Region:Global
Author(s):Rebecca
Product Code:KRAD5024
Pages:96
Published On:December 2025
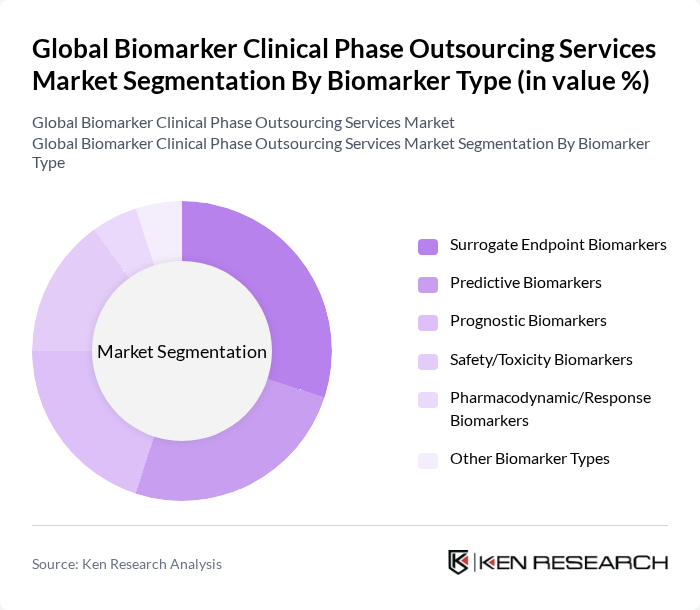
By Biomarker Type:The biomarker type segmentation includes various categories such as surrogate endpoint biomarkers, predictive biomarkers, prognostic biomarkers, safety/toxicity biomarkers, pharmacodynamic/response biomarkers, and other biomarker types. Among these, surrogate endpoint biomarkers are gaining traction due to their ability to act as substitutes for clinical endpoints and provide early indications of treatment efficacy, which is crucial for shortening trial duration and accelerating regulatory decision-making in clinical development.
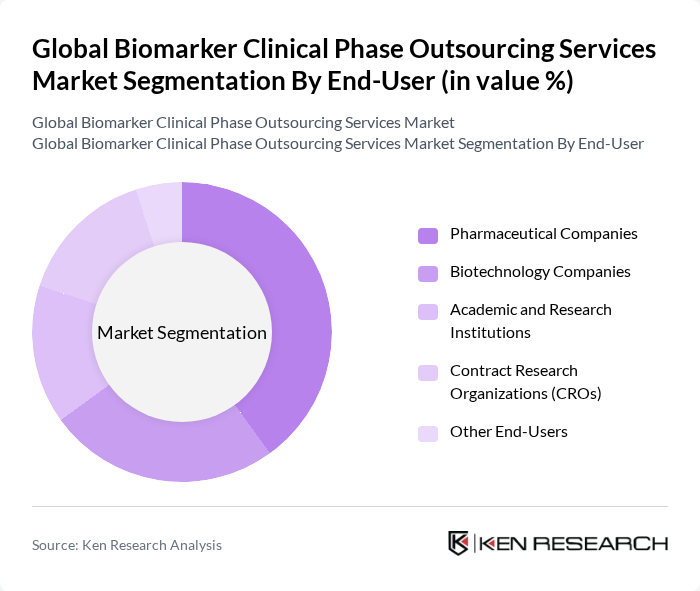
By End-User:The end-user segmentation encompasses pharmaceutical companies, biotechnology companies, academic and research institutions, contract research organizations (CROs), and other end-users. Pharmaceutical companies dominate this segment due to their substantial investments in biomarker research and development, extensive late?stage clinical pipelines, and increasing reliance on external partners for complex biomarker validation, testing, and companion diagnostic development that are essential for drug discovery and clinical validation processes.
