Region:Middle East
Author(s):Rebecca
Product Code:KRAC4621
Pages:93
Published On:October 2025
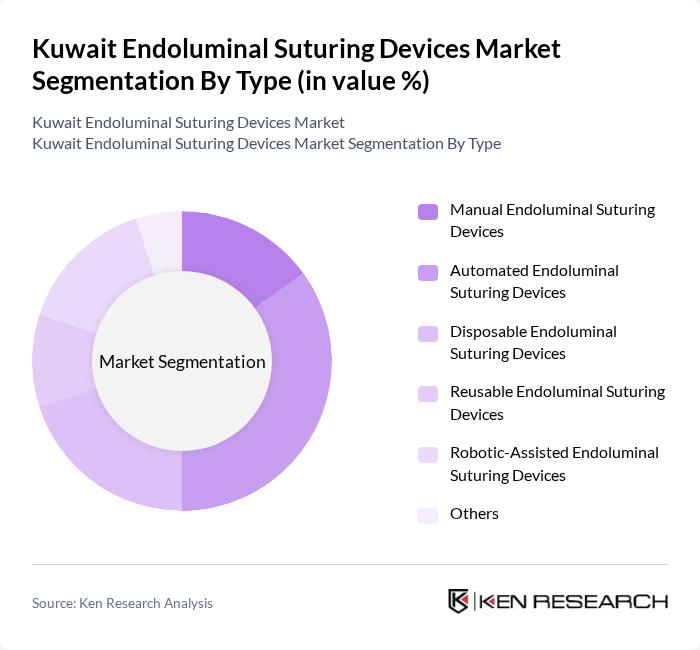
By Type:The market is segmented into Manual Endoluminal Suturing Devices, Automated Endoluminal Suturing Devices, Disposable Endoluminal Suturing Devices, Reusable Endoluminal Suturing Devices, Robotic-Assisted Endoluminal Suturing Devices, and Others. Automated Endoluminal Suturing Devices are gaining significant traction due to their superior precision, reduced procedural time, and enhanced outcomes in complex gastrointestinal surgeries. Robotic-assisted devices are also seeing increased adoption, particularly in tertiary care centers, as they enable advanced minimally invasive interventions.
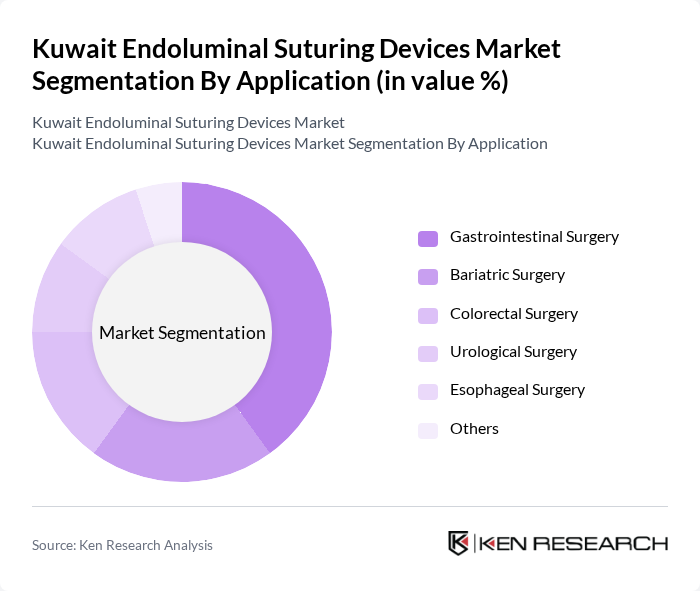
By Application:Endoluminal suturing devices are primarily used in Gastrointestinal Surgery, Bariatric Surgery, Colorectal Surgery, Urological Surgery, Esophageal Surgery, and Others. Gastrointestinal Surgery is the leading application segment, reflecting the high incidence of gastrointestinal diseases and the strong clinical preference for minimally invasive techniques. Bariatric and colorectal surgeries also represent substantial demand, driven by rising obesity rates and increased screening for colorectal disorders.
The Kuwait Endoluminal Suturing Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (Ethicon Endo-Surgery), Apollo Endosurgery, Inc., Boston Scientific Corporation, Olympus Corporation, CONMED Corporation, Cook Medical LLC, B. Braun Melsungen AG, Intuitive Surgical, Inc., Medline Industries, LP, Merit Medical Systems, Inc., Teleflex Incorporated, Aesculap, Inc. (a B. Braun company), Surgical Innovations Group plc, Ovesco Endoscopy AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait endoluminal suturing devices market appears promising, driven by technological advancements and increasing healthcare investments. As the government prioritizes healthcare innovation, the integration of AI and robotic-assisted surgeries is expected to enhance surgical precision and outcomes. Additionally, the growing emphasis on outpatient procedures will likely lead to a higher demand for minimally invasive solutions, positioning endoluminal suturing devices as a critical component of modern surgical practices in Kuwait.
| Segment | Sub-Segments |
|---|---|
| By Type | Manual Endoluminal Suturing Devices Automated Endoluminal Suturing Devices Disposable Endoluminal Suturing Devices Reusable Endoluminal Suturing Devices Robotic-Assisted Endoluminal Suturing Devices Others |
| By Application | Gastrointestinal Surgery Bariatric Surgery Colorectal Surgery Urological Surgery Esophageal Surgery Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Research & Academic Institutions Others |
| By Distribution Channel | Direct Sales Distributors/Dealers Online Sales Others |
| By Region | Capital Governorate (Kuwait City) Hawalli Governorate Farwaniya Governorate Ahmadi Governorate Mubarak Al-Kabeer Governorate Jahra Governorate Others |
| By Price Range | Low Price Range Mid Price Range High Price Range Others |
| By Brand | Established Brands Emerging Brands Private Labels Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gastroenterology Surgical Procedures | 90 | Surgeons, Gastroenterologists |
| Hospital Procurement Insights | 70 | Procurement Managers, Supply Chain Managers |
| Medical Device Distribution Channels | 50 | Distributors, Sales Managers |
| Clinical Application Feedback | 60 | Clinical Researchers, Medical Practitioners |
| Regulatory Compliance and Trends | 45 | Regulatory Affairs Specialists, Compliance Officers |
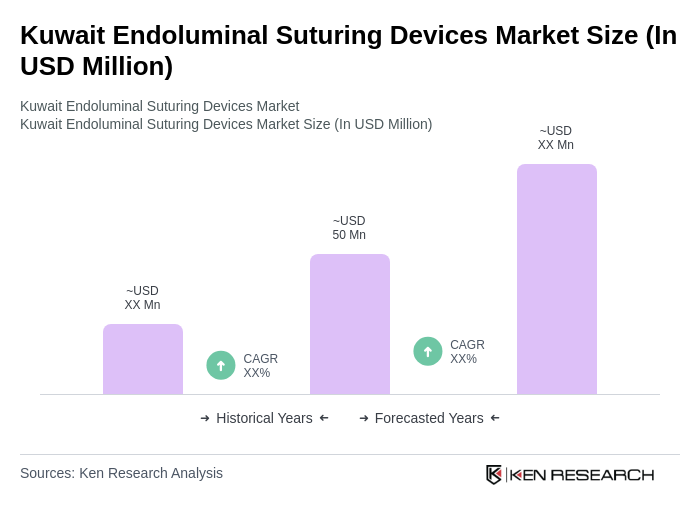
The Kuwait Endoluminal Suturing Devices Market is valued at approximately USD 50 million, reflecting strong demand driven by the increasing prevalence of gastrointestinal disorders and the rising adoption of minimally invasive surgical procedures.