About the Report
Base Year 2024Indonesia Cephalosporin Drugs Market Overview
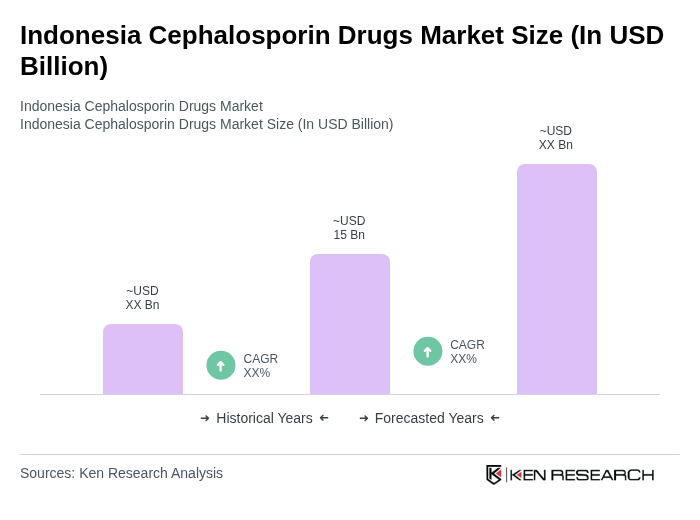
- The Indonesia Cephalosporin Drugs Market is valued at USD 15 billion, based on recent analysis. Growth is driven by expanding universal healthcare coverage under the JKN scheme, which has substantially increased antibiotic consumption including cephalosporins, and by broader improvements in healthcare infrastructure and access. Additionally, rising infectious disease burden and better diagnostics are supporting increased cephalosporin demand.
- Key players in this market include Kalbe Farma, Kimia Farma, and Bio Farma, which leverage extensive distribution networks and integrated manufacturing capabilities to supply both generic and injectable antibiotics, fostering market leadership. Indonesia’s rising role in Southeast Asia’s cephalosporin intermediate consumption is also supported by secondary manufacturing growth in the region including the country, complementing global supply dynamics.
- In 2024, Indonesia's government announced a strategic plan to enhance antibiotic stewardship through the National Action Plan on AMR and One Health efforts, which include surveillance expansion via GLASS enrollment, multisectoral stewardship programs, and regulations such as banning antibiotics for livestock use to curb resistance.
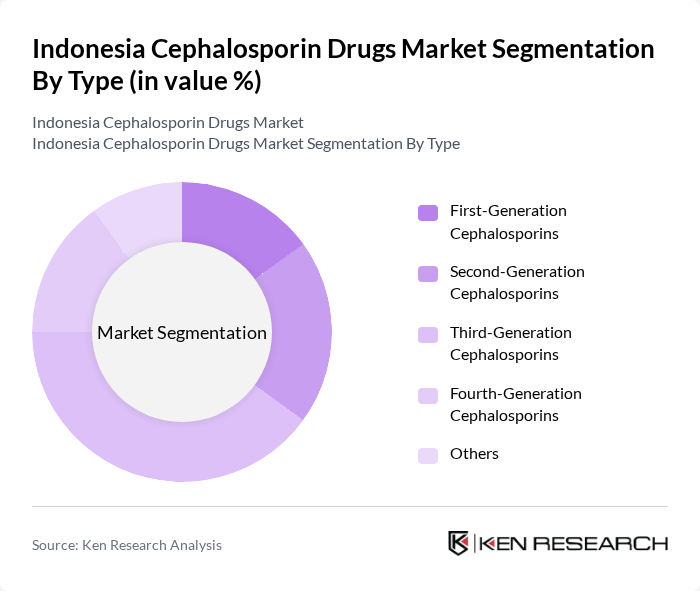
Indonesia Cephalosporin Drugs Market Segmentation
By Type:The cephalosporin drugs market is segmented into various types, including first-generation, second-generation, third-generation, fourth-generation, and others. Among these, third-generation cephalosporins dominate the market due to their broad-spectrum activity against gram-negative bacteria, making them a preferred choice in hospitals and clinics. The increasing prevalence of infections and the need for effective treatment options further bolster the demand for this sub-segment.
