Region:Middle East
Author(s):Geetanshi
Product Code:KRAA9163
Pages:96
Published On:November 2025
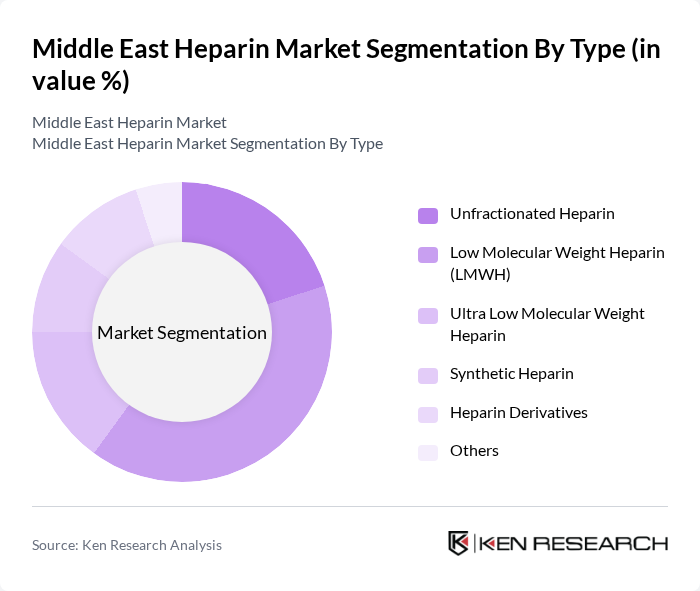
By Type:The heparin market can be segmented into several types, including Unfractionated Heparin, Low Molecular Weight Heparin (LMWH), Ultra Low Molecular Weight Heparin, Synthetic Heparin, Heparin Derivatives, and Others. Among these, Low Molecular Weight Heparin (LMWH) is the most dominant due to its ease of administration, lower risk of side effects, and favorable pharmacokinetic profile, making it a preferred choice for both healthcare providers and patients .
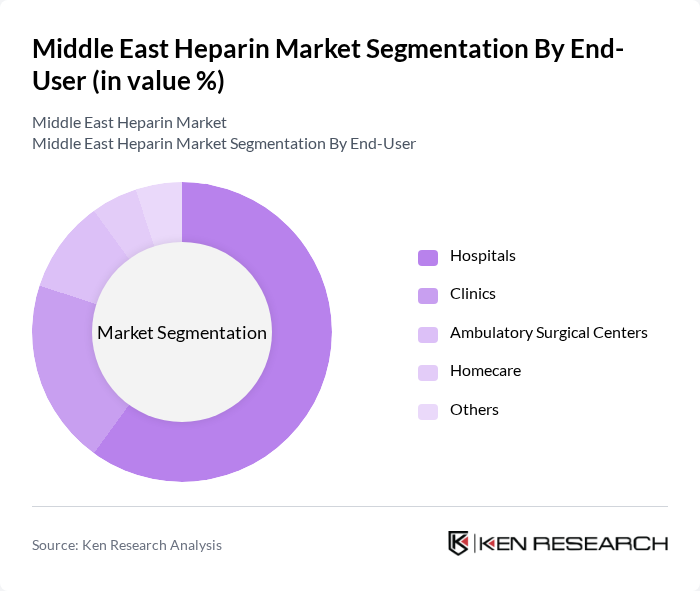
By End-User:The end-user segmentation includes Hospitals, Clinics, Ambulatory Surgical Centers, Homecare, and Others. Hospitals are the leading end-users of heparin products, primarily due to the high volume of surgical procedures, critical care treatments, and the need for effective thrombosis management. The increasing number of surgeries and the prevalence of chronic conditions requiring anticoagulation therapy in hospitals drive the demand for heparin .
The Middle East Heparin Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi, Pfizer, B. Braun Melsungen AG, GlaxoSmithKline plc, Novartis AG, Teva Pharmaceutical Industries Ltd., Hikma Pharmaceuticals plc, Fresenius Kabi AG, Boehringer Ingelheim GmbH, Viatris Inc. (formerly Mylan N.V.), LEO Pharma A/S, Amgen Inc., Sandoz (a Novartis division), Aurobindo Pharma Ltd., Baxter International Inc., Julphar (Gulf Pharmaceutical Industries), Pharco Pharmaceuticals, SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Tabuk Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space.
The future of the heparin market in the Middle East appears promising, driven by advancements in medical technology and increasing healthcare investments. The shift towards personalized medicine is expected to enhance treatment efficacy, while the adoption of digital health technologies will streamline patient management. Additionally, the growing focus on biosimilars may provide cost-effective alternatives, further expanding market access and improving patient care in the region, fostering a competitive landscape for heparin products.
| Segment | Sub-Segments |
|---|---|
| By Type | Unfractionated Heparin Low Molecular Weight Heparin (LMWH) Ultra Low Molecular Weight Heparin Synthetic Heparin Heparin Derivatives Others |
| By End-User | Hospitals Clinics Ambulatory Surgical Centers Homecare Others |
| By Route of Administration | Subcutaneous Intravenous Oral Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies & Drug Stores Online Pharmacies Others |
| By Geography | GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Bahrain, Oman) Levant Region (Jordan, Lebanon, Syria, Iraq, Palestine) North Africa (Egypt, Morocco, Algeria, Tunisia, Libya) Others |
| By Application | Thrombosis Treatment Cardiovascular Surgery Dialysis Deep Vein Thrombosis (DVT) Pulmonary Embolism Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 60 | Procurement Managers, Supply Chain Coordinators |
| Pharmaceutical Distributors | 50 | Sales Managers, Distribution Executives |
| Healthcare Providers (Clinics & Hospitals) | 80 | Physicians, Nursing Staff, Pharmacists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Policy Makers |
| Research Institutions | 40 | Clinical Researchers, Academic Professors |
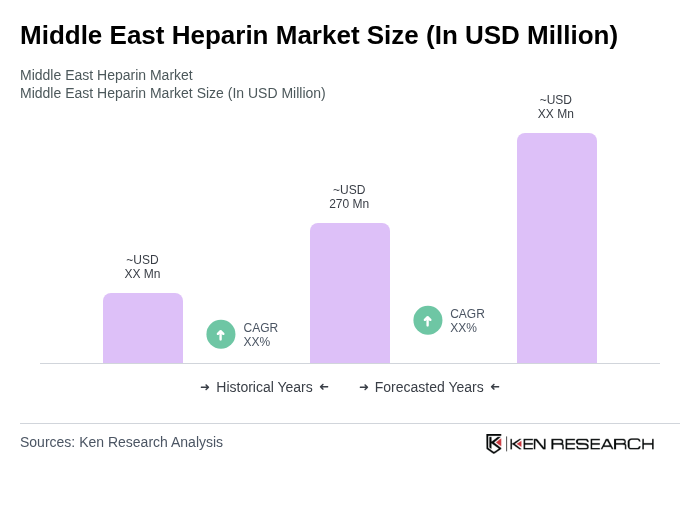
The Middle East Heparin Market is valued at approximately USD 270 million, driven by the increasing prevalence of thromboembolic and cardiovascular disorders, an aging population, and advancements in healthcare infrastructure across the region.