices Pregnancy and Fertility Testing Kits Others |
| By End-User | Hospitals Diagnostic Laboratories Home Care Settings Urgent Care Centers Others |
| By Application | Cardiovascular Diseases Infectious Diseases Diabetes Management Cancer Screening Others |
| By Distribution Channel | Direct Sales Online Retail Distributors Pharmacies Others |
| By Technology | Lateral Flow Assays Microfluidics Molecular Diagnostics Immunoassays Others |
| By Region | New South Wales Victoria Queensland Western Australia South Australia Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Department of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Clinics
Technology Providers
Industry Associations (e.g., Medical Technology Association of Australia)
Financial Institutions
Players Mentioned in the Report:
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Cepheid
Alere Inc.
Becton, Dickinson and Company
Thermo Fisher Scientific
Quidel Corporation
Hologic, Inc.
Bio-Rad Laboratories
Ortho Clinical Diagnostics
Medtronic
Abbott Point of Care
Danaher Corporation
PerkinElmer, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Point of Care POC Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Point of Care POC Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Point of Care POC Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for rapid diagnostic results
3.1.3 Technological advancements in diagnostic devices
3.1.4 Growing focus on home healthcare solutions
3.2 Market Challenges
3.2.1 High costs of advanced diagnostic equipment
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Limited reimbursement policies
3.2.4 Competition from traditional laboratory testing
3.3 Market Opportunities
3.3.1 Expansion of telehealth services
3.3.2 Increasing investments in healthcare infrastructure
3.3.3 Development of portable diagnostic devices
3.3.4 Rising awareness of preventive healthcare
3.4 Market Trends
3.4.1 Integration of AI in diagnostic processes
3.4.2 Shift towards personalized medicine
3.4.3 Growth of point-of-care testing in remote areas
3.4.4 Increasing collaboration between tech companies and healthcare providers
3.5 Government Regulation
3.5.1 Therapeutic Goods Administration (TGA) guidelines
3.5.2 Compliance with ISO standards
3.5.3 Medicare reimbursement policies
3.5.4 Data protection regulations in healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Point of Care POC Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Point of Care POC Diagnostics Market Segmentation
8.1 By Type
8.1.1 Blood Glucose Monitoring Devices
8.1.2 Infectious Disease Testing Kits
8.1.3 Cardiac Markers Testing Devices
8.1.4 Coagulation Testing Devices
8.1.5 Pregnancy and Fertility Testing Kits
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Home Care Settings
8.2.4 Urgent Care Centers
8.2.5 Others
8.3 By Application
8.3.1 Cardiovascular Diseases
8.3.2 Infectious Diseases
8.3.3 Diabetes Management
8.3.4 Cancer Screening
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Online Retail
8.4.3 Distributors
8.4.4 Pharmacies
8.4.5 Others
8.5 By Technology
8.5.1 Lateral Flow Assays
8.5.2 Microfluidics
8.5.3 Molecular Diagnostics
8.5.4 Immunoassays
8.5.5 Others
8.6 By Region
8.6.1 New South Wales
8.6.2 Victoria
8.6.3 Queensland
8.6.4 Western Australia
8.6.5 South Australia
8.6.6 Others
8.7 By Policy Support
8.7.1 Government Subsidies
8.7.2 Tax Incentives
8.7.3 Research Grants
8.7.4 Others
9. Australia Point of Care POC Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Product Innovation Rate
9.2.7 Distribution Efficiency
9.2.8 Pricing Strategy
9.2.9 Customer Satisfaction Score
9.2.10 Operational Efficiency Ratio
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Abbott Laboratories
9.5.2 Roche Diagnostics
9.5.3 Siemens Healthineers
9.5.4 Cepheid
9.5.5 Alere Inc.
9.5.6 Becton, Dickinson and Company
9.5.7 Thermo Fisher Scientific
9.5.8 Quidel Corporation
9.5.9 Hologic, Inc.
9.5.10 Bio-Rad Laboratories
9.5.11 Ortho Clinical Diagnostics
9.5.12 Medtronic
9.5.13 Abbott Point of Care
9.5.14 Danaher Corporation
9.5.15 PerkinElmer, Inc.
10. Australia Point of Care POC Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Key decision-making processes
10.1.2 Budget allocation trends
10.1.3 Preferred suppliers and vendors
10.1.4 Evaluation criteria for procurement
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment trends in healthcare infrastructure
10.2.2 Budgeting for diagnostic technologies
10.2.3 Corporate partnerships and collaborations
10.2.4 Spending on R&D for diagnostics
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges faced by hospitals
10.3.2 Issues in home care settings
10.3.3 Barriers for diagnostic laboratories
10.3.4 Needs of urgent care centers
10.4 User Readiness for Adoption
10.4.1 Awareness of POC diagnostics
10.4.2 Training and education needs
10.4.3 Infrastructure readiness
10.4.4 User feedback and adaptation
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case studies of successful implementations
10.5.3 Expansion into new use cases
10.5.4 Long-term benefits analysis
11. Australia Point of Care POC Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Business model development
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health organizations and government health departments
- Review of published articles and white papers on POC diagnostics trends in Australia
- Examination of regulatory frameworks and guidelines from the Therapeutic Goods Administration (TGA)
Primary Research
- Interviews with healthcare professionals and laboratory managers in hospitals and clinics
- Surveys targeting manufacturers and distributors of POC diagnostic devices
- Focus groups with end-users, including patients and healthcare providers
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including industry reports and expert opinions
- Triangulation of quantitative data with qualitative insights from interviews
- Sanity checks through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure
- Segmentation of the market by product type, application, and end-user demographics
- Incorporation of growth rates from historical data and emerging trends in telehealth
Bottom-up Modeling
- Collection of sales data from leading POC diagnostic device manufacturers
- Estimation of market share based on unit sales and revenue figures
- Analysis of pricing strategies and average selling prices across different product categories
Forecasting & Scenario Analysis
- Development of predictive models using historical growth rates and market drivers
- Scenario analysis based on potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital POC Diagnostics | 120 | Clinical Laboratory Managers, Medical Technologists |
| Home Healthcare POC Devices | 100 | Home Care Nurses, Patients using POC devices |
| Point of Care Testing in Pharmacies | 80 | Pharmacy Managers, Pharmacists |
| POC Diagnostics in Remote Areas | 70 | Rural Health Practitioners, Community Health Workers |
| Emerging Technologies in POC Diagnostics | 60 | Biomedical Engineers, Product Development Managers |
Frequently Asked Questions
What is the current value of the Australia Point of Care (POC) Diagnostics Market?
The Australia Point of Care Diagnostics Market is valued at approximately USD 410 million, driven by the increasing prevalence of chronic diseases, demand for rapid diagnostic tests, and advancements in technology that enhance diagnostic accuracy and efficiency.