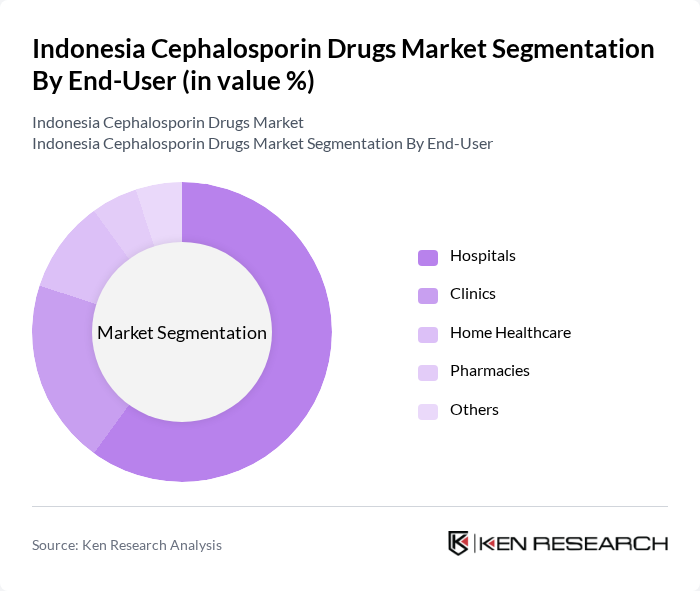
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, pharmacies, and others. Hospitals are the leading end-users of cephalosporin drugs, primarily due to the high volume of patients requiring antibiotic treatment for various infections. The increasing number of hospital admissions and the growing focus on infection control measures contribute to the dominance of this segment.
Indonesia Cephalosporin Drugs Market Competitive Landscape
The Indonesia Cephalosporin Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Kalbe Farma Tbk, PT. Kimia Farma Tbk, PT. Indofarma Tbk, PT. Sanbe Farma, PT. Merck Sharp & Dohme, PT. Pfizer Indonesia, PT. Novartis Indonesia, PT. Roche Indonesia, PT. Sandoz Indonesia, PT. GlaxoSmithKline Indonesia, PT. Bayer Indonesia, PT. AstraZeneca Indonesia, PT. Abbott Indonesia, PT. Darya-Varia Laboratoria Tbk, PT. Soho Global Health contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Cephalosporin Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Bacterial Infections:The rise in bacterial infections in Indonesia is significant, with the World Health Organization reporting over 1.5 million cases annually. This surge is attributed to urbanization and lifestyle changes, leading to increased healthcare demands. The healthcare expenditure in Indonesia is projected to reach IDR 1,200 trillion (approximately USD 81 billion) in future, driving the need for effective antibiotic treatments, including cephalosporins, to combat these infections.
- Rising Demand for Advanced Antibiotics:The demand for advanced antibiotics, particularly cephalosporins, is escalating due to their effectiveness against resistant bacterial strains. In future, the Indonesian government plans to allocate IDR 50 trillion (around USD 3.3 billion) for antibiotic procurement and distribution. This investment reflects a commitment to enhancing treatment options, thereby increasing the market for cephalosporin drugs as healthcare providers seek reliable solutions for complex infections.
- Expanding Healthcare Infrastructure:Indonesia's healthcare infrastructure is undergoing rapid expansion, with the government aiming to build 1,000 new healthcare facilities in future. This initiative is part of a broader strategy to improve healthcare access, particularly in rural areas. As healthcare facilities increase, the demand for cephalosporin drugs is expected to rise, supported by a projected increase in healthcare spending to IDR 1,500 trillion (approximately USD 100 billion) in future, enhancing drug availability.
Market Challenges
- Stringent Regulatory Requirements:The Indonesian pharmaceutical market faces stringent regulatory requirements, which can delay the approval of new cephalosporin drugs. The National Agency of Drug and Food Control (BPOM) has implemented rigorous testing and approval processes, which can take up to 24 months. This regulatory landscape poses challenges for companies seeking to introduce innovative products, potentially limiting market growth and access to new treatments for patients.
