About the Report
Base Year 2024Kuwait Orphan Drugs Market Overview
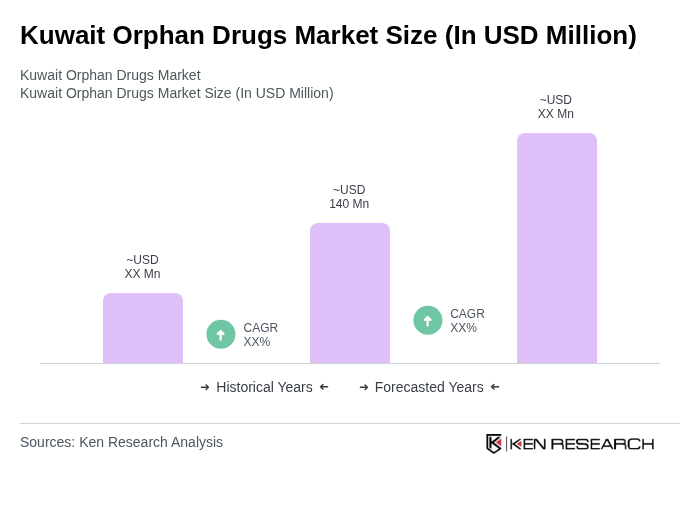
- The Kuwait Orphan Drugs Market is valued at USD 140 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of rare diseases, advancements in biotechnology, and a growing awareness of orphan drugs among healthcare professionals and patients. The market is also supported by government initiatives aimed at improving healthcare access and affordability for rare disease treatments.
- Kuwait City is the dominant region in the orphan drugs market due to its advanced healthcare infrastructure and the presence of leading pharmaceutical companies. The concentration of healthcare facilities and specialized medical professionals in the capital enhances the accessibility of orphan drugs, making it a focal point for patients seeking treatment for rare diseases. Additionally, the government's commitment to healthcare innovation further solidifies its position.
- The Ministerial Resolution No. 606 of 2021 issued by the Ministry of Health mandates a streamlined registration and approval process for orphan drugs, requiring submission of clinical data specific to rare diseases affecting fewer than 200 patients in Kuwait, with expedited review timelines not exceeding 90 days for priority cases. This regulation covers drugs for conditions designated as rare, enforces compliance through post-marketing surveillance, and requires manufacturers to meet Good Manufacturing Practice standards for licensing.
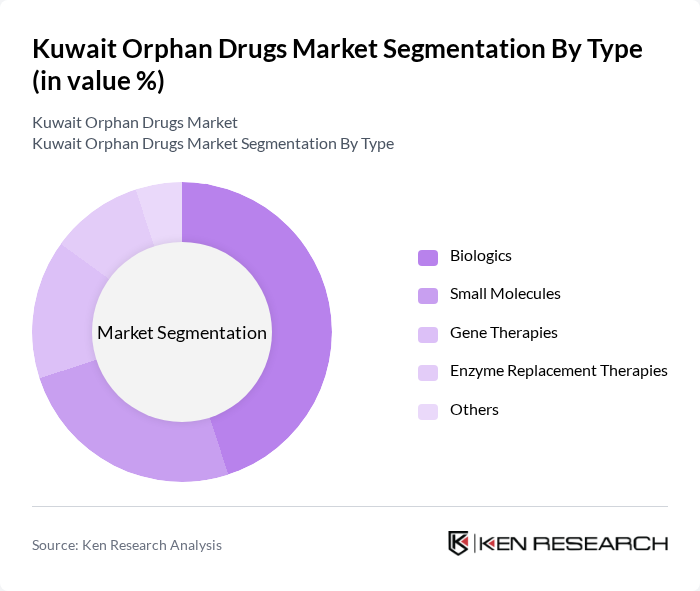
Kuwait Orphan Drugs Market Segmentation
By Type:The orphan drugs market can be segmented into various types, including Biologics, Small Molecules, Gene Therapies, Enzyme Replacement Therapies, and Others. Among these, Biologics are currently leading the market due to their effectiveness in treating complex rare diseases and their growing acceptance among healthcare providers. The increasing investment in research and development of biologics is also contributing to their dominance in the market.
