GCC Medical Device Design Development Services Market Overview
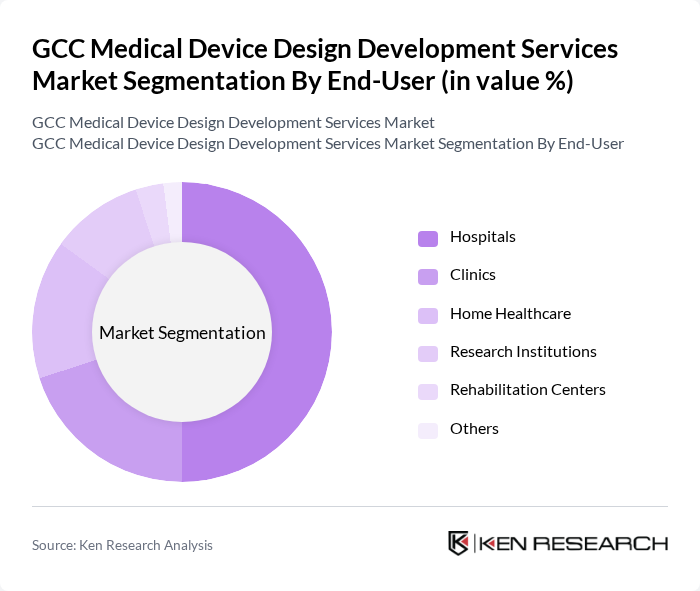
- The GCC Medical Device Design Development Services Market is valued at USD 1.1 billion, based on a five-year historical analysis. This valuation reflects the segment’s share within the broader GCC medical device market, which is driven by increasing demand for advanced medical technologies, rising healthcare expenditures, and a growing emphasis on patient-centric care. Growth is further supported by rapid adoption of digital health solutions, telehealth, and innovative medical devices that enhance diagnostic and therapeutic capabilities. Trends such as miniaturization, wearable devices, and connected healthcare are reshaping the market landscape, with service providers specializing in regulatory-compliant IoT integration and modular device design benefiting from expanding opportunities .
- Key players in this market include Saudi Arabia, the UAE, and Qatar, which dominate due to robust healthcare infrastructure, significant investments in healthcare technology, and government initiatives aimed at improving healthcare services. The presence of leading medical device manufacturers and a growing population with increasing healthcare needs further contribute to market expansion in these regions. The GCC region’s healthcare sector is characterized by strong R&D focus, geographic expansion, and strategic partnerships among major global and regional medical device companies .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Medical Devices Interim Regulation,” issued by the Saudi Food and Drug Authority, requiring all medical devices to undergo rigorous testing and certification before market entry. This regulation mandates comprehensive safety, performance, and quality standards, including conformity assessment procedures and post-market surveillance, thereby enhancing patient safety and fostering trust in healthcare technologies across the GCC region .
GCC Medical Device Design Development Services Market Segmentation
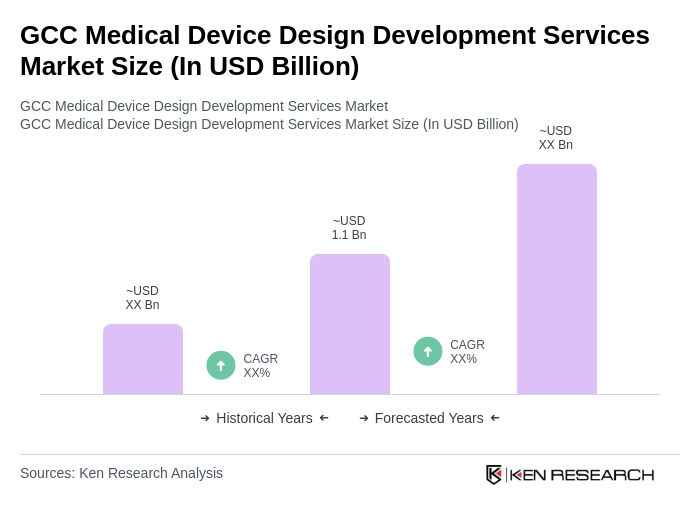
By Type:The market is segmented into various types of medical devices, including diagnostic devices, therapeutic devices, monitoring devices, surgical instruments, imaging equipment, consumables, and others. Among these, diagnostic devices currently lead the market, driven by the increasing prevalence of chronic diseases and the growing demand for early detection and diagnosis. The trend towards personalized medicine, miniaturization, and advancements in diagnostic technologies—such as point-of-care testing and wearable diagnostic solutions—further bolster the growth of this segment .