- High Competition Among Pharmaceutical Companies:The cephalosporin market in Indonesia is characterized by intense competition, with over 30 local and international companies vying for market share. This competitive environment leads to price wars and reduced profit margins. In future, the market is expected to see a 15% increase in the number of generic cephalosporin products, further intensifying competition and making it challenging for companies to differentiate their offerings effectively.
Indonesia Cephalosporin Drugs Market Future Outlook
The future of the Indonesia cephalosporin drugs market appears promising, driven by increasing healthcare investments and a growing focus on antibiotic stewardship. As the government enhances healthcare access and infrastructure, the demand for effective antibiotics will likely rise. Additionally, the integration of digital health technologies is expected to streamline drug distribution and patient management, fostering a more efficient healthcare system. These trends indicate a robust market environment for cephalosporin drugs in the coming years.
Market Opportunities
- Growth in Generic Cephalosporin Drugs:The increasing acceptance of generic drugs presents a significant opportunity for market players. With the Indonesian government promoting generic medications, the market for generic cephalosporins is projected to grow, potentially reaching IDR 20 trillion (approximately USD 1.3 billion) in future, allowing companies to capture a larger share of the market.
- Collaborations with Local Manufacturers:Collaborating with local manufacturers can enhance market penetration and reduce production costs. In future, partnerships with local firms are expected to increase by 30%, enabling foreign companies to leverage local expertise and distribution networks, thus expanding their reach in the Indonesian cephalosporin market effectively.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | First-Generation Cephalosporins Second-Generation Cephalosporins Third-Generation Cephalosporins Fourth-Generation Cephalosporins Others |
| By End-User | Hospitals Clinics Home Healthcare Pharmacies Others |
| By Distribution Channel | Direct Sales Wholesalers Online Pharmacies Retail Pharmacies Others |
| By Formulation | Oral Injectable Topical Others |
| By Therapeutic Area | Respiratory Infections Skin Infections Urinary Tract Infections Others |
| By Patient Demographics | Pediatric Adult Geriatric Others |
| By Region | Java Sumatra Kalimantan Sulawesi Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Agency of Drug and Food Control, Ministry of Health)
Pharmaceutical Manufacturers and Producers
Distributors and Wholesalers
Healthcare Providers and Hospitals
Pharmacy Chains and Retail Pharmacies
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Health Insurance Companies
Players Mentioned in the Report:
PT. Kalbe Farma Tbk
PT. Kimia Farma Tbk
PT. Indofarma Tbk
PT. Sanbe Farma
PT. Merck Sharp & Dohme
PT. Pfizer Indonesia
PT. Novartis Indonesia
PT. Roche Indonesia
PT. Sandoz Indonesia
PT. GlaxoSmithKline Indonesia
PT. Bayer Indonesia
PT. AstraZeneca Indonesia
PT. Abbott Indonesia
PT. Darya-Varia Laboratoria Tbk
PT. Soho Global Health
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Cephalosporin Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Cephalosporin Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Cephalosporin Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of bacterial infections
3.1.2 Rising demand for advanced antibiotics
3.1.3 Expanding healthcare infrastructure
3.1.4 Government initiatives to improve healthcare access
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition among pharmaceutical companies
3.2.3 Rising costs of drug development
3.2.4 Issues related to antibiotic resistance
3.3 Market Opportunities
3.3.1 Growth in generic cephalosporin drugs
3.3.2 Expansion into rural healthcare markets
3.3.3 Collaborations with local manufacturers
3.3.4 Increasing investment in R&D for new formulations
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Adoption of digital health technologies
3.4.3 Focus on sustainable and eco-friendly production
3.4.4 Growing awareness of antibiotic stewardship
3.5 Government Regulation
3.5.1 New drug approval processes
3.5.2 Pricing regulations for pharmaceuticals
3.5.3 Policies promoting local manufacturing