The Global Biomarker Clinical Phase Outsourcing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Laboratory Corporation of America Holdings (Labcorp Drug Development), ICON plc, Eurofins Scientific SE, WuXi AppTec Co., Ltd., IQVIA Inc., Syneos Health, Inc., PPD, Inc. (Thermo Fisher Scientific), Charles River Laboratories International, Inc., Covance Inc. (Labcorp Drug Development), Celerion, Inc., Parexel International Corporation, Medpace Holdings, Inc., Novotech Health Holdings, Precision for Medicine, Inc. contribute to innovation, geographic expansion, and service delivery in this space through capabilities in biomarker validation, central lab services, bioanalytical testing, and biomarker-informed clinical trial management.
The future of biomarker clinical phase outsourcing services appears promising, driven by the increasing integration of artificial intelligence and machine learning in biomarker analysis. These technologies are expected to enhance data interpretation and accelerate the discovery process. Additionally, the expansion of clinical trials in emerging markets, where regulatory environments are becoming more favorable, will provide new avenues for growth. As the industry adapts to these trends, collaboration between biotech firms and research institutions will further strengthen the market landscape.
| Segment | Sub-Segments |
|---|---|
| By Biomarker Type | Surrogate Endpoint Biomarkers Predictive Biomarkers Prognostic Biomarkers Safety/Toxicity Biomarkers Pharmacodynamic/Response Biomarkers Other Biomarker Types |
| By End-User | Pharmaceutical Companies Biotechnology Companies Academic and Research Institutions Contract Research Organizations (CROs) Other End-Users |
| By Therapeutic Area | Oncology Neurology Cardiology Autoimmune and Inflammatory Diseases Infectious Diseases Other Therapeutic Areas |
| By Service Type | Biomarker Validation Services Biomarker Testing Services (Central Lab & Specialty Lab) Biomarker Sample Analysis (Bioanalytical & Imaging) Data Management, Biostatistics & Reporting Services Regulatory Consulting & Submission Support Other Biomarker Clinical Phase Services |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology Platform | Genomic Technologies (NGS, qPCR, Microarrays) Proteomic Technologies (Mass Spectrometry, Immunoassays) Metabolomic Technologies Flow Cytometry & Cell-Based Assays Imaging-Based Biomarker Technologies Other Technologies |
| By Clinical Phase | Phase I Phase II Phase III Phase IV/Post-Marketing Studies Early Clinical/Translational Studies |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Biomarker Development | 120 | Clinical Research Coordinators, Oncologists |
| Neurology Biomarker Validation | 90 | Neuroscientists, Clinical Trial Managers |
| Cardiovascular Biomarker Testing | 80 | Cardiologists, Laboratory Technicians |
| Biomarker Regulatory Compliance | 60 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Biomarker Research Funding | 70 | Grant Managers, Research Directors |
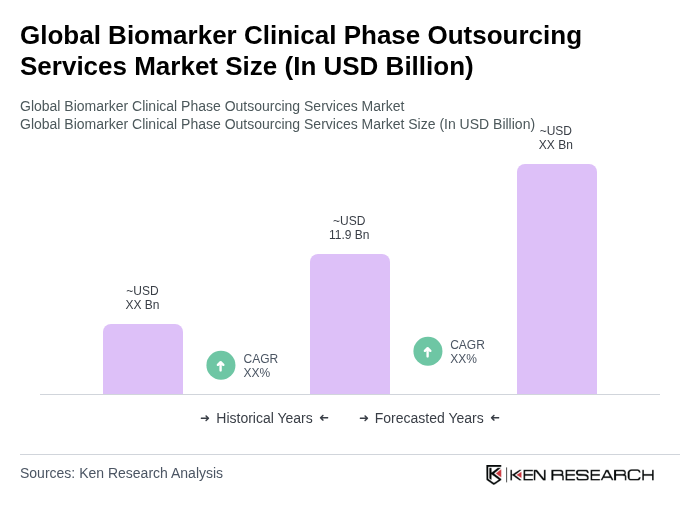
The Global Biomarker Clinical Phase Outsourcing Services Market is valued at approximately USD 11.9 billion, reflecting significant growth driven by advancements in personalized medicine, genomics, and the rising prevalence of chronic diseases requiring biomarker-driven trials.