About the Report
Base Year 2024US Biological Indicators Market Overview
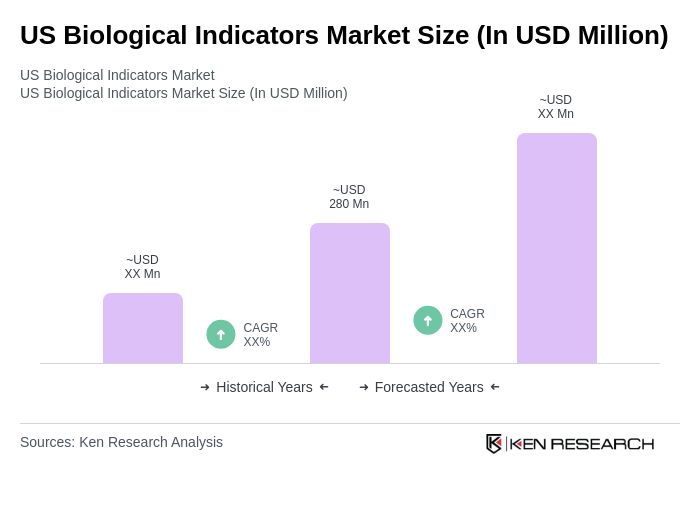
- The US Biological Indicators Market is valued at USD 280 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for sterilization processes in healthcare settings, coupled with the rising awareness of infection control measures. The market is also supported by advancements in technology and the growing number of surgical procedures performed annually.
- Key players in this market include major cities such as New York, Los Angeles, and Chicago, which dominate due to their extensive healthcare infrastructure and research institutions. These urban centers are hubs for medical innovation and have a high concentration of hospitals and laboratories, driving the demand for biological indicators.
- In 2023, the US government implemented the FDA's updated guidelines for sterilization processes, mandating the use of biological indicators in healthcare facilities. This regulation aims to enhance patient safety and ensure compliance with sterilization standards, thereby increasing the adoption of biological indicators across various healthcare settings.
US Biological Indicators Market Segmentation
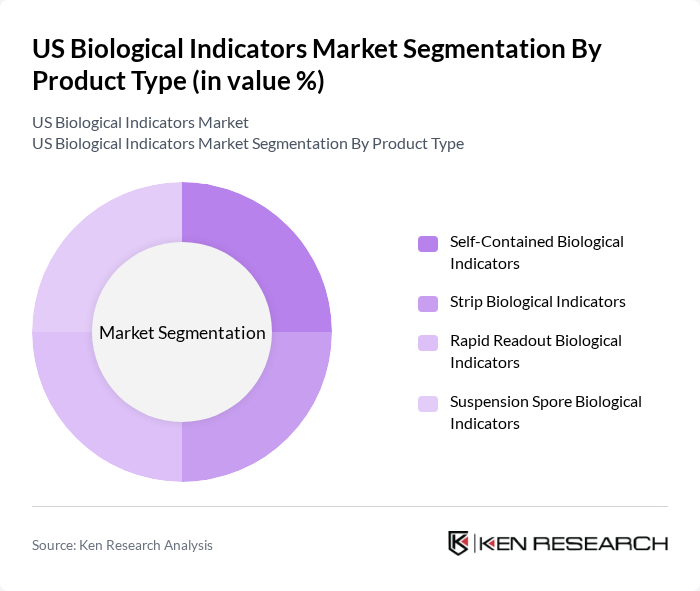
By Product Type:The product type segmentation includes Self-Contained Biological Indicators, Strip Biological Indicators, Rapid Readout Biological Indicators, and Suspension Spore Biological Indicators. Among these, Self-Contained Biological Indicators are leading the market due to their ease of use and reliability in providing immediate results. The demand for these indicators is driven by their application in various sterilization processes, particularly in hospitals and surgical centers.
