Region:Middle East
Author(s):Shubham
Product Code:KRAD1021
Pages:82
Published On:November 2025
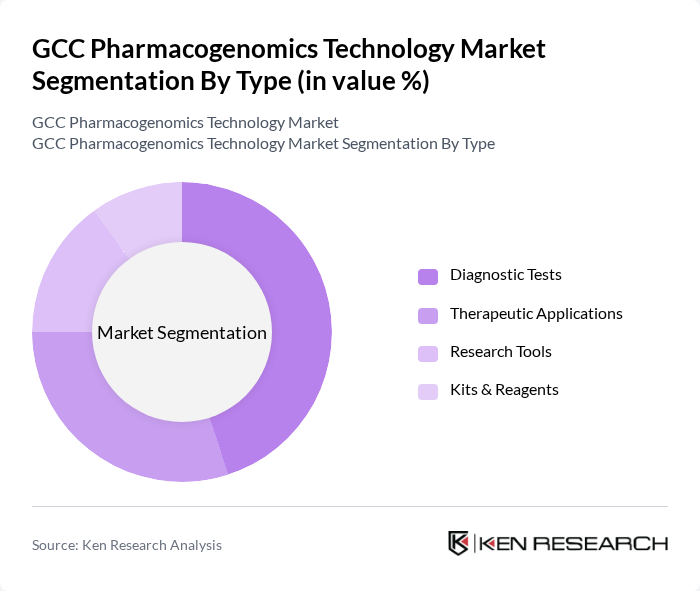
By Type:The market is segmented into Diagnostic Tests, Therapeutic Applications, Research Tools, and Kits & Reagents. Among these, Diagnostic Tests are leading due to their critical role in identifying genetic variations that affect drug response. The increasing focus on precision medicine and the growing number of genetic tests being adopted in clinical settings are driving this segment's growth. Therapeutic Applications are also significant, as they encompass treatments tailored to individual genetic profiles, enhancing treatment efficacy .
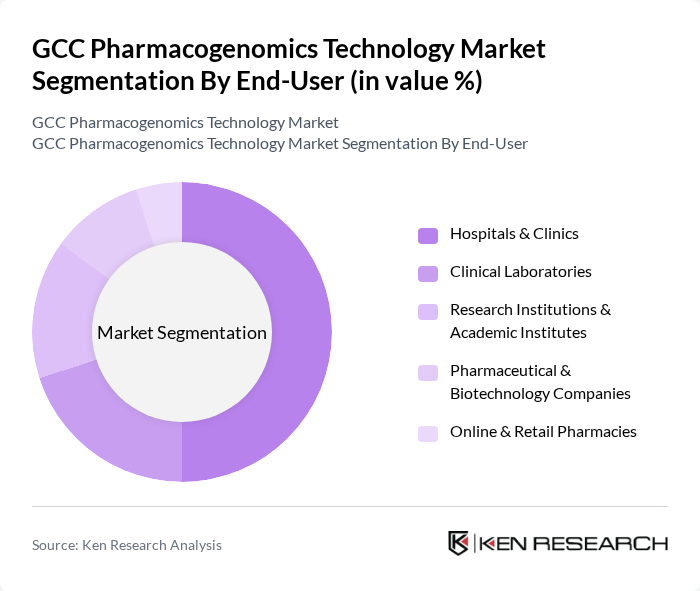
By End-User:The end-user segmentation includes Hospitals & Clinics, Clinical Laboratories, Research Institutions & Academic Institutes, Pharmaceutical & Biotechnology Companies, and Online & Retail Pharmacies. Hospitals & Clinics dominate this segment as they are the primary settings for patient care and genetic testing. The increasing adoption of pharmacogenomic testing in clinical practice to optimize drug therapy is a key factor driving this segment's growth. Pharmaceutical & Biotechnology Companies are also significant, as they utilize pharmacogenomics for drug development and personalized therapies .
The GCC Pharmacogenomics Technology Market is characterized by a dynamic mix of regional and international players. Leading participants such as Illumina, Inc., Thermo Fisher Scientific Inc., Roche Diagnostics, Agilent Technologies, Inc., QIAGEN N.V., Myriad Genetics, Inc., Genomic Health, Inc., BGI Genomics, PerkinElmer, Inc., Eurofins Scientific, F. Hoffmann-La Roche AG, Siemens Healthineers, Bio-Rad Laboratories, Inc., Abbott Laboratories, Hologic, Inc., Al Borg Diagnostics, King Faisal Specialist Hospital & Research Centre (KFSH&RC), G42 Healthcare, National Reference Laboratory (NRL), Centogene N.V. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC pharmacogenomics market appears promising, driven by increasing investments in healthcare infrastructure and a growing emphasis on personalized medicine. As governments in the region allocate more resources to healthcare, the integration of pharmacogenomics into clinical practice is expected to accelerate. Additionally, the rise of telemedicine is likely to enhance access to genetic testing, enabling more patients to benefit from tailored therapies and improving overall health outcomes in the GCC.
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Tests Therapeutic Applications Research Tools Kits & Reagents |
| By End-User | Hospitals & Clinics Clinical Laboratories Research Institutions & Academic Institutes Pharmaceutical & Biotechnology Companies Online & Retail Pharmacies |
| By Application | Oncology Cardiovascular Diseases Neurological & Psychiatric Disorders Infectious Diseases Pain Management Others |
| By Technology | Next-Generation Sequencing (NGS) Polymerase Chain Reaction (PCR) Microarray Technology Mass Spectrometry Others |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Investment Source | Private Investments Government Funding Venture Capital Others |
| By Policy Support | Subsidies for Research Tax Incentives Grants for Innovation Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Physicians, Pharmacists, Genetic Counselors |
| Genetic Testing Laboratories | 60 | Laboratory Managers, Technical Directors |
| Pharmaceutical Companies | 50 | R&D Managers, Product Development Leads |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Patient Representatives |
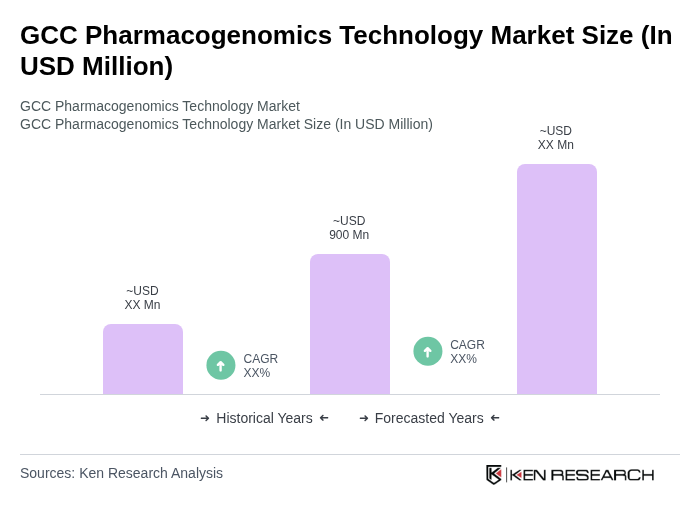
The GCC Pharmacogenomics Technology Market is valued at approximately USD 900 million, driven by the increasing prevalence of chronic diseases, advancements in genomic research, and the demand for personalized medicine.