Region:Asia
Author(s):Shubham
Product Code:KRAC3563
Pages:82
Published On:October 2025
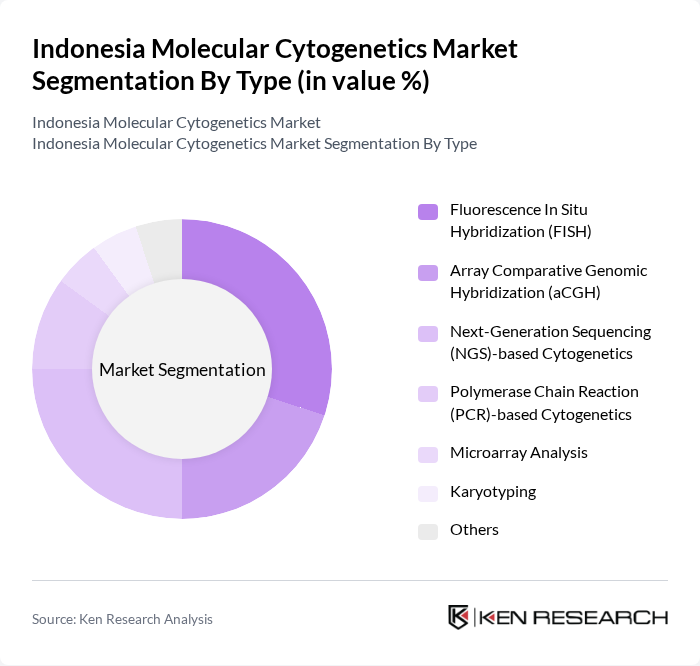
By Type:The market is segmented into various types of molecular cytogenetics techniques, including Fluorescence In Situ Hybridization (FISH), Array Comparative Genomic Hybridization (aCGH), Next-Generation Sequencing (NGS)-based Cytogenetics, Polymerase Chain Reaction (PCR)-based Cytogenetics, Microarray Analysis, Karyotyping, and Others. Among these, FISH and NGS-based techniques are gaining significant traction due to their accuracy and efficiency in detecting chromosomal abnormalities.
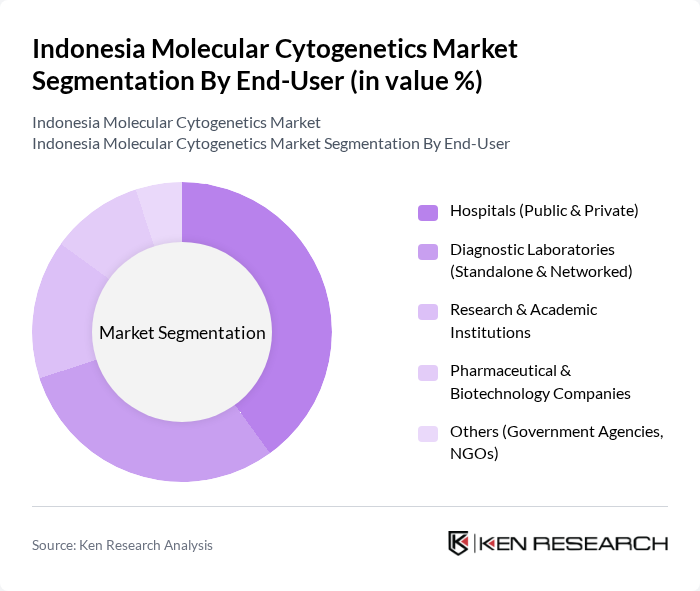
By End-User:The end-user segmentation includes Hospitals (Public & Private), Diagnostic Laboratories (Standalone & Networked), Research & Academic Institutions, Pharmaceutical & Biotechnology Companies, and Others (Government Agencies, NGOs). Hospitals are the primary end-users, driven by the increasing demand for genetic testing in clinical settings and the need for accurate diagnostics in patient care.
