About the Report
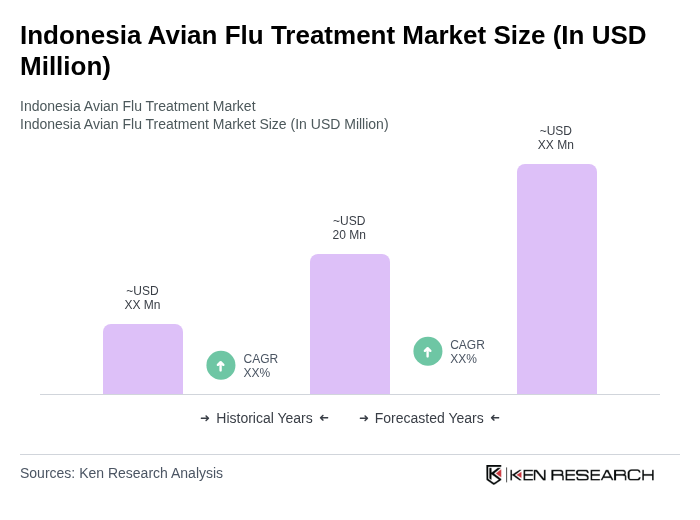
Base Year 2024Indonesia Avian Flu Treatment Market Overview
- The Indonesia Avian Flu Treatment Market is valued at USD 20 million, based on a five-year historical analysis.This growth is primarily driven by the increasing prevalence of avian influenza outbreaks in poultry and sporadic human infections, which necessitate effective treatment and control options.The rising awareness of public health, expanded surveillance for zoonotic influenza, and the need for preventive measures against zoonotic diseases have further fueled the demand for avian flu vaccines, antivirals, and biosecurity interventions in the region.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their high population density, concentration of healthcare facilities, and strong linkages to major poultry production and trading corridors in Java.These urban centers are critical for the distribution of avian flu treatments, as they serve as hubs for both human and veterinary healthcare services, national vaccine distribution (including state-owned manufacturers), and rapid outbreak response networks, ensuring faster containment and more effective management of public health.
- In response to the avian flu threat, the Indonesian government has implemented regulations and control programs that include mandatory vaccination of poultry in high-risk areas and integrated disease control measures. The key framework is the National Strategic Plan for Avian Influenza Control and Pandemic Influenza Preparedness, reinforced through instruments such as the Minister of Agriculture Regulation No. 28/Permentan/OT.140/5/2008 on the Control of Avian Influenza in Poultry, issued by the Ministry of Agriculture, which sets requirements for vaccination, surveillance, reporting, movement control, and biosecurity in commercial and backyard flocks.This regulatory framework aims to reduce the incidence of the disease in birds, thereby minimizing the risk of transmission to humans, and compliance with these measures is crucial for poultry farmers, veterinary services, and healthcare providers involved in avian flu prevention and treatment.
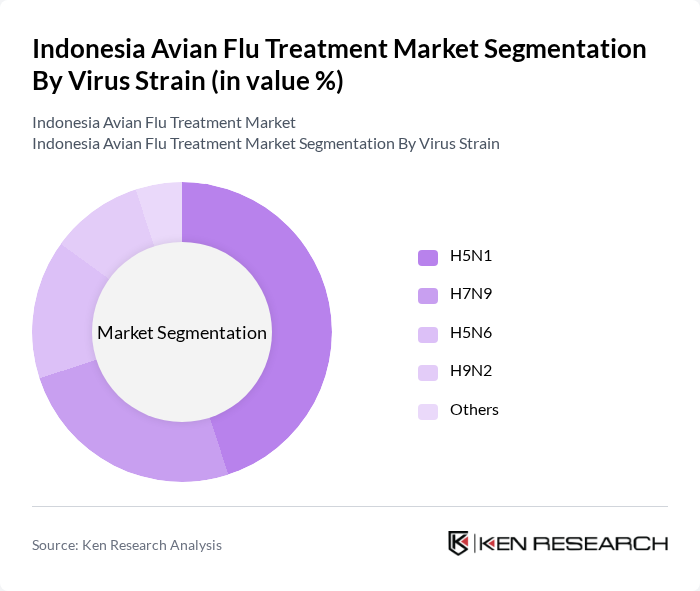
Indonesia Avian Flu Treatment Market Segmentation
By Virus Strain:The market is segmented based on various virus strains that cause avian influenza. The primary strains include H5N1, H7N9, H5N6, H9N2, and others. In Indonesia, highly pathogenic H5Nx clades (historically H5N1 and later reassortant H5 variants) remain the dominant concern in poultry and have been responsible for most reported human cases, driving demand for targeted vaccines and antiviral strategies.The increasing awareness of the risks associated with these strains, along with surveillance of emerging subtypes including H9N2 in regional poultry systems, has prompted healthcare and veterinary providers to focus on strain-specific vaccines, improved diagnostics, and rapid-response treatment protocols.
