Region:Asia
Author(s):Rebecca
Product Code:KRAC2623
Pages:86
Published On:October 2025
By Type:The sterilization monitoring market is segmented into Chemical Indicators, Biological Indicators, Physical Indicators, Integrating Indicators, Monitoring Systems, Accessories, and Others. Chemical indicators are widely used for routine process checks, while biological indicators are preferred for critical process validation due to their ability to confirm the destruction of microbial spores. Physical indicators, such as temperature and pressure monitoring devices, are essential for continuous process control. Integrating indicators and advanced monitoring systems are gaining traction for their ability to provide comprehensive validation and traceability, especially in high-throughput hospital and laboratory environments. Accessories include supporting products such as indicator tapes and test packs, which complement the main monitoring solutions .
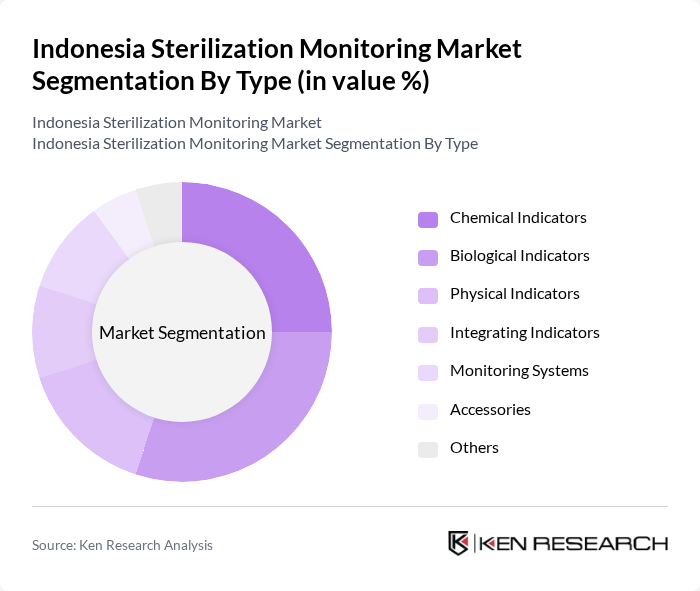
The Biological Indicators subsegment is currently dominating the market due to their critical role in validating sterilization processes. These indicators provide reliable evidence that sterilization has been achieved, which is essential for compliance with health regulations. The increasing focus on patient safety and infection control in healthcare facilities has led to a higher adoption rate of biological indicators, making them a preferred choice among healthcare providers. Rapid-readout biological indicators and automated loggers are further enhancing efficiency and traceability in sterilization validation .
By End-User:The market is segmented by end-users, including Hospitals, Clinics, Laboratories, Pharmaceutical Companies, Research Institutions, Food Processing Facilities, and Others. Hospitals require comprehensive sterilization monitoring due to the high volume of surgical procedures and stringent infection control standards. Clinics and laboratories rely on monitoring solutions for routine instrument reprocessing and sample integrity. Pharmaceutical companies and research institutions utilize advanced monitoring systems for regulatory compliance and product safety. Food processing facilities adopt sterilization monitoring to ensure product safety and meet food hygiene standards .
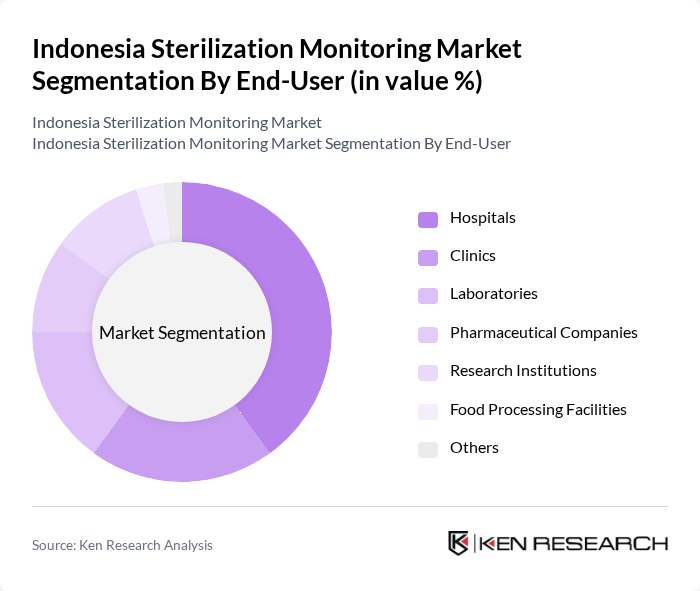
Hospitals are the leading end-user segment, accounting for a significant portion of the market. This dominance is attributed to the high volume of surgical procedures and the stringent sterilization requirements in hospital settings. The increasing number of hospitals and the growing emphasis on infection control measures further drive the demand for sterilization monitoring solutions in this sector. The expansion of ambulatory surgery centers and outpatient facilities is also contributing to the rising adoption of advanced monitoring systems .
