Region:Global
Author(s):Geetanshi
Product Code:KRAB1637
Pages:85
Published On:January 2026
By Type:The market is segmented into various types of dendritic cell vaccines, including Autologous Dendritic Cell Vaccines, Allogeneic Dendritic Cell Vaccines, Peptide-based Dendritic Cell Vaccines, RNA-based Dendritic Cell Vaccines, and Others. Each type serves different patient needs and treatment protocols, contributing to the overall market dynamics.
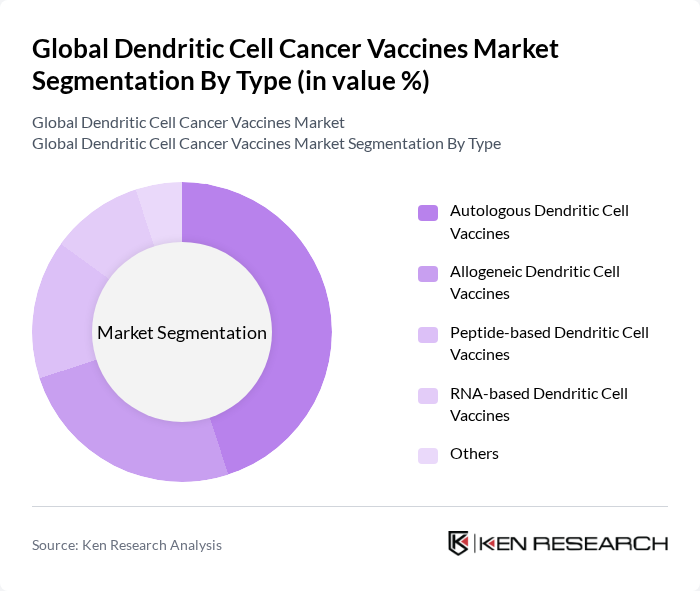
The Autologous Dendritic Cell Vaccines segment is currently leading the market due to their personalized approach, which enhances the immune response against cancer cells. Patients receive vaccines made from their own dendritic cells, which are more effective in eliciting a robust immune response. This segment's growth is further supported by increasing clinical trials demonstrating their efficacy in various cancer types, leading to higher adoption rates among oncologists and patients alike.
By End-User:The market is segmented based on end-users, including Hospitals, Research Institutions, Cancer Treatment Centers, and Others. Each end-user category plays a crucial role in the distribution and administration of dendritic cell cancer vaccines.
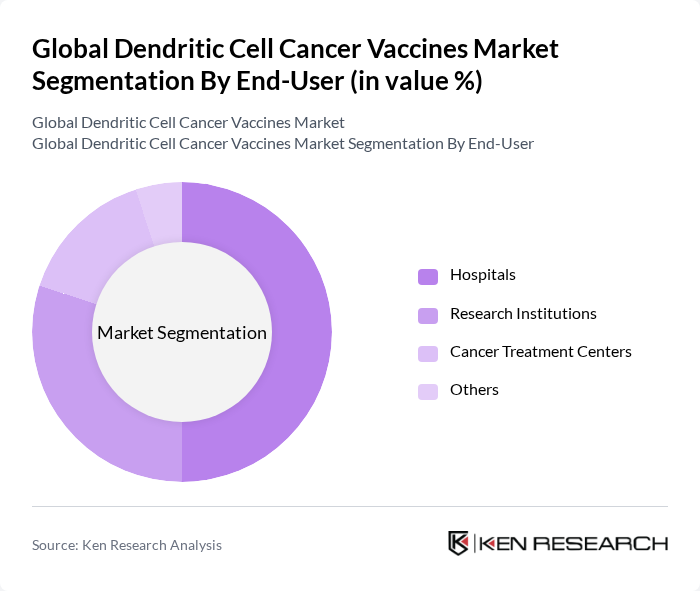
Hospitals dominate the end-user segment due to their comprehensive cancer treatment facilities and access to advanced medical technologies. They are equipped to conduct complex procedures required for the administration of dendritic cell vaccines, making them the primary choice for patients seeking innovative cancer therapies. The increasing number of hospitals adopting these vaccines as part of their treatment protocols further solidifies their leading position in the market.
The Global Dendritic Cell Cancer Vaccines Market is characterized by a dynamic mix of regional and international players. Leading participants such as Dendreon Pharmaceuticals, ImmunoCellular Therapeutics, Northwest Biotherapeutics, Celldex Therapeutics, OncoSec Medical, BioNTech SE, Moderna, Inc., Merck & Co., Inc., AstraZeneca, Novartis AG, Pfizer Inc., GSK plc, Roche Holding AG, Sanofi S.A., Amgen Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of dendritic cell cancer vaccines appears promising, driven by ongoing advancements in personalized medicine and immunotherapy. As research continues to unveil the potential of these vaccines, the integration of artificial intelligence in their development is expected to enhance efficacy and streamline production processes. Furthermore, the increasing collaboration between pharmaceutical companies and biotech firms will likely accelerate innovation, leading to more effective treatment options for cancer patients and improved patient outcomes in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Autologous Dendritic Cell Vaccines Allogeneic Dendritic Cell Vaccines Peptide-based Dendritic Cell Vaccines RNA-based Dendritic Cell Vaccines Others |
| By End-User | Hospitals Research Institutions Cancer Treatment Centers Others |
| By Indication | Melanoma Prostate Cancer Breast Cancer Lung Cancer Others |
| By Route of Administration | Intravenous Subcutaneous Intradermal Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Research Phase | Preclinical Clinical Trials Commercialization Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists Specializing in Immunotherapy | 100 | Medical Doctors, Cancer Treatment Specialists |
| Pharmaceutical Executives in Vaccine Development | 80 | Product Managers, R&D Directors |
| Clinical Trial Coordinators | 60 | Clinical Research Associates, Trial Managers |
| Patients Receiving Dendritic Cell Vaccines | 75 | Patients, Caregivers |
| Healthcare Policy Makers | 50 | Health Economists, Regulatory Affairs Specialists |
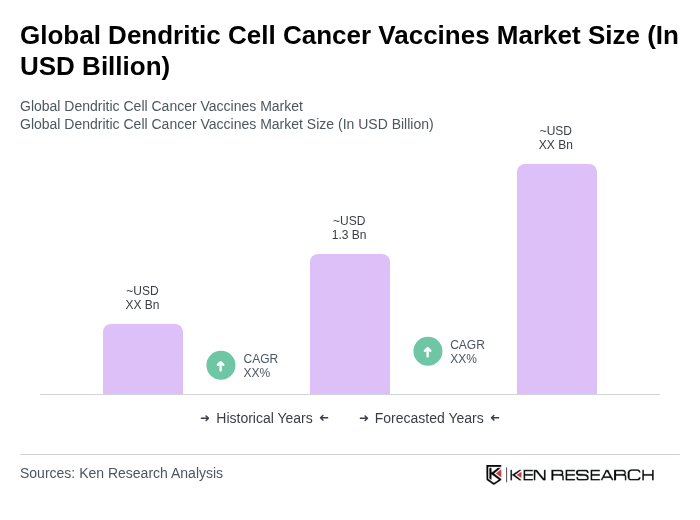
The Global Dendritic Cell Cancer Vaccines Market is valued at approximately USD 1.3 billion, driven by advancements in cancer immunotherapy, increasing cancer prevalence, and enhanced clinical trial infrastructures, particularly in North America and Asia Pacific.