Region:Middle East
Author(s):Rebecca
Product Code:KRAC3927
Pages:99
Published On:October 2025
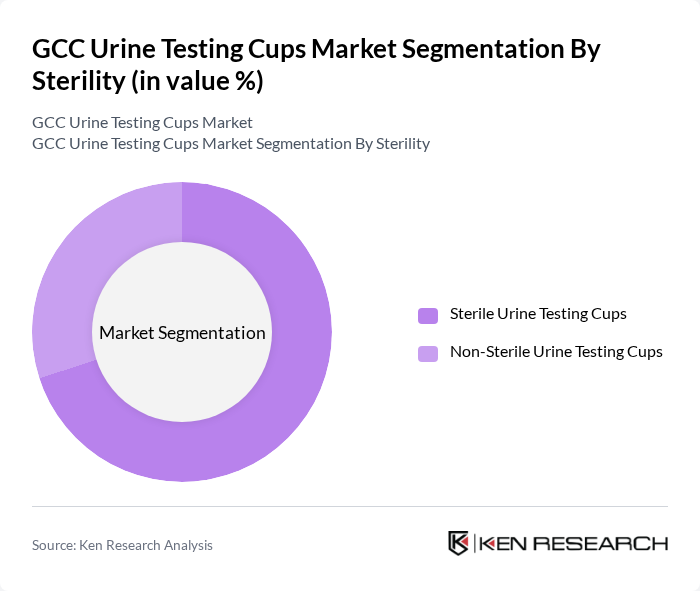
By Sterility:This segmentation focuses on the sterility of urine testing cups, which is crucial for ensuring accurate test results and preventing contamination. The two subsegments include Sterile Urine Testing Cups and Non-Sterile Urine Testing Cups. Sterile cups are preferred in clinical settings, hospitals, and diagnostic laboratories to maintain hygiene and comply with stringent quality standards, while non-sterile options are often used in less critical environments such as workplace screening programs and home-based testing applications.
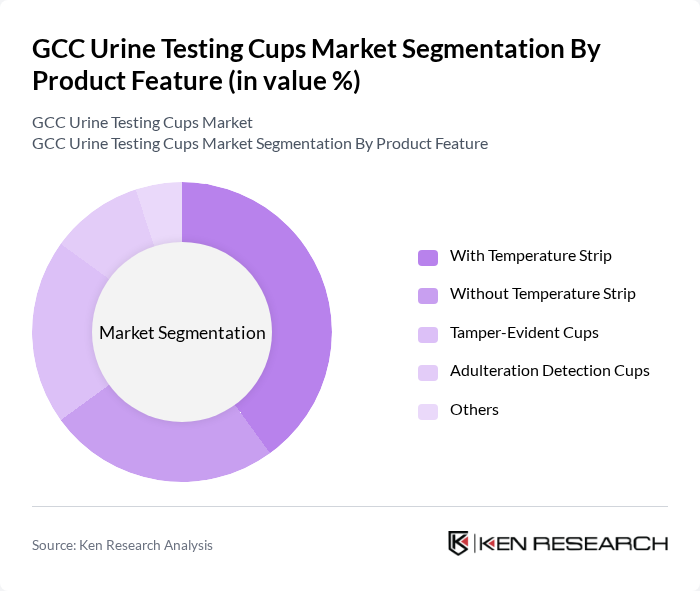
By Product Feature:This segmentation categorizes urine testing cups based on their features, which enhance usability and reliability. The subsegments include With Temperature Strip, Without Temperature Strip, Tamper-Evident Cups, Adulteration Detection Cups, and Others. The demand for cups with temperature strips is particularly high, as they help ensure the integrity of the sample by verifying that specimens are within acceptable temperature ranges immediately after collection. Tamper-evident features and adulteration detection capabilities have become increasingly important in workplace drug testing and law enforcement applications, where sample authenticity is critical. The market has also witnessed growing adoption of smart urine testing cups that incorporate IoT integration and digital connectivity for automated result recording and analysis.
The GCC Urine Testing Cups Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers, Roche Diagnostics, Bio-Rad Laboratories, Inc., Becton, Dickinson and Company, Medline Industries, LP, Premier Biotech, Inc., Alere Inc. (now part of Abbott), Chembio Diagnostic Systems, Inc., AccuBioTech Co., Ltd., LabCorp (Laboratory Corporation of America Holdings), Quest Diagnostics Incorporated, Medtox Scientific, Inc. (a LabCorp company), Tecan Group Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC urine testing cups market appears promising, driven by ongoing technological advancements and an increasing focus on health and wellness. As healthcare systems evolve, the integration of digital health technologies is expected to enhance testing accuracy and accessibility. Furthermore, the growing trend towards home-based testing solutions will likely expand market reach, catering to a broader demographic. These developments will create a dynamic environment for innovation and growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Sterility | Sterile Urine Testing Cups Non-Sterile Urine Testing Cups |
| By Product Feature | With Temperature Strip Without Temperature Strip Tamper-Evident Cups Adulteration Detection Cups Others |
| By Application | Drug Testing Pregnancy Testing Disease Detection Health Screening Others |
| By End-Use | Hospitals & Clinics Diagnostic Laboratories Home Use Rehabilitation Centers Corporate Health Programs Others |
| By Distribution Channel | Direct Sales Medical Supply Distributors Pharmacies Online Retail Others |
| By Region | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain Others |
| By Price Range | Low Price Range Mid Price Range High Price Range Others |
| By Brand | Established Brands Emerging Brands Private Labels Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 100 | Laboratory Managers, Medical Technologists |
| Diagnostic Centers | 60 | Procurement Officers, Operations Managers |
| Healthcare Clinics | 50 | Clinic Administrators, Healthcare Practitioners |
| Manufacturers of Testing Equipment | 40 | Product Development Managers, Sales Directors |
| Regulatory Bodies | 40 | Policy Makers, Health Inspectors |
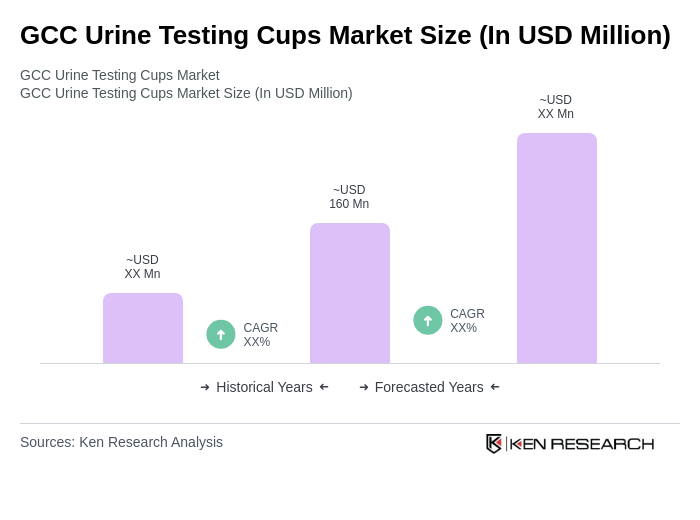
The GCC Urine Testing Cups Market is valued at approximately USD 160 million, reflecting a significant growth driven by increased drug testing, health screening initiatives, and advancements in urine testing technology across the region.