The Indonesia Molecular Cytogenetics Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Genetika Science, PT Bio Farma, PT Prodia Widyahusada Tbk, PT Siloam International Hospitals Tbk, PT Kimia Farma Tbk, PT Medisafe Technologies, PT Bina Sehat Lestari, PT Sehat Selalu, PT Genomics Indonesia, PT Alat Kesehatan Indonesia, PT Citra Medika, PT Diagnos Laboratorium Utama, PT Genetika Medika, PT Bioteknologi Indonesia, PT Medika Citra, PT Kalbe Genomics, PT Nusantara Genomics, PT Indogen Laboratory, PT Daya Medika Pratama, PT PathGen Diagnostik Teknologi contribute to innovation, geographic expansion, and service delivery in this space.
The future of the molecular cytogenetics market in Indonesia appears promising, driven by technological advancements and increasing healthcare investments. As the government prioritizes healthcare infrastructure, the integration of telemedicine and remote diagnostics is expected to enhance access to genetic testing. Furthermore, collaborations with research institutions will foster innovation, leading to the development of cost-effective solutions that can address the high costs associated with molecular cytogenetic tests, ultimately improving patient care and outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Fluorescence In Situ Hybridization (FISH) Array Comparative Genomic Hybridization (aCGH) Next-Generation Sequencing (NGS)-based Cytogenetics Polymerase Chain Reaction (PCR)-based Cytogenetics Microarray Analysis Karyotyping Others |
| By End-User | Hospitals (Public & Private) Diagnostic Laboratories (Standalone & Networked) Research & Academic Institutions Pharmaceutical & Biotechnology Companies Others (Government Agencies, NGOs) |
| By Application | Cancer Diagnostics (Solid Tumors, Hematological Malignancies) Prenatal & Postnatal Testing Genetic Disorder Screening Infectious Disease Testing Personalized Medicine Others (Rare Disease Diagnosis, Pharmacogenomics) |
| By Distribution Channel | Direct Sales (Manufacturers to End-Users) Online Sales (e-Commerce Platforms) Distributors/Dealers Others (Tender-based Procurement) |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Papua Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing |
| By Technology | Traditional Cytogenetics (Conventional Karyotyping) Molecular Cytogenetics (FISH, aCGH, NGS) Advanced Genomic Technologies (Multi-omics, AI-driven Analysis) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 60 | Laboratory Managers, Geneticists |
| Healthcare Providers | 50 | Oncologists, Genetic Counselors |
| Research Institutions | 40 | Research Scientists, Academic Professors |
| Government Health Agencies | 40 | Policy Makers, Health Program Directors |
| Private Genetic Testing Companies | 40 | Business Development Managers, Product Managers |
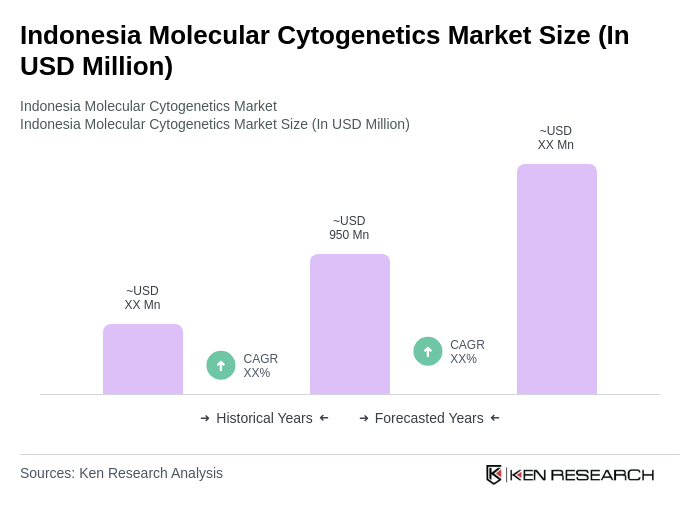
The Indonesia Molecular Cytogenetics Market is valued at approximately USD 950 million, reflecting significant growth driven by advancements in genetic testing technologies and increasing demand for personalized medicine.