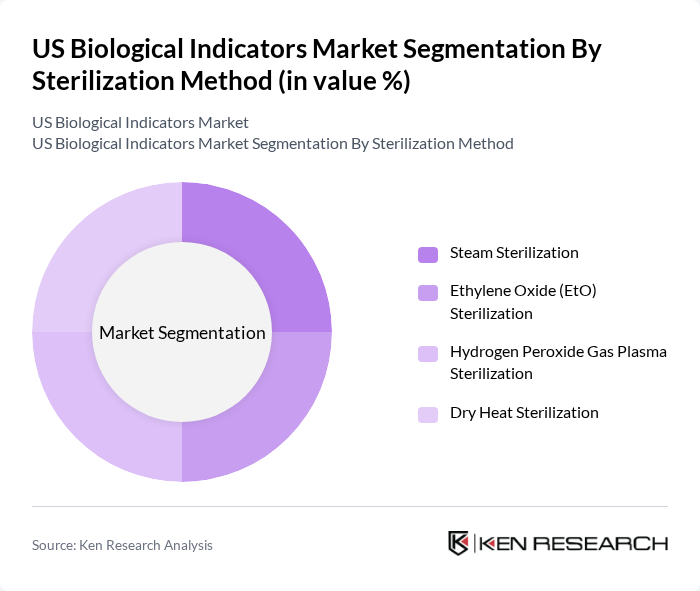
By Sterilization Method:The sterilization method segmentation includes Steam Sterilization, Ethylene Oxide (EtO) Sterilization, Hydrogen Peroxide Gas Plasma Sterilization, and Dry Heat Sterilization. Steam Sterilization is the dominant method due to its effectiveness and widespread use in healthcare facilities. The increasing focus on infection control and the need for efficient sterilization processes are driving the adoption of this method.
US Biological Indicators Market Competitive Landscape
The US Biological Indicators Market is characterized by a dynamic mix of regional and international players. Leading participants such as 3M Company, Getinge Group, STERIS plc, Mesa Labs, Inc., Cantel Medical Corp. (Sotera Health), Ecolab Inc., Advanced Sterilization Products (ASP), Becton, Dickinson and Company, Thermo Fisher Scientific Inc., Cardinal Health, Inc., VWR International, LLC, Terragene LLC, SSI Diagnostica A/S, Propper Manufacturing Company, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
US Biological Indicators Market Industry Analysis
Growth Drivers
- Increasing Demand for Sterilization in Healthcare Settings:The US healthcare sector is projected to reach $4.6 trillion in the future, driving the need for effective sterilization methods. With over 6,000 hospitals and numerous outpatient facilities, the demand for biological indicators is surging. The Centers for Disease Control and Prevention (CDC) reported that healthcare-associated infections affect 1 in 31 hospital patients, emphasizing the critical need for reliable sterilization processes to ensure patient safety and compliance with health standards.
- Rising Awareness of Infection Control:The global focus on infection control has intensified, particularly post-COVID-19. The US infection control market is expected to exceed $22 billion in the future, highlighting the increasing emphasis on safety protocols. Educational initiatives and training programs are being implemented across healthcare facilities, leading to a heightened understanding of the importance of biological indicators in monitoring sterilization efficacy, thus driving market growth.
- Technological Advancements in Biological Indicators:The US market is witnessing significant innovations, with investments in R&D projected to reach $210 billion in the future. Advanced biological indicators, such as rapid-readout systems, are becoming more prevalent, enhancing the efficiency of sterilization processes. These technologies not only improve accuracy but also reduce turnaround times, making them essential for modern healthcare facilities striving for operational excellence and patient safety.
Market Challenges
- High Costs Associated with Advanced Biological Indicators:The adoption of advanced biological indicators often comes with substantial costs, with some systems priced over $11,000. This financial barrier can deter smaller healthcare facilities from investing in these technologies, limiting their ability to ensure effective sterilization. As a result, the market faces challenges in achieving widespread adoption, particularly in budget-constrained environments where cost-effectiveness is paramount.
- Limited Awareness Among Small Healthcare Facilities:Approximately 32% of small healthcare facilities lack adequate knowledge about the benefits of biological indicators. This gap in awareness can lead to suboptimal sterilization practices, increasing the risk of healthcare-associated infections. Educational outreach and training programs are essential to bridge this knowledge gap, but the slow uptake of information poses a significant challenge to market growth in this segment.
US Biological Indicators Market Future Outlook
The US biological indicators market is poised for transformative growth, driven by technological advancements and an increasing focus on infection control. As healthcare facilities adopt automated sterilization processes, the demand for rapid biological indicators will rise. Additionally, the integration of digital technologies for monitoring sterilization efficacy will enhance operational efficiency. The emphasis on sustainability will also shape product development, leading to eco-friendly biological indicators that meet regulatory standards while addressing environmental concerns.
Market Opportunities
- Expansion in Emerging Healthcare Markets:The US healthcare market is expanding into underserved regions, with investments projected to reach $55 billion in the future. This growth presents opportunities for biological indicator manufacturers to penetrate new markets, providing essential sterilization solutions to facilities that previously lacked access to advanced technologies.
