Region:Middle East
Author(s):Geetanshi
Product Code:KRAC8352
Pages:83
Published On:November 2025
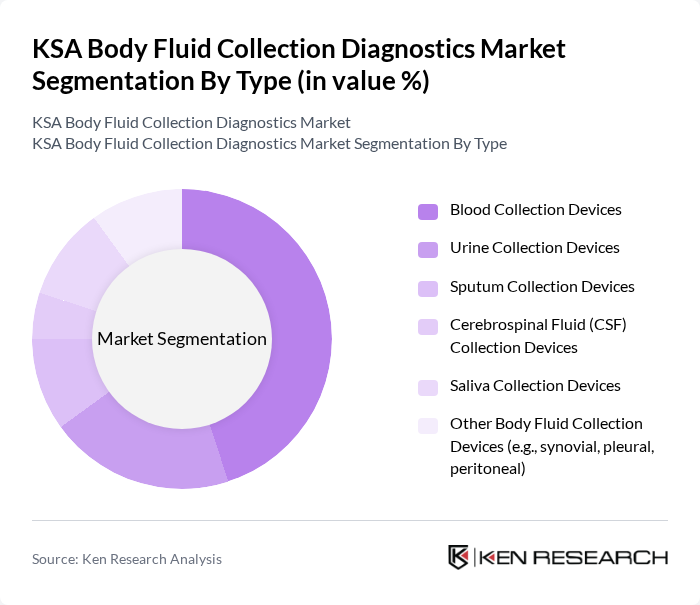
By Type:The market is segmented into various types of body fluid collection devices, which include blood collection devices, urine collection devices, sputum collection devices, cerebrospinal fluid (CSF) collection devices, saliva collection devices, and other body fluid collection devices such as synovial, pleural, and peritoneal collection devices. Among these,blood collection devicesdominate the market due to their widespread use in clinical settings for a broad range of diagnostic tests. The increasing number of blood tests performed annually, driven by the rise in chronic diseases, routine health check-ups, and national screening initiatives, significantly contributes to the growth of this segment .
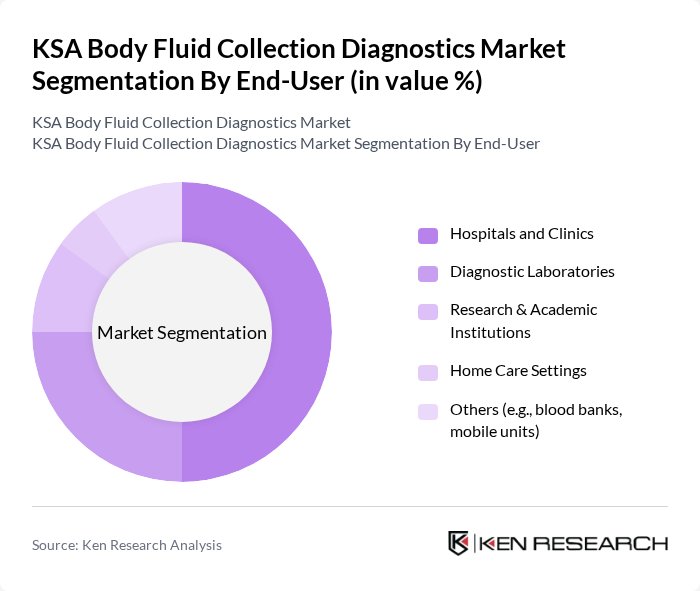
By End-User:The end-user segmentation includes hospitals and clinics, diagnostic laboratories, research and academic institutions, home care settings, and others such as blood banks and mobile units.Hospitals and clinicsare the leading end-users, accounting for a significant share of the market. This dominance is attributed to the high volume of diagnostic tests conducted in these facilities, the increasing patient population seeking medical care, and the expansion of point-of-care testing capabilities. The adoption of digital health solutions and government investment in healthcare infrastructure further enhance the demand for body fluid collection diagnostics in these settings .
The KSA Body Fluid Collection Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Becton, Dickinson and Company (BD), Thermo Fisher Scientific, Bio-Rad Laboratories, Hologic, Inc., Ortho Clinical Diagnostics (now part of QuidelOrtho Corporation), Sysmex Corporation, Mindray Medical International Limited, Grifols S.A., PerkinElmer, Inc. (now Revvity, Inc.), Agilent Technologies, QIAGEN N.V., DiaSorin S.p.A., Illumina, Inc., Guardant Health, Inc., Biocept, Inc., Johnson & Johnson (Janssen Diagnostics), Laboratory Corporation of America Holdings (LabCorp) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the KSA body fluid collection diagnostics market appears promising, driven by ongoing investments in healthcare infrastructure and technological innovation. As the government continues to enhance healthcare facilities and promote research and development, the adoption of advanced diagnostic tools is expected to rise. Additionally, the increasing focus on personalized medicine will likely lead to tailored diagnostic solutions, further propelling market growth and improving patient outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Blood Collection Devices Urine Collection Devices Sputum Collection Devices Cerebrospinal Fluid (CSF) Collection Devices Saliva Collection Devices Other Body Fluid Collection Devices (e.g., synovial, pleural, peritoneal) |
| By End-User | Hospitals and Clinics Diagnostic Laboratories Research & Academic Institutions Home Care Settings Others (e.g., blood banks, mobile units) |
| By Application | Infectious Disease Diagnostics Oncology (Cancer) Diagnostics Cardiometabolic Disease Diagnostics Drug Testing & Toxicology Disease Screening & Routine Health Checks Others |
| By Distribution Channel | Direct Sales Distributors/Wholesalers Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Technology | Manual Collection Techniques Automated Collection Systems Point-of-Care Collection Devices Next-Generation Sequencing (NGS) & Molecular Diagnostics Others |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 120 | Laboratory Managers, Medical Technologists |
| Diagnostic Centers | 90 | Clinical Pathologists, Operations Managers |
| Healthcare Providers | 80 | General Practitioners, Nurses |
| Research Institutions | 60 | Research Scientists, Lab Technicians |
| Regulatory Bodies | 40 | Health Policy Analysts, Regulatory Affairs Specialists |
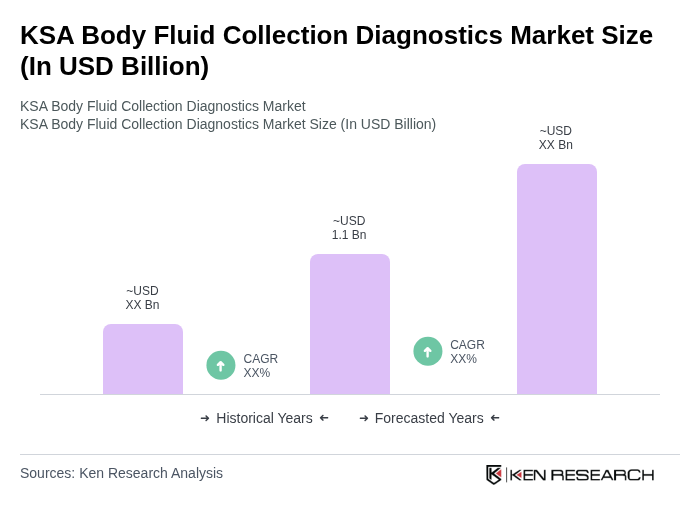
The KSA Body Fluid Collection Diagnostics Market is valued at approximately USD 1.1 billion, reflecting significant growth driven by the rising prevalence of chronic diseases and advancements in diagnostic technologies.