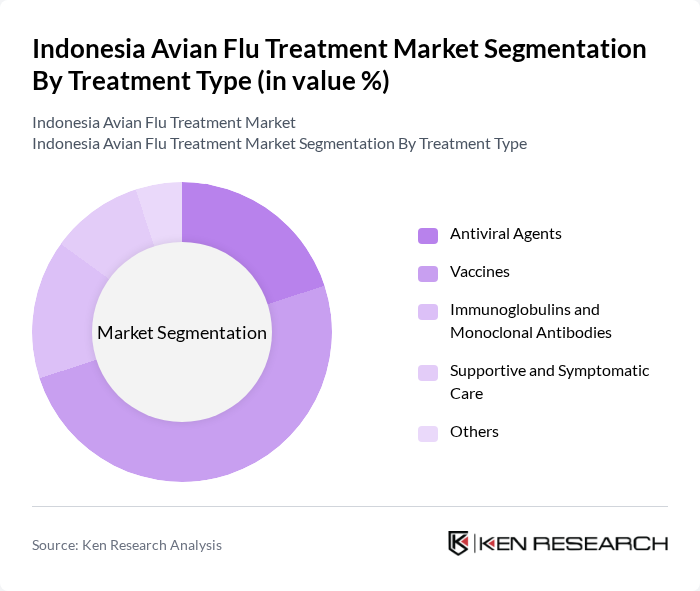
By Treatment Type:The treatment options for avian influenza are categorized into antiviral agents, vaccines, immunoglobulins and monoclonal antibodies, supportive and symptomatic care, and others. Vaccines are particularly significant in this market, as poultry vaccination is central to Indonesia’s avian influenza control strategy and underpins demand for inactivated and recombinant vaccines tailored to circulating H5 strains.In parallel, global and regional guidelines emphasize early use of neuraminidase-inhibitor antivirals for human cases, along with robust supportive and symptomatic care, which together with growing R&D in monoclonal antibodies and combination therapies are expanding the overall treatment landscape.
Indonesia Avian Flu Treatment Market Competitive Landscape
The Indonesia Avian Flu Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Bio Farma (Persero), PT Kimia Farma Tbk, PT Indofarma Tbk, PT Sanbe Farma, PT Merck Sharp & Dohme Pharma, PT GlaxoSmithKline Indonesia, PT Novartis Indonesia, PT Pfizer Indonesia, PT Johnson & Johnson Indonesia, PT Bayer Indonesia, PT Sandoz Indonesia, PT Abbott Indonesia, PT AstraZeneca Indonesia, PT Roche Indonesia, PT Takeda Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Avian Flu Treatment Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Avian Flu Cases:The rise in avian flu cases in Indonesia has been alarming, with over 250 reported outbreaks in poultry in the recent past, according to the Ministry of Agriculture. This surge has heightened the urgency for effective treatment options, driving demand for avian flu treatments. The World Health Organization reported that Indonesia has one of the highest incidences of H5N1 avian influenza, necessitating increased healthcare interventions and resources to combat the disease effectively.
- Government Initiatives for Disease Control:The Indonesian government has allocated approximately IDR 2 trillion (around USD 133 million) for avian flu control measures in the upcoming year. This funding supports vaccination programs and public health campaigns aimed at reducing the spread of the virus among poultry. Enhanced regulatory frameworks and partnerships with international health organizations are also being established to ensure effective disease management, thereby fostering a conducive environment for the avian flu treatment market to thrive.
- Advancements in Treatment Technologies:The avian flu treatment landscape is evolving, with significant investments in research and development. In the recent past, the Indonesian government partnered with local biotech firms, resulting in the development of three new antiviral drugs specifically targeting avian influenza. These innovations are expected to improve treatment efficacy and accessibility, thereby increasing the overall market potential. The focus on biotechnology is projected to enhance the country's capacity to respond to future outbreaks effectively.
Market Challenges
- Limited Access to Healthcare Facilities:In rural areas of Indonesia, access to healthcare facilities remains a significant challenge, with only 35% of the population having access to adequate healthcare services. This limitation hampers timely diagnosis and treatment of avian flu cases, leading to higher mortality rates among affected poultry and potential zoonotic transmission to humans. The disparity in healthcare infrastructure creates barriers to effective disease management and treatment delivery.
- High Cost of Treatment Options:The cost of avian flu treatment options can be prohibitive, with antiviral medications priced at approximately IDR 600,000 (USD 40) per treatment course. This expense is a significant burden for many poultry farmers, particularly smallholders, who may struggle to afford necessary treatments. The financial constraints limit the adoption of effective treatment protocols, ultimately affecting the overall health of poultry populations and increasing the risk of disease spread.
