Region:Global
Author(s):Rebecca
Product Code:KRAA2866
Pages:94
Published On:August 2025
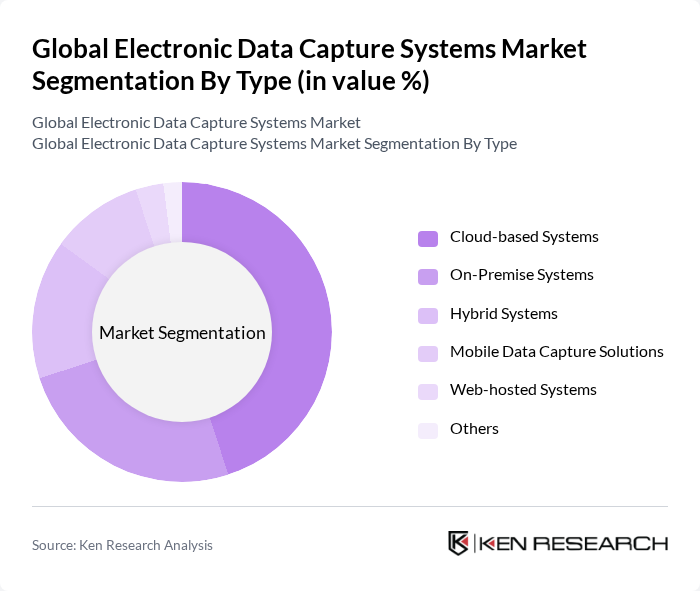
By Type:The market is segmented into Cloud-based Systems, On-Premise Systems, Hybrid Systems, Mobile Data Capture Solutions, Web-hosted Systems, and Others.Cloud-based Systemsare gaining significant traction due to their scalability, cost-effectiveness, and ease of access. The increasing reliance on remote data collection, decentralized clinical trials, and the need for real-time data sharing are driving the adoption of cloud solutions. On-Premise Systems, while still relevant for organizations with strict data residency requirements, are witnessing a decline as the industry shifts toward more flexible and scalable cloud-based options.
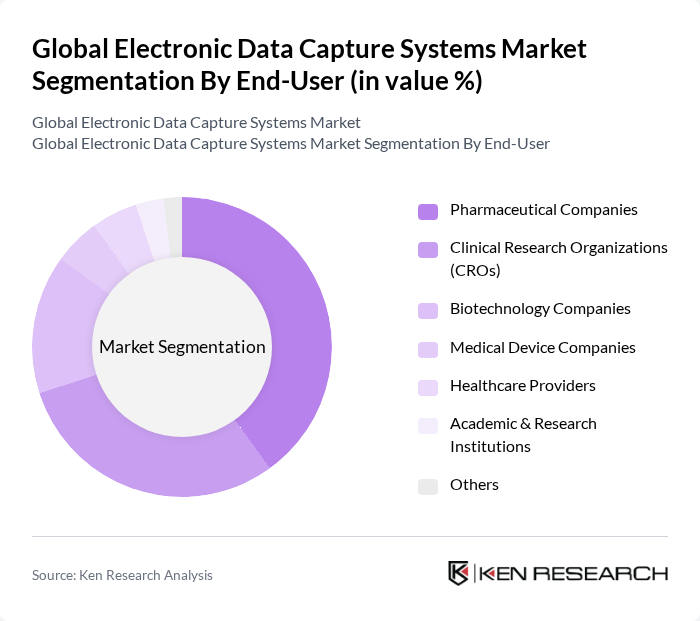
By End-User:The end-user segmentation includes Pharmaceutical Companies, Clinical Research Organizations (CROs), Biotechnology Companies, Medical Device Companies, Healthcare Providers, Academic & Research Institutions, and Others.Pharmaceutical Companiesare the leading end-users, driven by their need for efficient data management in clinical trials and regulatory compliance. CROs also play a significant role, as they require robust electronic data capture systems to manage multiple clinical trials simultaneously, ensuring data integrity and compliance with regulatory standards. The adoption of EDC systems among biotechnology companies and healthcare providers is also increasing, reflecting the broader trend toward digital transformation in clinical research and healthcare delivery.
The Global Electronic Data Capture Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Inc., Oracle Corporation, Veeva Systems Inc., IBM Watson Health, Parexel International Corporation, CRF Health (now Signant Health), Medrio, Inc., Castor EDC, ClinCapture, TrialSpark, eClinical Solutions LLC, BioClinica, Inc. (now part of Clario), Data MATRIX, OmniComm Systems, Inc. (an Anju Software company), nPhase, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of electronic data capture systems is poised for transformative growth, driven by technological advancements and evolving regulatory landscapes. As organizations increasingly embrace decentralized clinical trials, the demand for innovative data capture solutions will rise. Furthermore, the integration of artificial intelligence and machine learning will enhance data analytics capabilities, enabling real-time insights. This evolution will not only improve operational efficiency but also foster a more patient-centric approach to data collection, ultimately reshaping the industry landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Cloud-based Systems On-Premise Systems Hybrid Systems Mobile Data Capture Solutions Web-hosted Systems Others |
| By End-User | Pharmaceutical Companies Clinical Research Organizations (CROs) Biotechnology Companies Medical Device Companies Healthcare Providers Academic & Research Institutions Others |
| By Application | Clinical Trials (Phase I-IV) Patient Registries Observational Studies Post-Market Surveillance Real-World Evidence (RWE) Studies Others |
| By Component | Software Services Hardware Others |
| By Deployment Mode | Public Cloud Private Cloud Hybrid Cloud On-Premise Others |
| By Sales Channel | Direct Sales Distributors Online Sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Management | 60 | Clinical Research Coordinators, Project Managers |
| Pharmaceutical Data Management | 40 | Data Analysts, IT Managers |
| Patient Registry Systems | 50 | Registry Managers, Healthcare Administrators |
| Post-Marketing Surveillance | 45 | Pharmacovigilance Officers, Compliance Managers |
| EDC Software Adoption | 55 | IT Directors, Clinical Operations Managers |
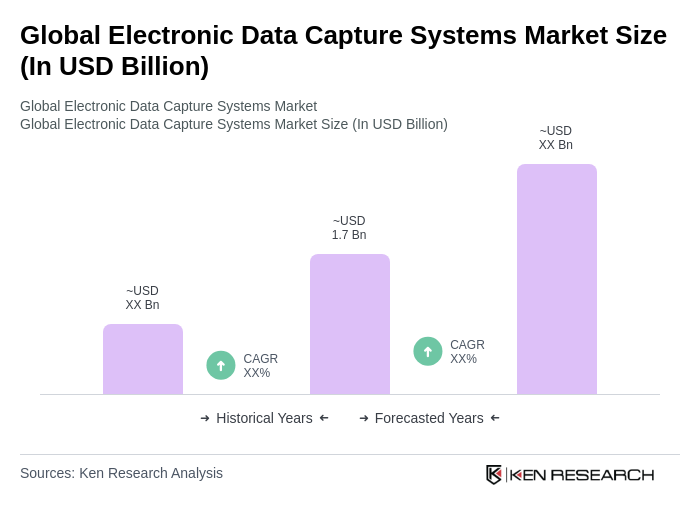
The Global Electronic Data Capture Systems Market is valued at approximately USD 1.7 billion, reflecting significant growth driven by the demand for efficient data management solutions in clinical trials and the adoption of cloud-based technologies.