Region:Asia
Author(s):Rebecca
Product Code:KRAE4301
Pages:80
Published On:March 2026
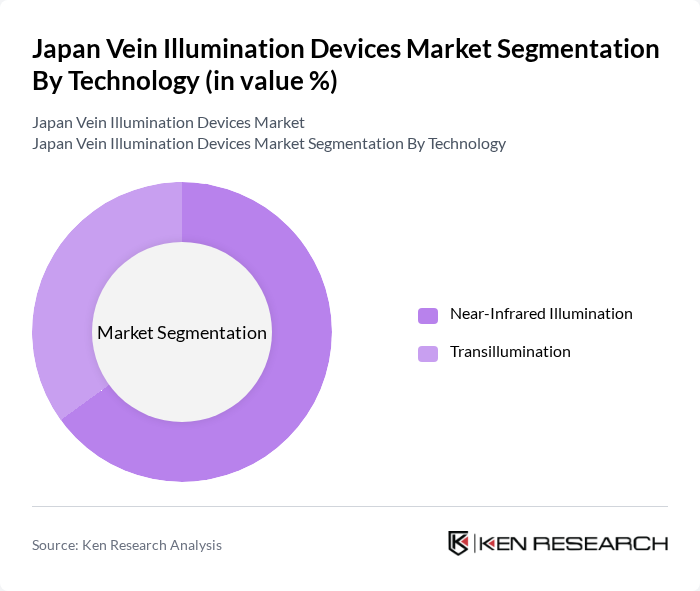
By Technology:The technology segment includes various methods used in vein illumination devices, primarily focusing on Near-Infrared Illumination and Transillumination. Near-Infrared Illumination is widely adopted due to its effectiveness in visualizing veins beneath the skin, enabling real-time, non-contact mapping of superficial veins across a wide range of skin tones and body habitus.Transillumination, which uses high-intensity visible or LED light, offers alternative methods for vein detection, especially in neonates and infants where backlighting the limb is advantageous.The market is witnessing a growing preference for Near-Infrared Illumination technology, attributed to its higher first‑attempt success rates, reduced procedure time, and better performance in difficult venous access cases in emergency, oncology, and dialysis settings.
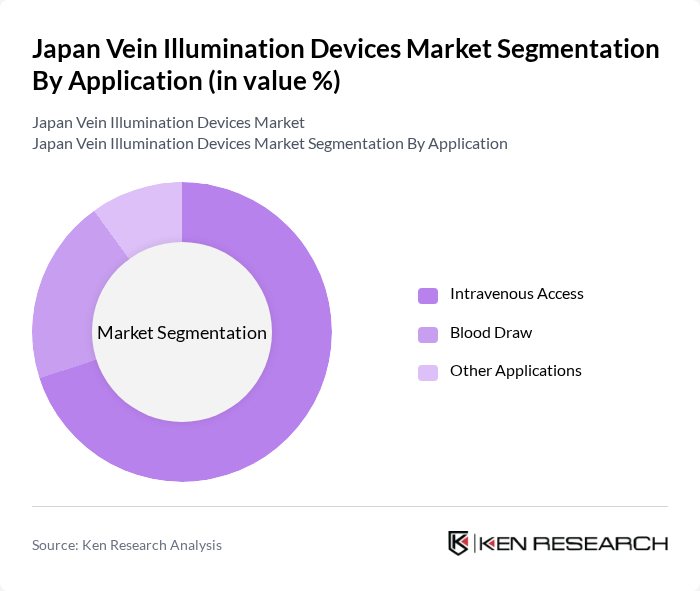
By Application:The application segment encompasses various uses of vein illumination devices, including Intravenous Access, Blood Draw, and Other Applications. Intravenous Access is the leading application, driven by the increasing number of patients requiring intravenous therapy for oncology, cardiovascular, renal, and infectious diseases, as well as the growing use of peripheral IV, central venous, and peripherally inserted central catheter (PICC) lines.Accurate vein visualization reduces failed cannulation attempts, helps avoid complications such as hematoma or arterial puncture, and improves clinical workflow efficiency in operating rooms, emergency departments, and intensive care units.Blood Draw is also significant, as healthcare providers aim to minimize patient discomfort and improve the efficiency of routine and high‑volume phlebotomy procedures, including in outpatient laboratories, dialysis units, and home-care settings, where portable vein illuminators support safer, more consistent venipuncture.
The Japan Vein Illumination Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as AccuVein Inc, Veinlite LLC, Christie Medical Holdings Inc, Near Infrared Imaging Inc, Venoscope LLC, TransLite LLC, ZD Medical Inc, AimVein Ltd, BLZ Medical SAS, Shenzhen Bestman Instrument Co Ltd, Vuetek Scientific LLC, Infrared Imaging Systems Inc, Vivolight LLC, Optics Medical LLC, Luminous Medical Inc contribute to innovation, geographic expansion, and service delivery in this space.
The future of the vein illumination devices market in Japan appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in device functionality is expected to enhance diagnostic accuracy and procedural efficiency. Additionally, the shift towards home healthcare solutions is likely to create new avenues for device utilization, as patients seek convenient and effective treatment options. These trends indicate a dynamic market landscape poised for growth and innovation in the future.
| Segment | Sub-Segments |
|---|---|
| By Technology | Near-Infrared Illumination Transillumination |
| By Application | Intravenous Access Blood Draw Other Applications |
| By End-User | Hospitals Blood Donation Camps |
| By Device Type | Handheld Vein Illuminators Fixed Vein Illumination Systems |
| By Transillumination Type | Fiber-Optic Transillumination Devices LED-Based Transillumination Devices |
| By Region | Kanto Kansai Chubu Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 120 | Procurement Managers, Medical Equipment Buyers |
| Private Clinics and Dermatology Centers | 90 | Clinic Owners, Dermatologists |
| Medical Device Distributors | 70 | Sales Managers, Distribution Executives |
| Healthcare Technology Experts | 50 | Medical Device Engineers, R&D Managers |
| Regulatory Bodies and Health Authorities | 40 | Regulatory Affairs Specialists, Policy Makers |
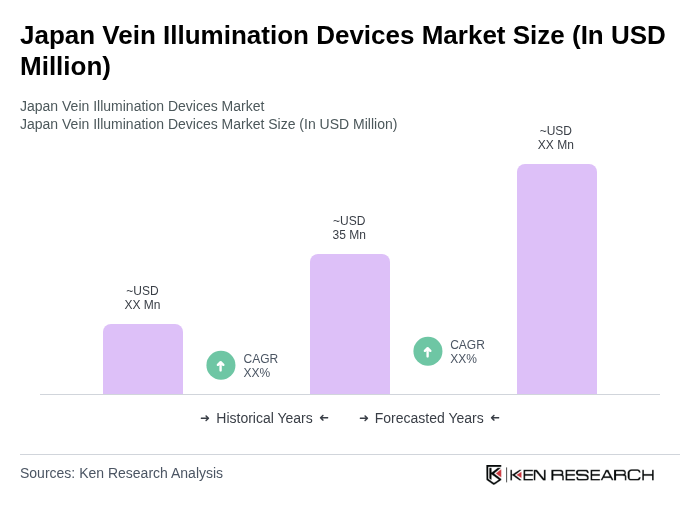
The Japan Vein Illumination Devices Market is valued at approximately USD 35 million, driven by the increasing prevalence of chronic diseases, an aging population, and advancements in medical technology that enhance intravenous procedures.