Region:Asia
Author(s):Rebecca
Product Code:KRAE4300
Pages:88
Published On:March 2026
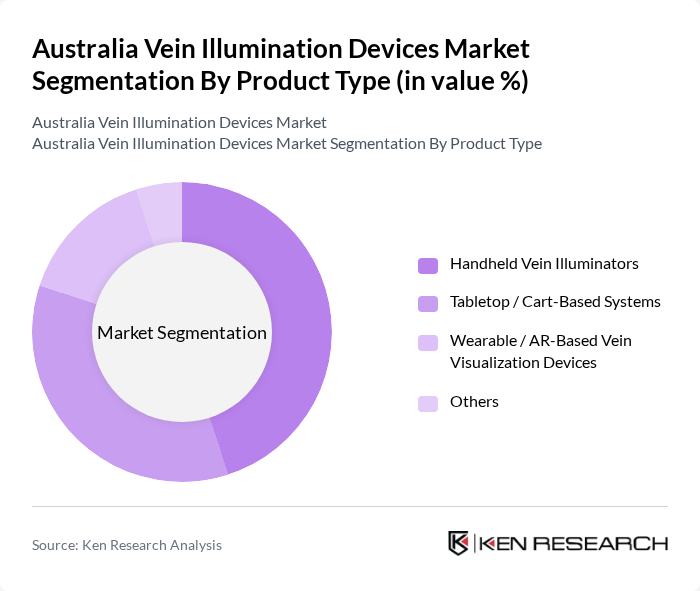
By Product Type:The product type segmentation includes handheld vein illuminators, tabletop/cart-based systems, wearable/AR-based vein visualization devices, and others. Handheld vein illuminators are widely adopted globally due to their portability, ease of use at the bedside, and suitability for emergency departments, oncology units, and outpatient phlebotomy, and this pattern is reflected in Australia as clinicians look for compact, battery‑operated solutions that improve first‑stick success.The demand for tabletop or cart-based systems is also significant, particularly in larger hospitals and day-surgery centers where higher‑end imaging and stable positioning can provide enhanced visualization for complex or high‑throughput procedures, while emerging wearable and AR-based devices are gradually entering advanced facilities that prioritize hands‑free workflow and real‑time visualization.
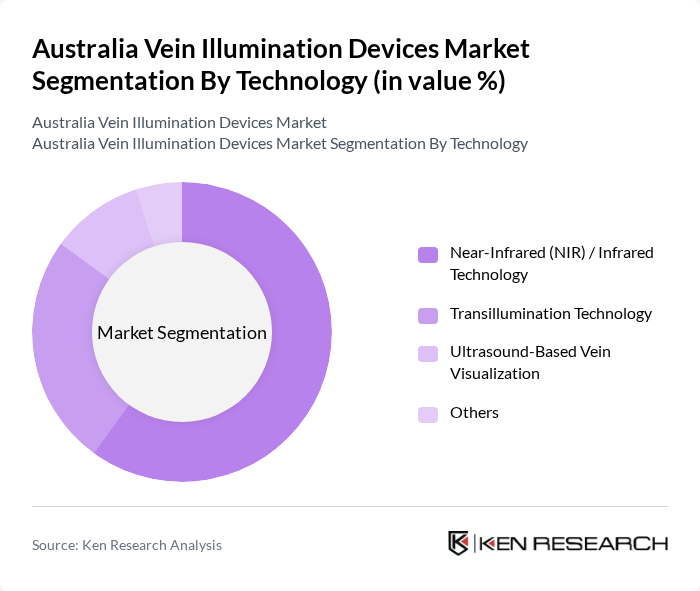
By Technology:The technology segmentation encompasses near-infrared (NIR) / infrared technology, transillumination technology, ultrasound-based vein visualization, and others. Near-infrared technology is the leading segment worldwide because hemoglobin strongly absorbs NIR light, enabling clear mapping of superficial veins and improving cannulation accuracy across adult, pediatric, and obese patient groups, which similarly underpins its dominance in Australian facilities adopting modern vein finders.Transillumination technology is also popular, particularly in neonatal and pediatric care, where compact, light‑based devices help visualize very small or fragile veins, while ultrasound-based vein visualization is increasingly used for deeper or more complex vascular access in critical care and anesthesia settings.