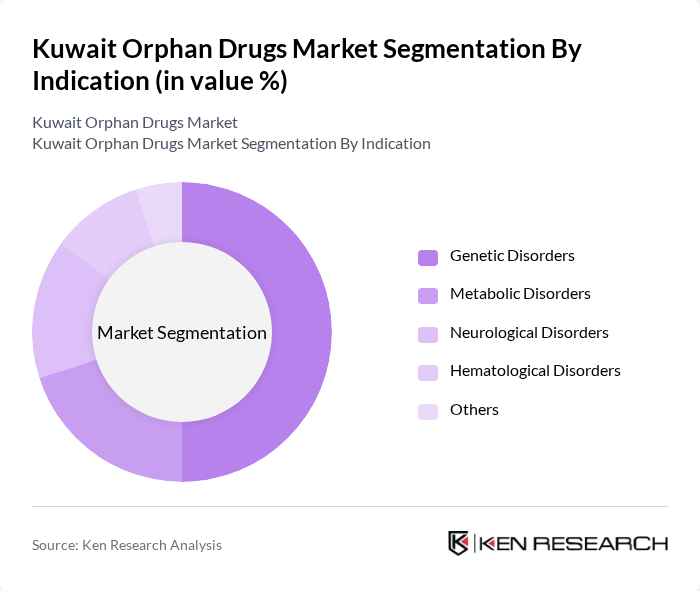
By Indication:The orphan drugs market is also categorized by indications, including Genetic Disorders, Metabolic Disorders, Neurological Disorders, Hematological Disorders, and Others. Genetic Disorders are the leading indication segment, driven by the high prevalence of conditions such as cystic fibrosis and muscular dystrophy. The increasing focus on genetic research and the development of targeted therapies are further propelling this segment's growth.
Kuwait Orphan Drugs Market Competitive Landscape
The Kuwait Orphan Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Novartis AG, Sanofi S.A., Roche Holding AG, Amgen Inc., Vertex Pharmaceuticals Incorporated, BioMarin Pharmaceutical Inc., Alexion Pharmaceuticals, Inc., Takeda Pharmaceutical Company Limited, Regeneron Pharmaceuticals, Inc., Gilead Sciences, Inc., CSL Behring, Incyte Corporation, Ultragenyx Pharmaceutical Inc., Horizon Therapeutics plc contribute to innovation, geographic expansion, and service delivery in this space.
Kuwait Orphan Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Rare Diseases:The prevalence of rare diseases in Kuwait is estimated to affect approximately 7% of the population, translating to around 200,000 individuals. This growing patient base drives demand for orphan drugs, as healthcare providers seek effective treatments. The World Health Organization reports that the number of rare diseases is increasing, with over 7,000 identified globally, further emphasizing the need for targeted therapies in Kuwait's healthcare system.
- Government Initiatives for Orphan Drug Development:The Kuwaiti government has allocated approximately $50 million in future to support orphan drug research and development. This funding aims to incentivize pharmaceutical companies to invest in rare disease treatments. Additionally, the Ministry of Health is implementing policies to streamline the approval process for orphan drugs, which is expected to enhance market access and encourage innovation in the sector.
- Advancements in Biotechnology:The biotechnology sector in Kuwait is projected to grow by 15% annually, driven by innovations in gene therapy and biologics. In future, investments in biotech research are expected to reach $30 million, fostering the development of orphan drugs. These advancements enable the creation of targeted therapies that address the unique needs of patients with rare diseases, thereby expanding treatment options and improving patient outcomes.
Market Challenges
- High Cost of Orphan Drugs:The average cost of orphan drugs in Kuwait can exceed $100,000 per patient annually, creating significant financial barriers for healthcare systems and patients. This high pricing often leads to limited access to essential treatments, as insurance coverage may not fully reimburse these costs. Consequently, many patients may remain untreated, exacerbating health disparities within the population.