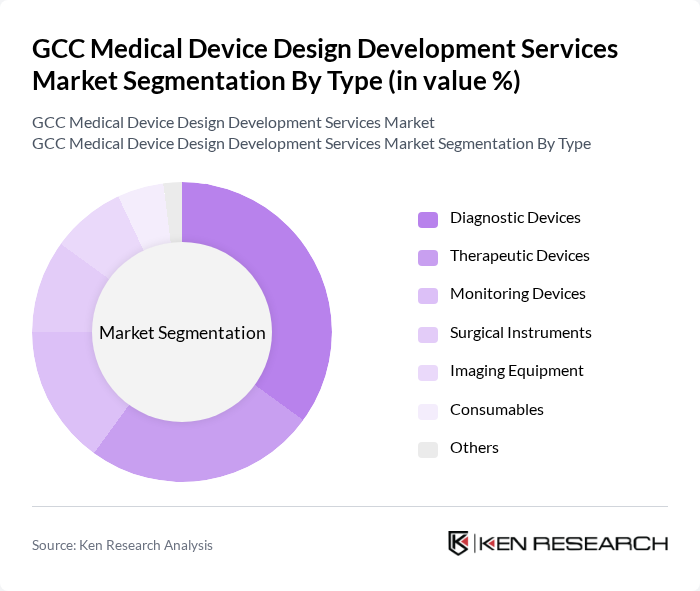
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, research institutions, rehabilitation centers, and others. Hospitals are the dominant end-user segment, driven by the increasing number of hospital admissions and the growing demand for advanced medical technologies. The expansion of healthcare facilities, integration of digital health systems, and adoption of remote monitoring solutions further support the growth of this segment. Home healthcare and clinics are also experiencing notable growth due to the rising adoption of portable and connected medical devices for chronic disease management and preventive care .
GCC Medical Device Design Development Services Market Competitive Landscape
The GCC Medical Device Design Development Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Jabil, Flex Ltd., Medtronic, Siemens Healthineers, Philips Healthcare, GE Healthcare, Abbott Laboratories, Boston Scientific, Johnson & Johnson, Stryker Corporation, B. Braun Melsungen AG, Canon Medical Systems, Olympus Corporation, Hologic, Inc., 3M Health Care contribute to innovation, geographic expansion, and service delivery in this space.
GCC Medical Device Design Development Services Market Industry Analysis
Growth Drivers
- Increasing Demand for Advanced Healthcare Solutions:The GCC region is witnessing a surge in demand for advanced healthcare solutions, driven by a population growth rate of 2.2% annually. This growth is accompanied by an increase in healthcare spending, projected to reach $116 billion in future. The rising expectations for quality healthcare services are pushing healthcare providers to adopt innovative medical devices, thereby enhancing the demand for design and development services in the medical device sector.
- Government Initiatives to Boost Healthcare Infrastructure:Governments in the GCC are investing heavily in healthcare infrastructure, with an estimated $35 billion allocated for healthcare projects in future. Initiatives such as the Saudi Vision 2030 and UAE Vision 2021 aim to improve healthcare access and quality. These investments are expected to create a favorable environment for medical device design and development services, as new facilities require advanced medical technologies to operate effectively.
- Rising Prevalence of Chronic Diseases:The GCC is experiencing a significant rise in chronic diseases, with diabetes rates projected to reach about 17% of the adult population in future. This increase necessitates the development of specialized medical devices for monitoring and treatment. The demand for innovative solutions to manage these conditions is driving growth in the medical device design and development sector, as healthcare providers seek to improve patient outcomes through advanced technologies.
Market Challenges
- Stringent Regulatory Requirements:The medical device industry in the GCC faces stringent regulatory requirements, which can delay product development timelines. For instance, the average time for medical device approval can exceed 14 months due to complex regulatory frameworks. Compliance with international standards, such as ISO 13485, adds to the challenges, requiring companies to invest significant resources in quality management systems to meet these regulations.
- High Development Costs:The costs associated with developing medical devices in the GCC are notably high, often exceeding $1.2 million for initial prototypes. This financial burden can deter smaller companies from entering the market, limiting innovation. Additionally, the need for extensive testing and validation processes further escalates costs, making it challenging for firms to maintain competitive pricing while ensuring product quality and compliance with regulatory standards.
GCC Medical Device Design Development Services Market Future Outlook
The future of the GCC medical device design and development services market appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence and machine learning into medical devices is expected to enhance diagnostic accuracy and patient care. Furthermore, the expansion of telemedicine services will create new avenues for device development, allowing for remote monitoring and treatment solutions that cater to the growing demand for accessible healthcare.
Market Opportunities
- Expansion of Telemedicine Services:The telemedicine market in the GCC is projected to grow to $2 billion in future, driven by increased smartphone penetration and internet access. This growth presents opportunities for developing medical devices that facilitate remote consultations and monitoring, enabling healthcare providers to reach underserved populations effectively.
- Increasing Focus on Personalized Medicine:The personalized medicine market in the GCC is expected to reach $3 billion in future, as healthcare providers seek tailored treatment solutions. This trend opens avenues for the design and development of customized medical devices that cater to individual patient needs, enhancing treatment efficacy and patient satisfaction.