- Development of Eco-Friendly Biological Indicators:With increasing regulatory pressure for sustainable practices, the demand for eco-friendly biological indicators is on the rise. Companies that innovate in this area can capture a growing segment of environmentally conscious healthcare providers, potentially increasing market share and enhancing brand reputation in a competitive landscape.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Self-Contained Biological Indicators Strip Biological Indicators Rapid Readout Biological Indicators Suspension Spore Biological Indicators |
| By Sterilization Method | Steam Sterilization Ethylene Oxide (EtO) Sterilization Hydrogen Peroxide Gas Plasma Sterilization Dry Heat Sterilization |
| By End-User | Hospitals and Surgical Centers Pharmaceutical Manufacturing Medical Device Manufacturing Clinical Laboratories Research and Academic Institutions |
| By Distribution Channel | Direct Sales to Healthcare Facilities Medical Device Distributors Online and E-Commerce Platforms Hospital Group Purchasing Organizations (GPOs) |
| By US Region | Northeast Midwest South West |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, Environmental Protection Agency)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Biotechnology and Pharmaceutical Companies
Industry Associations (e.g., Association for the Advancement of Medical Instrumentation)
Financial Institutions
Players Mentioned in the Report:
3M Company
Getinge Group
STERIS plc
Mesa Labs, Inc.
Cantel Medical Corp. (Sotera Health)
Ecolab Inc.
Advanced Sterilization Products (ASP)
Becton, Dickinson and Company
Thermo Fisher Scientific Inc.
Cardinal Health, Inc.
VWR International, LLC
Terragene LLC
SSI Diagnostica A/S
Propper Manufacturing Company, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Biological Indicators Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Biological Indicators Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Biological Indicators Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for sterilization in healthcare settings
3.1.2 Rising awareness of infection control
3.1.3 Technological advancements in biological indicators
3.1.4 Stringent regulatory requirements for sterilization processes
3.2 Market Challenges
3.2.1 High costs associated with advanced biological indicators
3.2.2 Limited awareness among small healthcare facilities
3.2.3 Competition from alternative sterilization methods
3.2.4 Regulatory compliance complexities
3.3 Market Opportunities
3.3.1 Expansion in emerging healthcare markets
3.3.2 Development of eco-friendly biological indicators
3.3.3 Increasing investments in healthcare infrastructure
3.3.4 Collaborations with research institutions
3.4 Market Trends
3.4.1 Shift towards automated sterilization processes
3.4.2 Growing preference for rapid biological indicators
3.4.3 Integration of digital technologies in monitoring
3.4.4 Focus on sustainability and eco-friendly products
3.5 Government Regulation
3.5.1 FDA regulations on biological indicators
3.5.2 Compliance with ISO standards for sterilization
3.5.3 State-specific regulations on healthcare practices
3.5.4 Guidelines from CDC on infection control
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Biological Indicators Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Biological Indicators Market Segmentation
8.1 By Product Type
8.1.1 Self-Contained Biological Indicators
8.1.2 Strip Biological Indicators
8.1.3 Rapid Readout Biological Indicators
8.1.4 Suspension Spore Biological Indicators
8.2 By Sterilization Method
8.2.1 Steam Sterilization
8.2.2 Ethylene Oxide (EtO) Sterilization
8.2.3 Hydrogen Peroxide Gas Plasma Sterilization
8.2.4 Dry Heat Sterilization
8.3 By End-User
8.3.1 Hospitals and Surgical Centers
8.3.2 Pharmaceutical Manufacturing
8.3.3 Medical Device Manufacturing
8.3.4 Clinical Laboratories
8.3.5 Research and Academic Institutions
8.4 By Distribution Channel
8.4.1 Direct Sales to Healthcare Facilities
8.4.2 Medical Device Distributors
8.4.3 Online and E-Commerce Platforms
8.4.4 Hospital Group Purchasing Organizations (GPOs)
8.5 By US Region
8.5.1 Northeast
8.5.2 Midwest
8.5.3 South
8.5.4 West
9. US Biological Indicators Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Key Performance Indicators (KPIs) for Cross Comparison of Key Players
9.2.1 Company Revenue and Market Share Percentage
9.2.2 Company Classification (Large-Cap, Mid-Cap, or Specialized)
9.2.3 Year-over-Year Revenue Growth Rate (%)
9.2.4 US Market Penetration Rate by End-User Segment (%)
9.2.5 Customer Retention and Repeat Order Rate (%)
9.2.6 New Product Launch Frequency and Adoption Rate
9.2.7 Distribution Network Coverage (Direct + Indirect Channels)
9.2.8 Pricing Strategy (Premium, Mid-Range, Value-Based)
9.2.9 Brand Recognition and Market Visibility Score
9.2.10 Regulatory Approvals and Certifications (FDA, ISO 11138)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 3M Company
