Region:Global
Author(s):Rebecca
Product Code:KRAC4014
Pages:97
Published On:October 2025
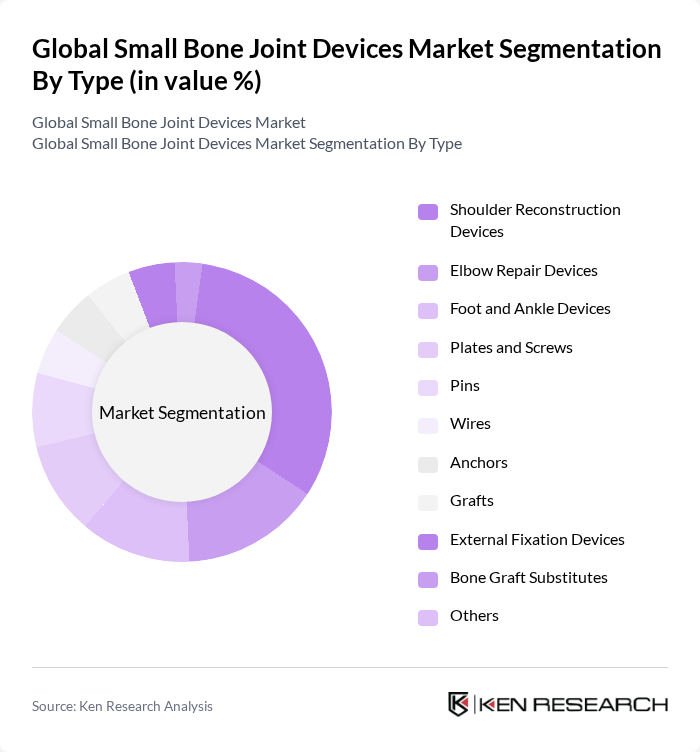
By Type:The market is segmented into various types of devices, including Shoulder Reconstruction Devices, Elbow Repair Devices, Foot and Ankle Devices, Plates and Screws, Pins, Wires, Anchors, Grafts, External Fixation Devices, Bone Graft Substitutes, and Others. Among these, the Shoulder Reconstruction Devices segment is currently leading the market due to the high incidence of shoulder injuries and the growing preference for arthroscopic surgeries, which utilize these devices extensively.
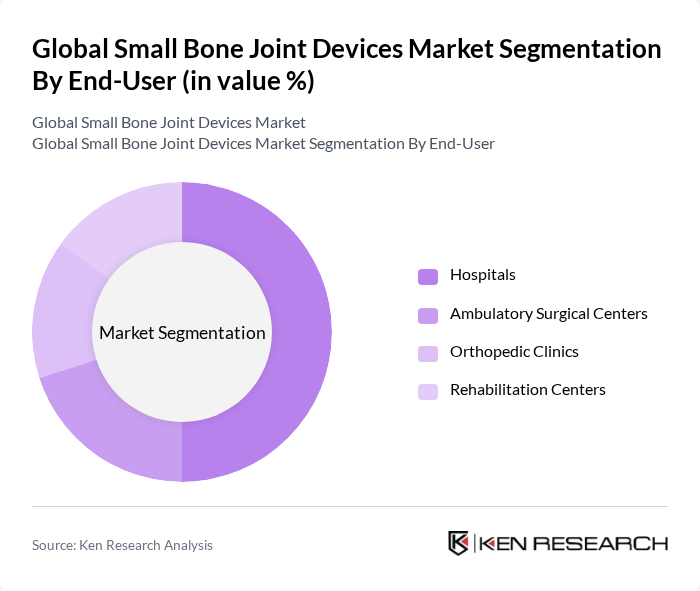
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Orthopedic Clinics, and Rehabilitation Centers. Hospitals are the leading end-user segment, primarily due to their capacity to perform complex surgeries and the availability of advanced surgical equipment and specialists. The increasing number of orthopedic procedures performed in hospitals further solidifies their dominant position in the market.
The Global Small Bone Joint Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stryker Corporation, DePuy Synthes (Johnson & Johnson), Zimmer Biomet Holdings, Inc., Medtronic plc, Smith & Nephew plc, Arthrex, Inc., NuVasive, Inc., Orthofix Medical Inc., Aesculap, Inc. (B. Braun Melsungen AG), B. Braun Melsungen AG, CONMED Corporation, RTI Surgical Holdings, Inc., Integra LifeSciences Holdings Corporation, DJO Global, Inc., Tecomet, Inc., Conventus Flower Orthopedics Corporation, Z-Medical GmbH + Co. KG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the small bone joint devices market appears promising, driven by technological advancements and an increasing focus on patient-centered care. Innovations such as 3D printing and digital surgical planning are expected to enhance device customization and surgical precision. Additionally, the rise of outpatient procedures will likely lead to increased adoption of minimally invasive techniques, further propelling market growth. Companies that invest in research and development will be well-positioned to capitalize on these trends and meet evolving patient needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Shoulder Reconstruction Devices Elbow Repair Devices Foot and Ankle Devices Plates and Screws Pins Wires Anchors Grafts External Fixation Devices Bone Graft Substitutes Others |
| By End-User | Hospitals Ambulatory Surgical Centers Orthopedic Clinics Rehabilitation Centers |
| By Application | Trauma Surgery Sports Medicine Joint Reconstruction Arthroscopy Foot and Ankle Orthopedic Procedures Hand Orthopedic Procedures Shoulder Orthopedic Procedures |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Material | Metal Polymer Composite Ceramic |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Price Range | Low Medium High |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Surgeons | 60 | Orthopedic Surgeons, Medical Directors |
| Hospital Procurement Managers | 40 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Distributors | 40 | Sales Managers, Distribution Executives |
| Clinical Researchers | 45 | Clinical Researchers, Product Development Specialists |
| Regulatory Affairs Professionals | 50 | Regulatory Affairs Managers, Compliance Officers |
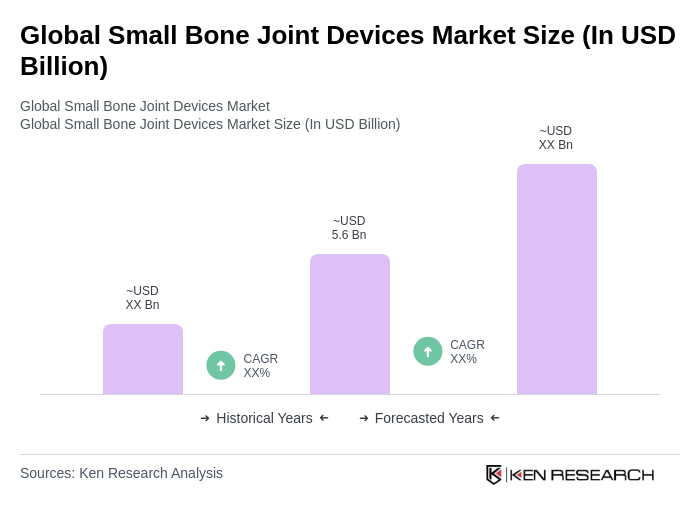
The Global Small Bone Joint Devices Market is valued at approximately USD 5.6 billion, reflecting a significant growth driven by the rising prevalence of orthopedic disorders and advancements in surgical techniques, particularly minimally invasive surgery.