The Australia Vein Illumination Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as AccuVein, Inc., Christie Medical Holdings, Inc. (VeinViewer), TransLite, LLC (Veinlite), B. Braun Melsungen AG, Koninklijke Philips N.V., GE Healthcare Technologies Inc., Nipro Corporation, Vivolight Medical Device & Technology Co., Ltd., Venoscope, LLC, VINO Optics, Inc., Hellovein, Medline Industries, LP, Siemens Healthineers AG, Shenzhen ZD Medical Co., Ltd., Mindray Medical International Limited contribute to innovation, geographic expansion, and service delivery in this space.
The future of the vein illumination devices market in Australia appears promising, driven by ongoing technological advancements and a growing emphasis on patient-centric care. As healthcare providers increasingly adopt innovative solutions, the integration of artificial intelligence and machine learning into these devices is expected to enhance their functionality. Additionally, the rise of telemedicine and remote monitoring will likely create new avenues for the application of vein illumination technologies, further expanding their reach and impact in the healthcare sector.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Handheld Vein Illuminators Tabletop / Cart-Based Systems Wearable / AR-Based Vein Visualization Devices Others |
| By Technology | Near-Infrared (NIR) / Infrared Technology Transillumination Technology Ultrasound-Based Vein Visualization Others |
| By Application | IV Access & Venipuncture Blood Collection & Donation Sclerotherapy & Vein Treatment Surgical, Emergency & Critical Care Pediatric, Geriatric & Difficult Venous Access Cases Others |
| By End-User | Hospitals Specialty & Outpatient Clinics Ambulatory Surgical & Infusion Centers Home Care & Long-Term Care Settings Others |
| By Distribution Channel | Direct Sales to Healthcare Providers Medical Device Distributors Online / E-commerce Channels Group Purchasing Organizations (GPOs) & Tender-Based Procurement Others |
| By Region | New South Wales Victoria Queensland Western Australia South Australia Others (Tasmania, Australian Capital Territory, Northern Territory) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Usage of Vein Illumination Devices | 120 | Nurses, Phlebotomists, Medical Directors |
| Clinical Adoption in Outpatient Settings | 90 | Clinic Managers, Healthcare Practitioners |
| Home Healthcare Applications | 70 | Home Care Nurses, Patient Care Coordinators |
| Market Insights from Distributors | 55 | Sales Representatives, Product Managers |
| Regulatory Perspectives on Device Usage | 45 | Regulatory Affairs Specialists, Compliance Officers |
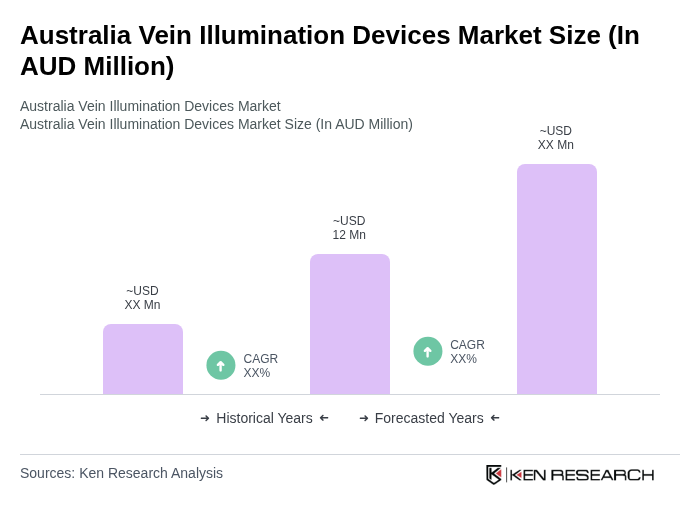
The Australia Vein Illumination Devices Market is valued at approximately AUD 12 million, reflecting a growing demand driven by the increasing prevalence of chronic diseases and advancements in medical technology.