Region:Middle East
Author(s):Shubham
Product Code:KRAA8606
Pages:89
Published On:November 2025
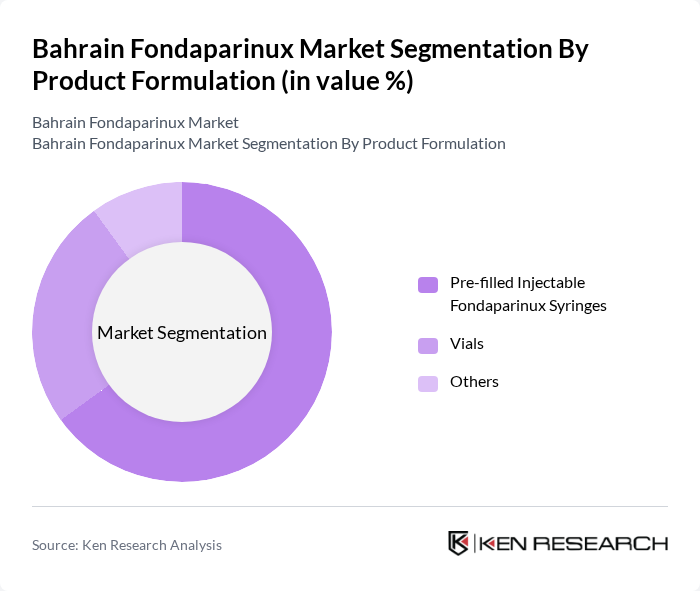
By Product Formulation:The product formulation segment includes pre-filled injectable Fondaparinux syringes, vials, and other forms. Among these, pre-filled injectable syringes account for the largest share of the Bahrain market, reflecting a global trend toward user-friendly and safe drug delivery systems that support both hospital and outpatient care. Vials remain important in hospital settings, especially for bulk purchasing and tailored dosing. The "Others" category comprises alternative delivery methods, such as ampoules or emerging device-assisted formats, which are gaining limited but increasing traction.
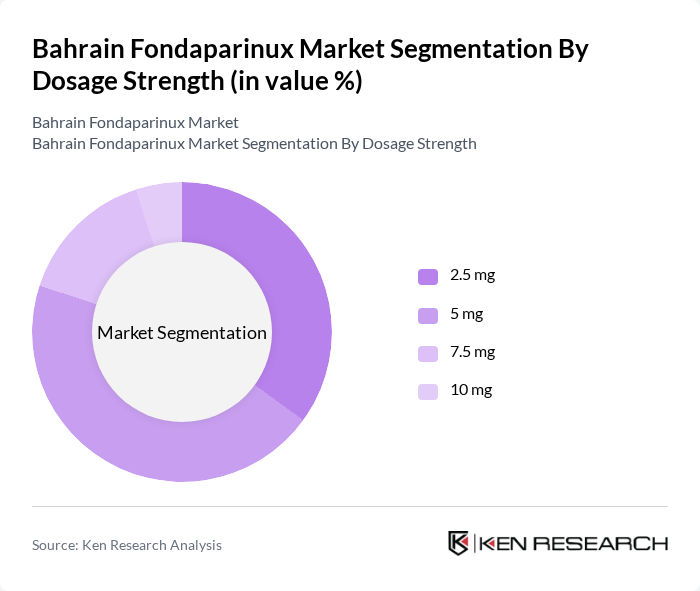
By Dosage Strength:This segment includes various dosage strengths of Fondaparinux, specifically 2.5 mg, 5 mg, 7.5 mg, and 10 mg. The 2.5 mg strength is most widely used in Bahrain, aligning with international clinical guidelines for VTE prophylaxis, particularly in surgical and moderate-risk patients. The 5 mg strength is also frequently prescribed for both prophylaxis and treatment, while higher strengths (7.5 mg and 10 mg) are reserved for severe or high-risk cases, contributing to their presence in the market.
The Bahrain Fondaparinux Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi, Dr. Reddy's Laboratories, Mylan (Viatris), Aspen Pharmacare, Pfizer, Novartis, Teva Pharmaceutical Industries, Sandoz, Cipla, Zydus Lifesciences, Abbott Laboratories, Hikma Pharmaceuticals, Sun Pharmaceutical Industries, Lupin Pharmaceuticals, and Apotex Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The Bahrain Fondaparinux market is poised for growth, driven by increasing healthcare investments and a rising prevalence of thromboembolic disorders. As the geriatric population expands, the demand for effective anticoagulant therapies will likely surge. Additionally, advancements in healthcare infrastructure will facilitate better access to treatments. The integration of digital health solutions and telemedicine services will further enhance patient management, ensuring timely access to Fondaparinux and improving overall health outcomes in the region.
| Segment | Sub-Segments |
|---|---|
| By Product Formulation | Pre-filled Injectable Fondaparinux Syringes Vials Others |
| By Dosage Strength | mg mg mg mg |
| By End-User | Hospitals Specialty Clinics Home Healthcare Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Therapeutic Application | Venous Thromboembolism (VTE) Prophylaxis Deep Vein Thrombosis (DVT) Treatment Pulmonary Embolism (PE) Treatment Acute Coronary Syndrome Others |
| By Patient Demographics | Adult Patients Geriatric Patients Pediatric Patients Others |
| By Region | Capital Governorate Northern Governorate Southern Governorate Muharraq Governorate |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 100 | Cardiologists, Clinical Pharmacists |
| Hematology Clinics | 60 | Hematologists, Nurse Practitioners |
| Pharmacy Chains | 50 | Pharmacy Managers, Pharmacists |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
| Health Insurance Providers | 40 | Underwriters, Claims Analysts |
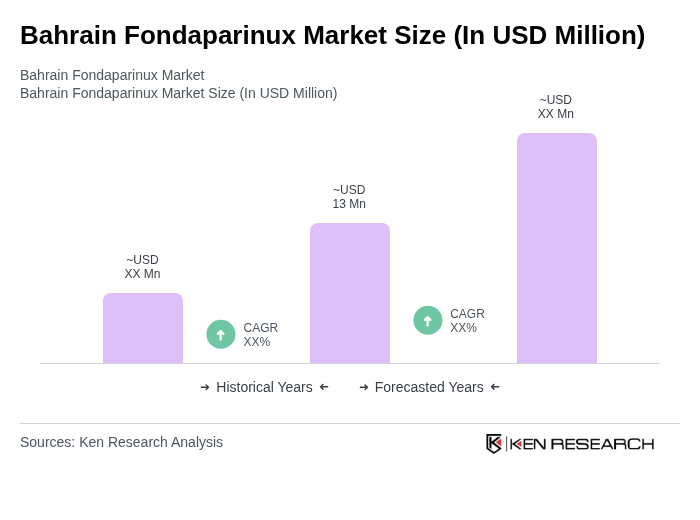
The Bahrain Fondaparinux Market is valued at approximately USD 13 million, reflecting a five-year historical analysis. This growth is driven by the increasing prevalence of venous thromboembolism and advancements in drug formulations.