Region:Global
Author(s):Rebecca
Product Code:KRAA2941
Pages:90
Published On:August 2025
By Product Type:
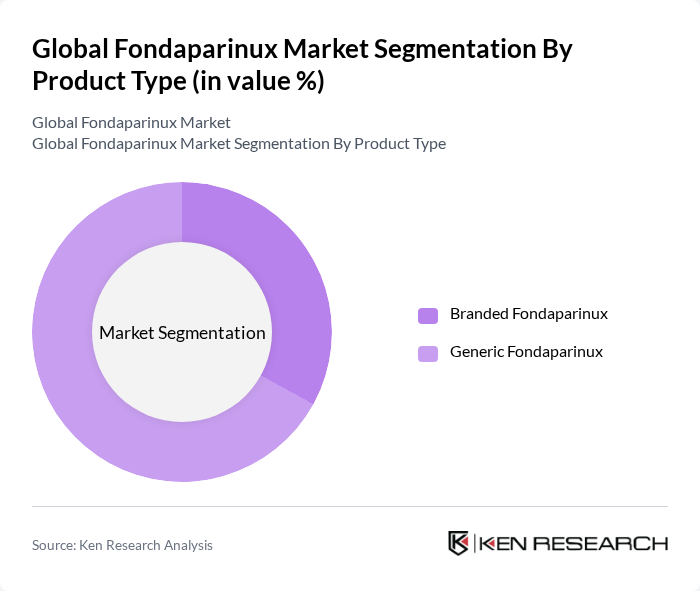
The product type segmentation reveals that generic fondaparinux is currently dominating the market. This trend is driven by the widespread availability and cost-effectiveness of generic formulations, which have gained significant acceptance among healthcare providers and payers. While branded fondaparinux maintains a reputation for quality and physician trust, the increasing pressure to reduce healthcare costs and the proven therapeutic equivalence of generics have shifted market preference toward generic alternatives, especially in hospital and retail pharmacy channels .
By Indication:
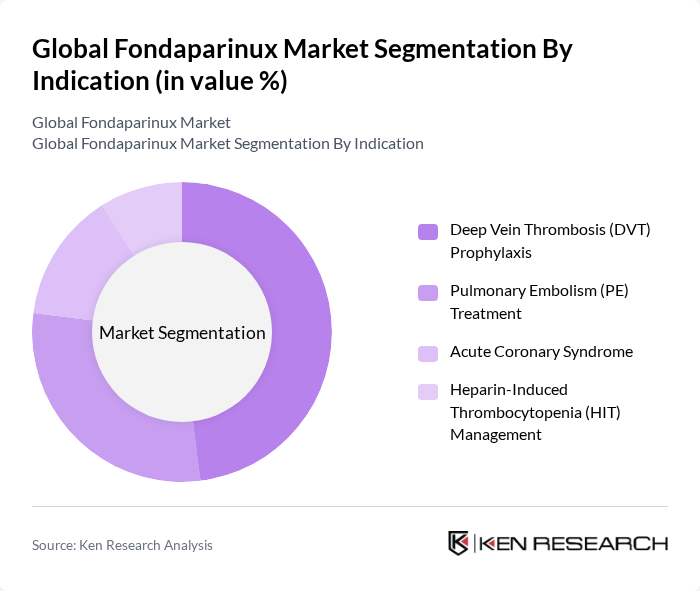
In terms of indication, the market is primarily driven by the need for deep vein thrombosis (DVT) prophylaxis, which accounts for the largest share. This is attributed to the increasing incidence of DVT in hospitalized patients and those undergoing major orthopedic or cancer-related surgeries. The effectiveness of fondaparinux in preventing DVT, combined with its favorable safety profile, has made it a preferred choice among healthcare providers. Pulmonary embolism (PE) treatment also holds a significant share, reflecting the critical need for effective management of this life-threatening condition. Acute coronary syndrome and heparin-induced thrombocytopenia (HIT) management represent smaller but important segments, supported by clinical guidelines and expanding clinical evidence .
The Global Fondaparinux Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi S.A., Viatris Inc., Dr. Reddy’s Laboratories Ltd., Pfizer Inc., Teva Pharmaceutical Industries Ltd., Sandoz International GmbH, Mylan N.V., Apotex Inc., Aurobindo Pharma Limited, Zydus Lifesciences Limited, Cipla Limited, Lupin Limited, Intas Pharmaceuticals Ltd., Glenmark Pharmaceuticals Ltd., Sun Pharmaceutical Industries Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the fondaparinux market appears promising, driven by the increasing prevalence of thromboembolic disorders and the aging population. Innovations in drug delivery systems and personalized medicine are expected to enhance treatment efficacy and patient adherence. Additionally, the integration of digital health technologies will facilitate better monitoring and management of anticoagulant therapies, ultimately improving patient outcomes and expanding market reach. The focus on preventive healthcare will further bolster the demand for effective anticoagulants in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Branded Fondaparinux Generic Fondaparinux |
| By Indication | Deep Vein Thrombosis (DVT) Prophylaxis Pulmonary Embolism (PE) Treatment Acute Coronary Syndrome Heparin-Induced Thrombocytopenia (HIT) Management |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By End-User | Hospitals Clinics Homecare Settings |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacy Management | 100 | Pharmacy Directors, Clinical Pharmacists |
| Cardiology Departments | 60 | Cardiologists, Nurse Practitioners |
| Oncology Treatment Centers | 50 | Oncologists, Treatment Coordinators |
| Health Insurance Providers | 40 | Medical Directors, Claims Analysts |
| Clinical Research Organizations | 70 | Clinical Research Associates, Project Managers |
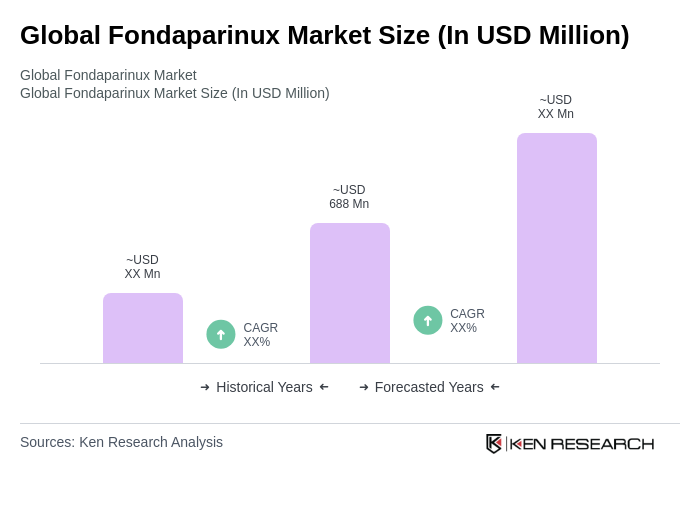
The Global Fondaparinux Market is valued at approximately USD 688 million, driven by the increasing prevalence of thromboembolic disorders and advancements in healthcare infrastructure. This market is expected to grow as demand for effective anticoagulants rises.