Indonesia Avian Flu Treatment Market Future Outlook
The future of the avian flu treatment market in Indonesia appears promising, driven by ongoing government initiatives and advancements in biotechnology. As healthcare infrastructure expands, particularly in rural areas, access to treatment is expected to improve significantly. Additionally, the focus on developing affordable treatment options will likely enhance market penetration. Collaborations with international health organizations will further bolster research efforts, paving the way for innovative solutions to combat avian influenza effectively in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Indonesian government plans to invest IDR 3 trillion (USD 200 million) in healthcare infrastructure by the mid-2020s. This investment will enhance access to treatment facilities, particularly in underserved regions, creating a favorable environment for avian flu treatment providers to expand their services and reach more patients effectively.
- Research and Development in Vaccine Production:With a growing emphasis on vaccine development, Indonesia is set to increase its R&D budget by 25% in the upcoming year. This focus on innovative vaccine solutions presents a significant opportunity for pharmaceutical companies to collaborate with local institutions, potentially leading to breakthroughs in avian flu prevention and treatment, thereby enhancing public health outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Virus Strain | H5N1 H7N9 H5N6 H9N2 Others |
| By Treatment Type | Antiviral Agents Vaccines Immunoglobulins and Monoclonal Antibodies Supportive and Symptomatic Care Others |
| By Vaccine Category | Inactivated Vaccines Live Attenuated / Recombinant Vaccines Subunit and DNA Vaccines Bivalent and Multivalent Vaccines Others |
| By Application | Veterinary Human Health Research and Surveillance Others |
| By End-User | Poultry Farms Veterinary Clinics and Hospitals Governmental and Public Health Organizations Research Institutes Others |
| By Distribution Channel | Direct Tenders Distributor / Wholesaler Channel Retail and Hospital Pharmacies Online Pharmacies and E-Procurement Others |
| By Region | Java Sumatra Bali and Nusa Tenggara Kalimantan Sulawesi Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Ministry of Agriculture)
Pharmaceutical Manufacturers
Veterinary Product Suppliers
Healthcare Providers and Hospitals
Animal Health Organizations
Biotechnology Firms
Insurance Companies
Players Mentioned in the Report:
PT Bio Farma (Persero)
PT Kimia Farma Tbk
PT Indofarma Tbk
PT Sanbe Farma
PT Merck Sharp & Dohme Pharma
PT GlaxoSmithKline Indonesia
PT Novartis Indonesia
PT Pfizer Indonesia
PT Johnson & Johnson Indonesia
PT Bayer Indonesia
PT Sandoz Indonesia
PT Abbott Indonesia
PT AstraZeneca Indonesia
PT Roche Indonesia
PT Takeda Indonesia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Avian Flu Treatment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Avian Flu Treatment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Avian Flu Treatment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Avian Flu cases
3.1.2 Government initiatives for disease control
3.1.3 Rising awareness about avian diseases
3.1.4 Advancements in treatment technologies
3.2 Market Challenges
3.2.1 Limited access to healthcare facilities
3.2.2 High cost of treatment options
3.2.3 Regulatory hurdles in drug approval
3.2.4 Lack of public awareness and education
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Development of affordable treatment options
3.3.3 Collaborations with international health organizations
3.3.4 Research and development in vaccine production
3.4 Market Trends
3.4.1 Increasing investment in biotechnology
3.4.2 Shift towards preventive healthcare measures
3.4.3 Growth of telemedicine in treatment delivery
3.4.4 Rising demand for organic and natural treatments
3.5 Government Regulation
3.5.1 Implementation of vaccination programs
3.5.2 Strict monitoring of poultry health
3.5.3 Regulations on drug approval processes
3.5.4 Guidelines for public health awareness campaigns
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Avian Flu Treatment Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Avian Flu Treatment Market Segmentation
8.1 By Virus Strain
8.1.1 H5N1
8.1.2 H7N9
8.1.3 H5N6
8.1.4 H9N2
8.1.5 Others
8.2 By Treatment Type
8.2.1 Antiviral Agents
8.2.2 Vaccines
8.2.3 Immunoglobulins and Monoclonal Antibodies
8.2.4 Supportive and Symptomatic Care
8.2.5 Others
8.3 By Vaccine Category
8.3.1 Inactivated Vaccines
8.3.2 Live Attenuated / Recombinant Vaccines
8.3.3 Subunit and DNA Vaccines
8.3.4 Bivalent and Multivalent Vaccines
8.3.5 Others
8.4 By Application
8.4.1 Veterinary
8.4.2 Human Health
8.4.3 Research and Surveillance
8.4.4 Others
8.5 By End-User
8.5.1 Poultry Farms
8.5.2 Veterinary Clinics and Hospitals
