Region:Asia
Author(s):Shubham
Product Code:KRAE0491
Pages:96
Published On:December 2025
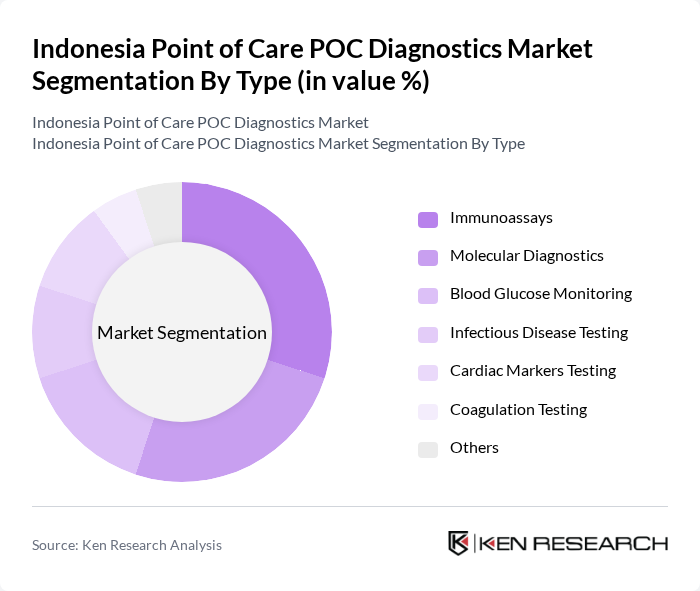
By Type:The market is segmented into various types of diagnostics, including Immunoassays, Molecular Diagnostics, Blood Glucose Monitoring, Infectious Disease Testing, Cardiac Markers Testing, Coagulation Testing, and Others. Among these, Immunoassays and Molecular Diagnostics are leading due to their rapid results and accuracy, which are crucial in emergency and chronic disease management.
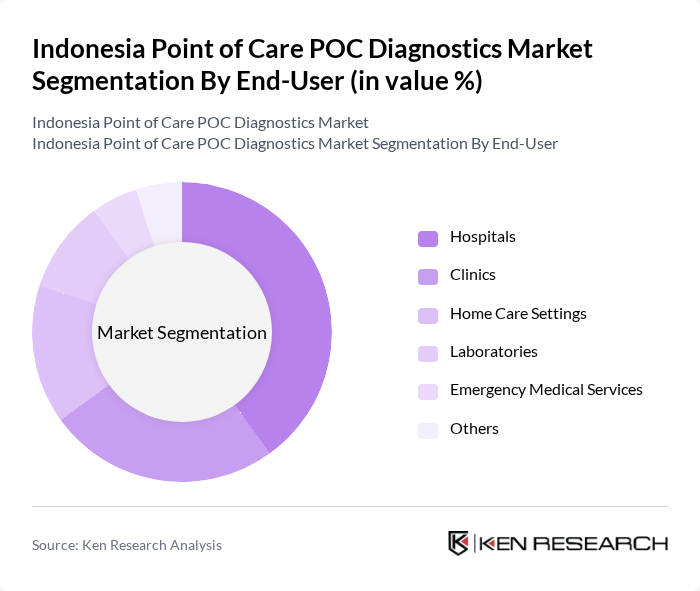
By End-User:The end-user segmentation includes Hospitals, Clinics, Home Care Settings, Laboratories, Emergency Medical Services, and Others. Hospitals are the dominant segment due to their need for immediate diagnostic results to facilitate timely treatment, especially in critical care settings.
The Indonesia Point of Care POC Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Cepheid, Bio-Rad Laboratories, Quidel Corporation, Becton, Dickinson and Company, Hologic, Inc., Danaher Corporation, Thermo Fisher Scientific, Abbott Point of Care, Ortho Clinical Diagnostics, GenMark Diagnostics, Mylab Discovery Solutions, and Medtronic contribute to innovation, geographic expansion, and service delivery in this space.
The future of the POC diagnostics market in Indonesia appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the shift towards home-based testing solutions will cater to the growing demand for convenience. Additionally, partnerships with local healthcare providers will facilitate the distribution of innovative POC devices, ensuring broader access to essential diagnostic services across the country.
| Segment | Sub-Segments |
|---|---|
| By Type | Immunoassays Molecular Diagnostics Blood Glucose Monitoring Infectious Disease Testing Cardiac Markers Testing Coagulation Testing Others |
| By End-User | Hospitals Clinics Home Care Settings Laboratories Emergency Medical Services Others |
| By Application | Disease Screening Monitoring Chronic Conditions Emergency Diagnostics Preventive Healthcare Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Technology | Lateral Flow Assays Microfluidics Nucleic Acid Amplification Biosensors Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Public-Private Partnerships Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 100 | Lab Managers, Medical Technologists |
| Private Clinics | 80 | General Practitioners, Clinic Owners |
| Public Health Facilities | 70 | Healthcare Administrators, Public Health Officials |
| Diagnostic Equipment Distributors | 60 | Sales Managers, Product Specialists |
| Patient Focus Groups | 50 | Patients, Caregivers |
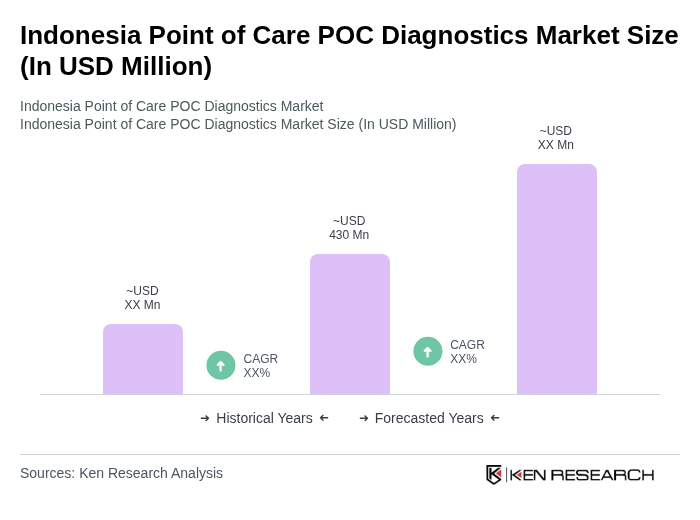
The Indonesia Point of Care (POC) Diagnostics Market is valued at approximately USD 430 million, reflecting significant growth driven by the increasing prevalence of infectious and chronic diseases, as well as technological innovations in diagnostics.