3.5.4 Guidelines for antibiotic usage in healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Cephalosporin Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Cephalosporin Drugs Market Segmentation
8.1 By Type
8.1.1 First-Generation Cephalosporins
8.1.2 Second-Generation Cephalosporins
8.1.3 Third-Generation Cephalosporins
8.1.4 Fourth-Generation Cephalosporins
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Home Healthcare
8.2.4 Pharmacies
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Wholesalers
8.3.3 Online Pharmacies
8.3.4 Retail Pharmacies
8.3.5 Others
8.4 By Formulation
8.4.1 Oral
8.4.2 Injectable
8.4.3 Topical
8.4.4 Others
8.5 By Therapeutic Area
8.5.1 Respiratory Infections
8.5.2 Skin Infections
8.5.3 Urinary Tract Infections
8.5.4 Others
8.6 By Patient Demographics
8.6.1 Pediatric
8.6.2 Adult
8.6.3 Geriatric
8.6.4 Others
8.7 By Region
8.7.1 Java
8.7.2 Sumatra
8.7.3 Kalimantan
8.7.4 Sulawesi
8.7.5 Others
9. Indonesia Cephalosporin Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Diversification Index
9.2.8 Distribution Network Efficiency
9.2.9 R&D Investment Ratio
9.2.10 Brand Equity Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT. Kalbe Farma Tbk
9.5.2 PT. Kimia Farma Tbk
9.5.3 PT. Indofarma Tbk
9.5.4 PT. Sanbe Farma
9.5.5 PT. Merck Sharp & Dohme
9.5.6 PT. Pfizer Indonesia
9.5.7 PT. Novartis Indonesia
9.5.8 PT. Roche Indonesia
9.5.9 PT. Sandoz Indonesia
9.5.10 PT. GlaxoSmithKline Indonesia
9.5.11 PT. Bayer Indonesia
9.5.12 PT. AstraZeneca Indonesia
9.5.13 PT. Abbott Indonesia
9.5.14 PT. Darya-Varia Laboratoria Tbk
9.5.15 PT. Soho Global Health
10. Indonesia Cephalosporin Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Facilities
10.2.2 Pharmaceutical Manufacturing
10.2.3 Research Institutions
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Pharmacies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Healthcare Professionals
10.4.2 Patients
10.4.3 Pharmacists
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvement
10.5.3 Market Expansion Opportunities
10.5.4 Others
11. Indonesia Cephalosporin Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical associations in Indonesia
- Review of government publications on drug approvals and regulations
- Examination of market trends through academic journals and healthcare publications
Primary Research
- Interviews with key opinion leaders in the pharmaceutical sector
- Surveys conducted with healthcare professionals and pharmacists
- Focus group discussions with patients and healthcare providers regarding cephalosporin usage
Validation & Triangulation
- Cross-validation of data from multiple sources including sales data and market reports
- Triangulation of findings from primary interviews and secondary research insights
- Sanity checks through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size in Indonesia as a baseline
- Segmentation of the cephalosporin market by therapeutic class and sales channels
- Incorporation of growth rates from historical data and market forecasts
Bottom-up Modeling
- Collection of sales data from major pharmaceutical distributors and manufacturers
- Estimation of average pricing and volume sold for various cephalosporin products
- Analysis of prescription patterns and hospital procurement data
Forecasting & Scenario Analysis
- Development of market forecasts based on demographic trends and healthcare spending
- Scenario analysis considering potential regulatory changes and market entry of new drugs
- Creation of baseline, optimistic, and pessimistic growth scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacists | 100 | Pharmacy Directors, Clinical Pharmacists |
| General Practitioners | 80 | Family Physicians, General Practitioners |
| Pharmaceutical Distributors | 70 | Sales Managers, Distribution Coordinators |
| Healthcare Policy Makers | 50 | Health Ministry Officials, Regulatory Affairs Specialists |
| Patients Using Cephalosporins | 90 | Chronic Illness Patients, Post-operative Patients |
Frequently Asked Questions
What is the current value of the Indonesia Cephalosporin Drugs Market?
The Indonesia Cephalosporin Drugs Market is valued at approximately USD 15 billion, driven by increased healthcare access, rising infectious disease burdens, and improved diagnostics, which have collectively boosted the demand for cephalosporin antibiotics.