Region:Middle East
Author(s):Dev
Product Code:KRAC1595
Pages:88
Published On:December 2025
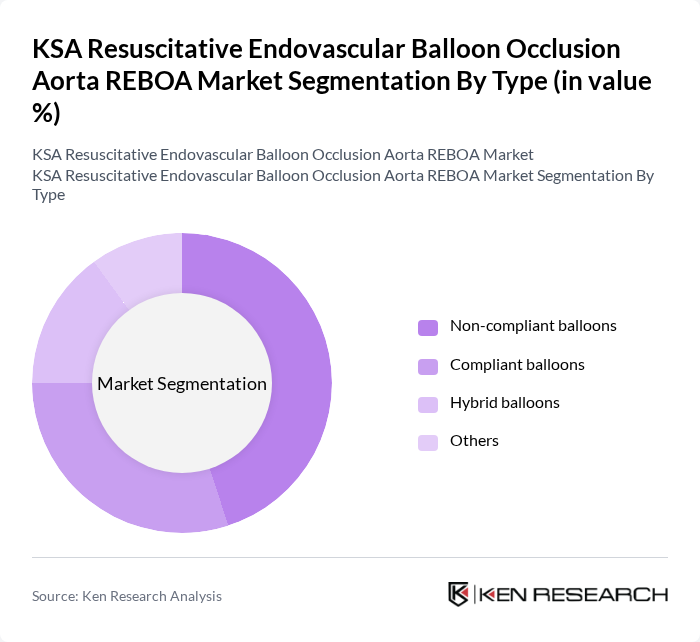
By Type:The market is segmented into various types of balloons used in REBOA procedures. Non-compliant balloons are designed to maintain a fixed volume and pressure, making them suitable for specific applications. Compliant balloons, on the other hand, can expand and contract, providing flexibility during procedures. Hybrid balloons combine features of both types, offering versatility. Other types may include specialized balloons for unique clinical scenarios. Non-compliant balloons currently dominate the market due to their reliability and effectiveness in critical situations.
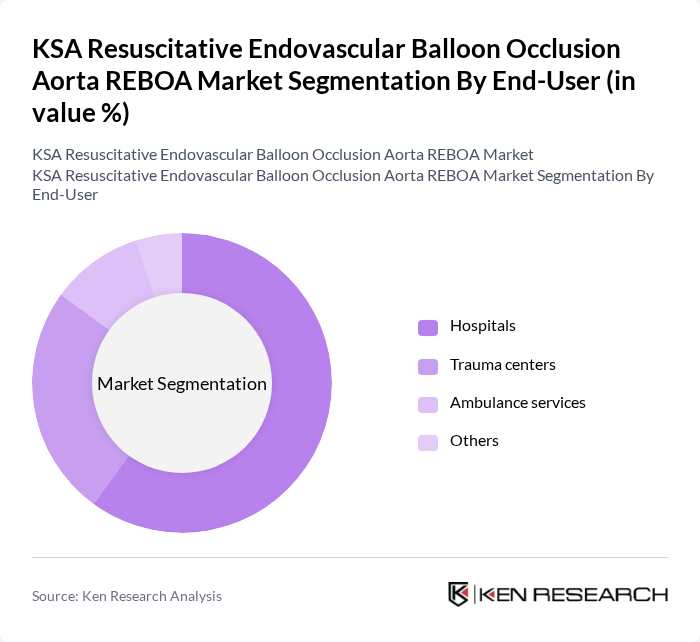
By End-User:The end-user segmentation includes hospitals, trauma centers, ambulance services, and other healthcare facilities. Hospitals are the primary users of REBOA technology, as they have the necessary infrastructure and trained personnel to perform these advanced procedures. Trauma centers also play a significant role, given their focus on emergency care. Ambulance services are increasingly integrating REBOA devices for pre-hospital care, enhancing patient outcomes during transport. Hospitals lead the market due to their comprehensive trauma care capabilities and resources.
The KSA Resuscitative Endovascular Balloon Occlusion Aorta REBOA Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corp, Medtronic PLC, Terumo Corp, Abbott Laboratories, and Cardinal Health Inc contribute to innovation, geographic expansion, and service delivery in this space.
The future of the KSA REBOA market appears promising, driven by emerging technologies and increased government funding for healthcare innovation. With over USD 1.5 billion allocated to health technology under Vision 2030, the integration of digital health solutions will enhance procedural planning and training for REBOA. Additionally, public-private partnerships are expected to foster localized development of advanced medical devices, creating a conducive environment for the adoption of REBOA systems in trauma care.
| Segment | Sub-Segments |
|---|---|
| By Type | Non-compliant balloons Compliant balloons Hybrid balloons Others |
| By End-User | Hospitals Trauma centers Ambulance services Others |
| By Application | Trauma management Surgical procedures Research and development Others |
| By Distribution Channel | Direct sales Distributors Online platforms Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Patient Demographics | Adults Pediatric Geriatric Others |
| By Technology Integration | Standalone devices Integrated systems Telemedicine-enabled devices Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Trauma Surgery Departments | 100 | Trauma Surgeons, Emergency Medicine Physicians |
| Hospital Procurement Teams | 80 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Distributors | 60 | Sales Representatives, Regional Managers |
| Healthcare Policy Makers | 50 | Health Administrators, Policy Analysts |
| Clinical Researchers in Trauma Care | 70 | Clinical Researchers, Academic Physicians |
The KSA REBOA Market is valued at approximately USD 78 million, driven by the increasing incidence of trauma cases and the adoption of minimally invasive hemorrhage control devices, alongside advancements in endovascular technologies and healthcare infrastructure.