8.5.3 Governmental and Public Health Organizations
8.5.4 Research Institutes
8.5.5 Others
8.6 By Distribution Channel
8.6.1 Direct Tenders
8.6.2 Distributor / Wholesaler Channel
8.6.3 Retail and Hospital Pharmacies
8.6.4 Online Pharmacies and E-Procurement
8.6.5 Others
8.7 By Region
8.7.1 Java
8.7.2 Sumatra
8.7.3 Bali and Nusa Tenggara
8.7.4 Kalimantan
8.7.5 Sulawesi
8.7.6 Others
9. Indonesia Avian Flu Treatment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Share in Indonesia (%)
9.2.3 Indonesia Avian Flu Treatment Revenue (USD Million)
9.2.4 3-year Revenue CAGR (%) in Avian Flu Segment
9.2.5 R&D Intensity (% of Revenue)
9.2.6 EBITDA Margin (%) for Avian Flu Portfolio
9.2.7 Average Selling Price Positioning (vs. Market Index)
9.2.8 Geographic Coverage (No. of Provinces Served)
9.2.9 Distribution Mix (Direct Tenders vs. Distributors vs. Retail, %)
9.2.10 Vaccine and Antiviral Portfolio Breadth (No. of SKUs)
9.2.11 Tender Win Rate (%) in Public Sector
9.2.12 Inventory Turnover (Times per Year)
9.2.13 Days Sales Outstanding (DSO)
9.2.14 Regulatory / WHO Prequalification Status
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Bio Farma (Persero)
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Indofarma Tbk
9.5.4 PT Sanbe Farma
9.5.5 PT Merck Sharp & Dohme Pharma
9.5.6 PT GlaxoSmithKline Indonesia
9.5.7 PT Novartis Indonesia
9.5.8 PT Pfizer Indonesia
9.5.9 PT Johnson & Johnson Indonesia
9.5.10 PT Bayer Indonesia
9.5.11 PT Sandoz Indonesia
9.5.12 PT Abbott Indonesia
9.5.13 PT AstraZeneca Indonesia
9.5.14 PT Roche Indonesia
9.5.15 PT Takeda Indonesia
10. Indonesia Avian Flu Treatment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Agriculture
10.1.3 Ministry of Environment
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research Initiatives
10.2.3 Budget Allocation for Public Health Campaigns
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Healthcare Providers
10.3.2 Patients
10.3.3 Government Agencies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Accessibility of Treatments
10.4.3 Training and Support Needs
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Effectiveness of Treatments
10.5.2 Cost Savings Analysis
10.5.3 Expansion into New Markets
10.5.4 Others
11. Indonesia Avian Flu Treatment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health reports and avian flu statistics from the Indonesian Ministry of Health
- Review of published studies and articles in medical journals focusing on avian flu treatment protocols
- Examination of market reports from health organizations and NGOs involved in avian flu research
Primary Research
- Interviews with healthcare professionals specializing in infectious diseases and avian flu treatment
- Surveys conducted with pharmaceutical companies involved in the development of avian flu medications
- Field interviews with veterinarians and poultry health experts regarding treatment practices
Validation & Triangulation
- Cross-validation of data through multiple sources including government health statistics and industry reports
- Triangulation of findings from expert interviews and published literature
- Sanity checks through feedback from a panel of healthcare experts and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the avian flu treatment market size based on national healthcare expenditure data
- Segmentation of the market by treatment type, including antiviral medications and vaccines
- Incorporation of government health initiatives aimed at controlling avian flu outbreaks
Bottom-up Modeling
- Collection of sales data from pharmaceutical companies producing avian flu treatments
- Estimation of treatment volumes based on reported cases and vaccination rates
- Cost analysis of treatment options to establish a pricing model for the market
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as poultry population growth and public health policies
- Scenario modeling based on potential future outbreaks and vaccine development timelines
- Baseline, optimistic, and pessimistic forecasts through 2030 based on historical trends
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Infectious Disease Specialists, General Practitioners |
| Pharmaceutical Companies | 90 | Product Managers, R&D Directors |
| Poultry Health Experts | 70 | Veterinarians, Animal Health Researchers |
| Government Health Officials | 60 | Public Health Administrators, Policy Makers |
| NGO Representatives | 50 | Health Program Coordinators, Research Analysts |
Frequently Asked Questions
What is the current value of the Indonesia Avian Flu Treatment Market?
The Indonesia Avian Flu Treatment Market is valued at approximately USD 20 million, driven by the increasing prevalence of avian influenza outbreaks in poultry and sporadic human infections, necessitating effective treatment and control options.