9.5.2 Getinge Group
9.5.3 STERIS plc
9.5.4 Mesa Labs, Inc.
9.5.5 Cantel Medical Corp. (Sotera Health)
9.5.6 Ecolab Inc.
9.5.7 Advanced Sterilization Products (ASP)
9.5.8 Becton, Dickinson and Company
9.5.9 Thermo Fisher Scientific Inc.
9.5.10 Cardinal Health, Inc.
9.5.11 VWR International, LLC
9.5.12 Terragene LLC
9.5.13 SSI Diagnostica A/S
9.5.14 Propper Manufacturing Company, Inc.
10. US Biological Indicators Market End-User Analysis
10.1 Procurement Behavior of Key End-Users
10.1.1 Healthcare facility procurement policies
10.1.2 Group Purchasing Organization (GPO) influence
10.1.3 Evaluation criteria for supplier selection
10.2 Corporate Spend on Infrastructure & Sterilization Technology
10.2.1 Investment in sterilization validation technologies
10.2.2 Budget allocation trends in hospitals and clinics
10.2.3 Spending on compliance and quality assurance
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in sterilization validation and monitoring
10.3.2 Issues with product reliability and turnaround time
10.3.3 Cost constraints and budget limitations
10.4 User Readiness for Adoption
10.4.1 Training and education needs for staff
10.4.2 Technology adoption barriers and resistance
10.4.3 Awareness of rapid-readout and digital monitoring benefits
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of sterilization effectiveness
10.5.2 Expansion into new sterilization methods and applications
10.5.3 Long-term cost savings and operational efficiency gains
11. US Biological Indicators Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategies
2.5 Digital marketing tactics
2.6 Trade show participation
2.7 Customer engagement initiatives
3. Distribution Plan
3.1 Healthcare facility direct sales strategies
3.2 Medical device distributor partnerships
3.3 Online and e-commerce channels
3.4 Group Purchasing Organization (GPO) relationships
3.5 Direct sales force deployment
3.6 Logistics and supply chain management
4. Channel & Pricing Gaps
4.1 Underserved market segments
4.2 Pricing bands analysis by product type
4.3 Competitor pricing comparison
4.4 Value-based pricing strategies
4.5 Volume discounts and promotional strategies
4.6 Customer feedback on pricing
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 End-user segments analysis
5.3 Emerging trends exploration
5.4 Product development opportunities
5.5 Market entry barriers
5.6 Customer pain points
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback mechanisms
6.4 Relationship management strategies
6.5 Community engagement
6.6 Customer education initiatives
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-effectiveness
7.4 Quality assurance
7.5 Innovation in product offerings
7.6 Customer-centric approach
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Market research
8.5 Training and development
8.6 Performance monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging considerations
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from industry associations such as the American Society for Microbiology
- Review of scientific publications and journals focusing on biological indicators and their applications
- Examination of regulatory frameworks and guidelines from the Environmental Protection Agency (EPA) and Food and Drug Administration (FDA)
Primary Research
- Interviews with product managers at leading biological indicator manufacturers
- Surveys with laboratory directors and quality assurance professionals in healthcare settings
- Field interviews with environmental scientists and microbiologists involved in indicator development
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of data from regulatory bodies, industry reports, and primary interviews
- Sanity checks through expert panel discussions to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and laboratory spending
- Segmentation by application areas such as healthcare, pharmaceuticals, and environmental monitoring
- Incorporation of trends in sterilization practices and infection control measures
Bottom-up Modeling
- Volume estimates derived from sales data of biological indicators across various sectors
- Cost analysis based on pricing models of leading manufacturers and distributors
- Calculation of market size using unit sales multiplied by average selling prices
Forecasting & Scenario Analysis
- Multi-variable forecasting using growth rates in healthcare and environmental sectors
- Scenario analysis based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Sector Biological Indicators | 120 | Laboratory Managers, Infection Control Specialists |
| Pharmaceutical Manufacturing Applications | 90 | Quality Assurance Managers, Production Supervisors |
| Environmental Monitoring Practices | 80 | Environmental Scientists, Compliance Officers |
| Research and Development in Microbiology | 70 | Microbiologists, R&D Directors |
| Regulatory Compliance and Standards | 60 | Regulatory Affairs Specialists, Policy Makers |
Frequently Asked Questions
What is the current value of the US Biological Indicators Market?
The US Biological Indicators Market is valued at approximately USD 280 million, reflecting a significant growth driven by the increasing demand for sterilization processes in healthcare settings and advancements in technology.