Region:Middle East
Author(s):Shubham
Product Code:KRAA8951
Pages:100
Published On:November 2025
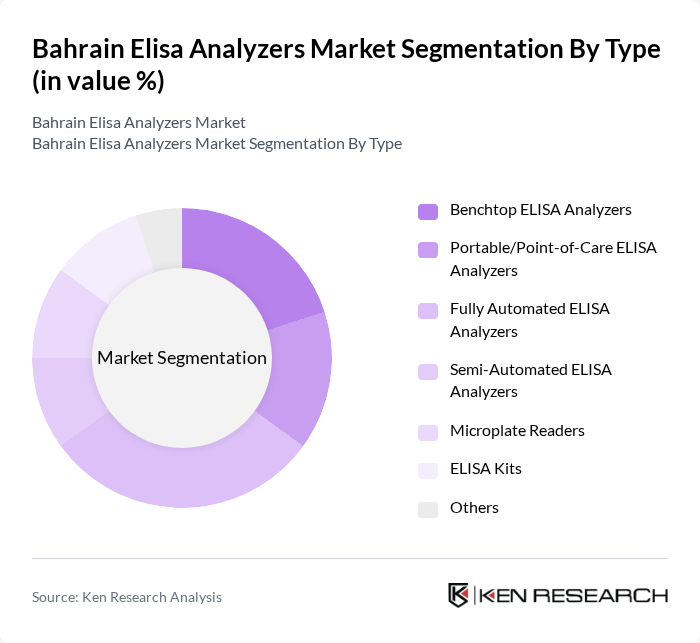
By Type:The market is segmented into various types of ELISA analyzers, including Benchtop ELISA Analyzers, Portable/Point-of-Care ELISA Analyzers, Fully Automated ELISA Analyzers, Semi-Automated ELISA Analyzers, Microplate Readers, ELISA Kits, and Others. Among these, Fully Automated ELISA Analyzers are gaining significant traction due to their efficiency, reduced manual intervention, and ability to handle high volumes of tests. This is particularly important in busy clinical and research settings, where rapid turnaround and scalability are essential. Benchtop analyzers remain widely used in centralized laboratories, while portable analyzers are increasingly adopted in decentralized and field settings for their mobility and rapid results .
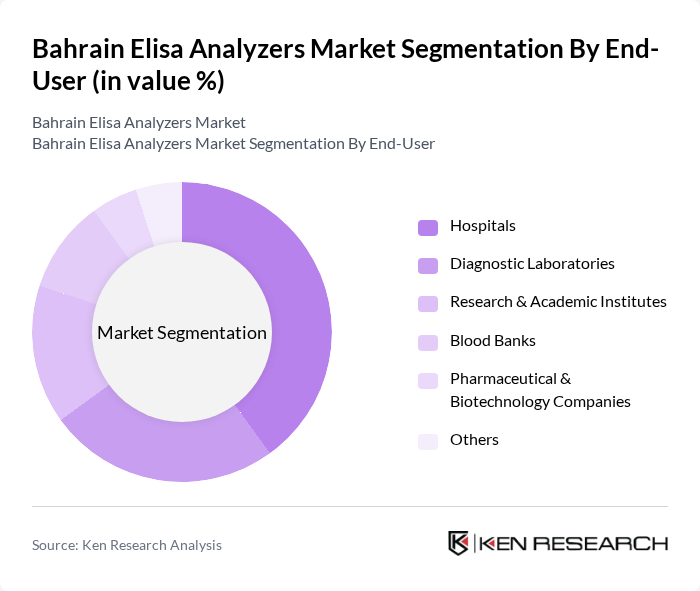
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research & Academic Institutes, Blood Banks, Pharmaceutical & Biotechnology Companies, and Others. Hospitals are the leading end-users, driven by the growing demand for rapid and accurate diagnostic testing in patient care. Diagnostic laboratories follow, supported by the expansion of laboratory networks and increased testing volumes. Research and academic institutes, as well as pharmaceutical and biotechnology companies, are also contributing to market growth through investments in clinical research and drug development .
The Bahrain Elisa Analyzers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Bio-Rad Laboratories, Thermo Fisher Scientific, Ortho Clinical Diagnostics, Beckman Coulter, PerkinElmer, Fujirebio, Meridian Bioscience, GenWay Biotech, Viroclinics Xplore, DiaSorin, MBL International, EMD Millipore (Merck Millipore) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain ELISA analyzers market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the shift towards point-of-care testing will make diagnostics more accessible. Additionally, the government's commitment to healthcare infrastructure development will likely facilitate the adoption of innovative diagnostic solutions, positioning the market for sustained growth in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Benchtop ELISA Analyzers Portable/Point-of-Care ELISA Analyzers Fully Automated ELISA Analyzers Semi-Automated ELISA Analyzers Microplate Readers ELISA Kits Others |
| By End-User | Hospitals Diagnostic Laboratories Research & Academic Institutes Blood Banks Pharmaceutical & Biotechnology Companies Others |
| By Application | Infectious Disease Testing Hormonal Testing Allergy Testing Autoimmune Disease Testing Oncology (Cancer Marker Testing) Drug Monitoring & Therapeutic Drug Management Others |
| By Region | Northern Governorate Southern Governorate Capital Governorate Muharraq Governorate Others |
| By Technology | Enzyme-Linked Immunosorbent Assay (ELISA) Chemiluminescent Immunoassay (CLIA) Fluorescent Immunoassay (FIA) Radioimmunoassay (RIA) Others |
| By Investment Source | Private Investments Government Funding International Grants Others |
| By Policy Support | Subsidies for Diagnostic Equipment Tax Incentives for Healthcare Providers Grants for Research and Development Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 80 | Laboratory Managers, Clinical Pathologists |
| Diagnostic Centers | 60 | Procurement Officers, Technical Directors |
| Research Institutions | 40 | Research Scientists, Lab Technicians |
| Public Health Organizations | 40 | Public Health Officials, Epidemiologists |
| Private Clinics | 50 | Clinic Owners, Medical Directors |
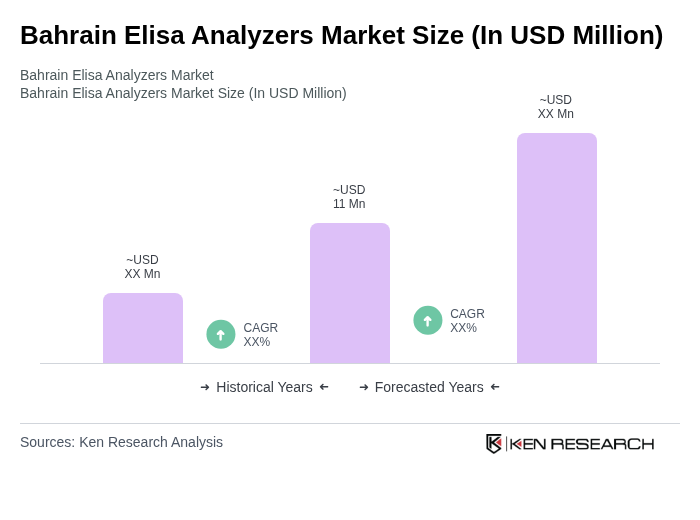
The Bahrain Elisa Analyzers Market is valued at approximately USD 11 million, reflecting a five-year historical analysis and recent data from the in vitro diagnostics sector. This growth is driven by increasing disease prevalence and advancements in diagnostic technologies.