The Indonesia Sterilization Monitoring Market is characterized by a dynamic mix of regional and international players. Leading participants such as 3M Company, Getinge AB, STERIS Corporation, Belimed AG, Cantel Medical Corporation, Ecolab Inc., Halyard Health, Inc., Advanced Sterilization Products (ASP, a Fortive company), Mesa Laboratories, Inc., Becton, Dickinson and Company (BD), Thermo Fisher Scientific Inc., Hu-Friedy Mfg. Co., LLC, Aesculap, Inc. (B. Braun Group), Cardinal Health, Inc., PT Enseval Medika Prima (Indonesia), PT Rajawali Nusindo (Indonesia), PT Kimia Farma Tbk (Indonesia) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian sterilization monitoring market appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on infection control. As hospitals and clinics increasingly adopt automated and IoT-enabled sterilization solutions, the market is expected to evolve rapidly. Additionally, government initiatives aimed at enhancing healthcare quality will likely spur further advancements in sterilization technologies, ensuring compliance with international standards and improving patient safety across the nation.
| Segment | Sub-Segments |
|---|---|
| By Type | Chemical Indicators Biological Indicators Physical Indicators Integrating Indicators Monitoring Systems Accessories Others |
| By End-User | Hospitals Clinics Laboratories Pharmaceutical Companies Research Institutions Food Processing Facilities Others |
| By Application | Surgical Procedures Laboratory Testing Pharmaceutical Manufacturing Medical Device Sterilization Food Safety Monitoring Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Outlets Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Papua Others |
| By Price Range | Low Medium High |
| By Technology | Manual Monitoring Automated Monitoring Digital Monitoring Rapid Readout Systems Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Sterilization Units | 100 | Sterilization Managers, Infection Control Officers |
| Private Clinics and Healthcare Facilities | 70 | Clinic Owners, Medical Directors |
| Medical Device Manufacturers | 50 | Product Managers, Sales Executives |
| Regulatory Bodies and Health Authorities | 40 | Policy Makers, Health Inspectors |
| Research Institutions and Universities | 40 | Healthcare Researchers, Academic Professors |
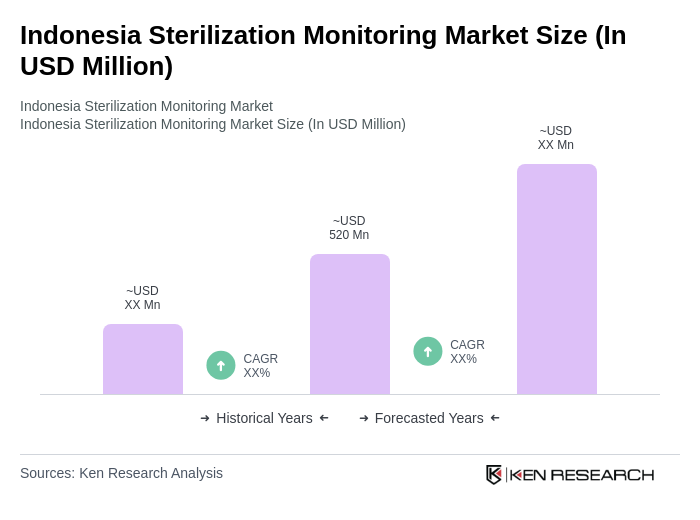
The Indonesia Sterilization Monitoring Market is valued at approximately USD 520 million, reflecting a significant growth driven by increasing demand for sterilization in healthcare facilities and advancements in monitoring technologies.