Region:Asia
Author(s):Rebecca
Product Code:KRAE4292
Pages:95
Published On:March 2026
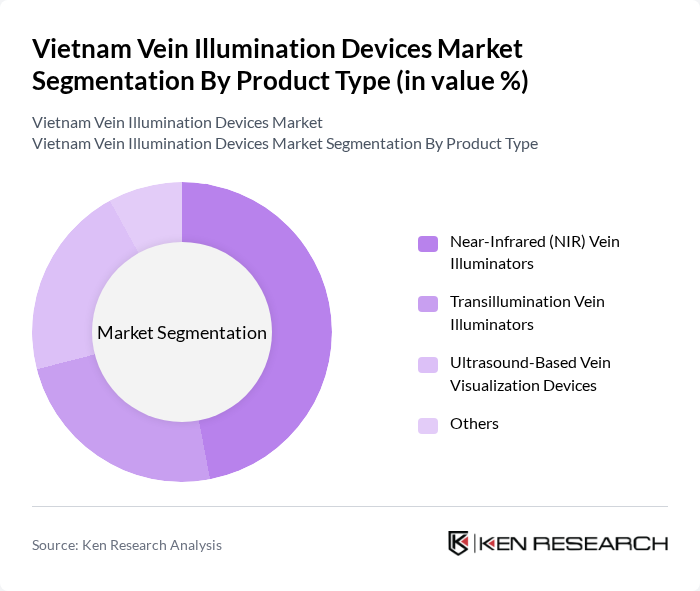
By Product Type:The market is segmented into various product types, including Near-Infrared (NIR) Vein Illuminators, Transillumination Vein Illuminators, Ultrasound-Based Vein Visualization Devices, and Others. Among these, Near-Infrared (NIR) Vein Illuminators are leading the market due to their effectiveness in visualizing veins, which significantly enhances the success rate of venipuncture procedures. The growing preference for non-invasive and user-friendly devices has also contributed to the increasing adoption of NIR technology in hospitals and clinics.
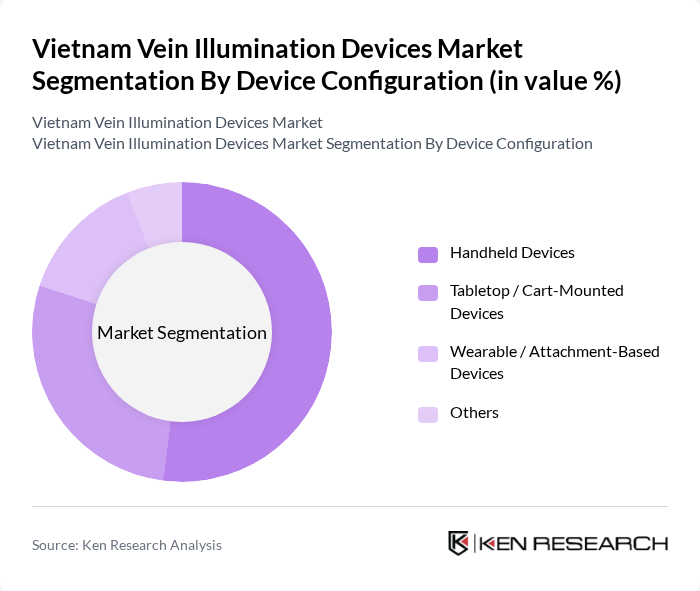
By Device Configuration:The segmentation by device configuration includes Handheld Devices, Tabletop / Cart-Mounted Devices, Wearable / Attachment-Based Devices, and Others. Handheld Devices are currently the most popular configuration due to their portability and ease of use, making them ideal for both hospital and home healthcare settings. The demand for these devices is driven by the need for quick and efficient venous access, particularly in emergency situations.
The Vietnam Vein Illumination Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as AccuVein Inc., Christie Medical Holdings, Inc. (VeinViewer), Medline Industries, LP, TransLite, LLC, Shandong Double Medical Technology, Shenzhen Bestman Instrument Co., Ltd., Shenzhen Vivolight Medical Device & Technology Co., Ltd., SIFSOF LLC, ZD Medical Inc., Rencongzhong (Beijing) Technology Co., Ltd., Near Infrared Imaging, Inc., BLZ Technology (and other local Vietnamese distributors), Philips Healthcare, GE HealthCare Technologies Inc., Siemens Healthineers AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam vein illumination devices market appears promising, driven by ongoing advancements in healthcare technology and increasing healthcare investments. As the government aims to enhance healthcare infrastructure, the integration of innovative medical devices is expected to rise. Additionally, the growing emphasis on patient-centered care will likely lead to increased demand for devices that improve patient experiences during procedures, fostering a more favorable environment for market expansion in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Near-Infrared (NIR) Vein Illuminators Transillumination Vein Illuminators Ultrasound-Based Vein Visualization Devices Others |
| By Device Configuration | Handheld Devices Tabletop / Cart-Mounted Devices Wearable / Attachment-Based Devices Others |
| By Application | Intravenous (IV) Access & Therapy Blood Draw & Phlebotomy Pediatric & Neonatal Care Emergency & Critical Care Cosmetic & Aesthetic Procedures Others |
| By End-User | Hospitals Ambulatory Surgical Centers Clinics & Physician Offices Blood Donation Centers Home Healthcare & Long-Term Care Facilities Others |
| By Distribution Channel | Direct Tender / Institutional Sales Medical Device Distributors Online / E-commerce Channels Others |
| By Region | Northern Vietnam Southern Vietnam Central Vietnam Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 120 | Procurement Managers, Medical Equipment Buyers |
| Outpatient Clinics | 90 | Clinic Owners, Healthcare Administrators |
| Medical Device Distributors | 70 | Sales Managers, Distribution Coordinators |
| Healthcare Professionals | 110 | Vascular Surgeons, Dermatologists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
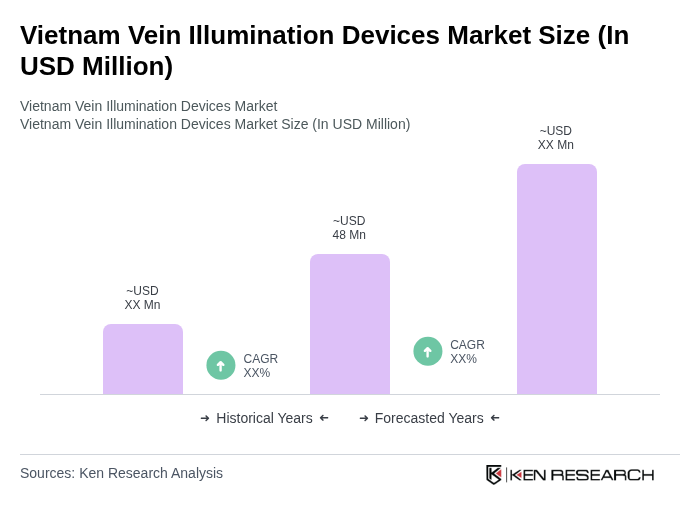
The Vietnam Vein Illumination Devices Market is valued at approximately USD 48 million, driven by the increasing prevalence of chronic diseases, an aging population, and advancements in medical technology that enhance patient care and venipuncture procedures.