- Regulatory Hurdles:The regulatory framework for orphan drugs in Kuwait is still evolving, with complex approval processes that can delay market entry. In future, it is estimated that the average time for drug approval could extend to 18 months, hindering timely access to innovative therapies. These regulatory challenges can deter pharmaceutical companies from investing in the orphan drug market, limiting treatment options for patients.
Kuwait Orphan Drugs Market Future Outlook
The future of the orphan drugs market in Kuwait appears promising, driven by increasing government support and advancements in biotechnology. As healthcare infrastructure expands, more patients will gain access to innovative therapies. Additionally, the rise of telemedicine and digital health technologies is expected to enhance patient management and monitoring, facilitating better outcomes. The focus on personalized medicine will further drive the development of targeted therapies, addressing the unique needs of patients with rare diseases.
Market Opportunities
- Expansion of Healthcare Infrastructure:Kuwait's healthcare expenditure is projected to reach $10 billion in future, facilitating the expansion of healthcare facilities. This growth presents opportunities for orphan drug manufacturers to establish partnerships with hospitals and clinics, enhancing distribution channels and improving patient access to rare disease treatments.
- Increased Investment in R&D:With the government’s commitment to allocate $50 million for orphan drug research, there is a significant opportunity for collaboration between public and private sectors. This investment can lead to breakthroughs in rare disease treatments, fostering innovation and attracting global pharmaceutical companies to Kuwait's burgeoning market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Biologics Small Molecules Gene Therapies Enzyme Replacement Therapies Others |
| By Indication | Genetic Disorders Metabolic Disorders Neurological Disorders Hematological Disorders Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Specialty Pharmacies Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Region | Capital Governorate Hawalli Governorate Al Ahmadi Governorate Farwaniya Governorate Others |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-Based Pricing Others |
| By Market Access Strategy | Direct Access Indirect Access Partnership Models Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Kuwait Ministry of Health)
Pharmaceutical Manufacturers and Producers
Distributors and Wholesalers
Healthcare Providers and Hospitals
Patient Advocacy Groups
Health Insurance Companies
Biotechnology Firms
Players Mentioned in the Report:
Pfizer Inc.
Novartis AG
Sanofi S.A.
Roche Holding AG
Amgen Inc.
Vertex Pharmaceuticals Incorporated
BioMarin Pharmaceutical Inc.
Alexion Pharmaceuticals, Inc.
Takeda Pharmaceutical Company Limited
Regeneron Pharmaceuticals, Inc.
Gilead Sciences, Inc.
CSL Behring
Incyte Corporation
Ultragenyx Pharmaceutical Inc.
Horizon Therapeutics plc
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Kuwait Orphan Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Kuwait Orphan Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Kuwait Orphan Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of rare diseases
3.1.2 Government initiatives for orphan drug development
3.1.3 Rising healthcare expenditure
3.1.4 Advancements in biotechnology
3.2 Market Challenges
3.2.1 High cost of orphan drugs
3.2.2 Limited patient population
3.2.3 Regulatory hurdles
3.2.4 Lack of awareness among healthcare providers
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with pharmaceutical companies
3.3.3 Increased investment in R&D
3.3.4 Growing demand for personalized medicine
3.4 Market Trends
3.4.1 Shift towards patient-centric approaches
3.4.2 Rise of telemedicine in rare disease management
3.4.3 Integration of digital health technologies
3.4.4 Focus on gene therapies and biologics
3.5 Government Regulation
3.5.1 Orphan drug designation policies
3.5.2 Pricing and reimbursement frameworks
3.5.3 Clinical trial regulations
3.5.4 Market exclusivity provisions
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Kuwait Orphan Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Kuwait Orphan Drugs Market Segmentation
8.1 By Type
8.1.1 Biologics
8.1.2 Small Molecules
8.1.3 Gene Therapies
8.1.4 Enzyme Replacement Therapies
8.1.5 Others
8.2 By Indication
8.2.1 Genetic Disorders
8.2.2 Metabolic Disorders
8.2.3 Neurological Disorders
8.2.4 Hematological Disorders
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Hospital Pharmacies
8.3.2 Retail Pharmacies
8.3.3 Online Pharmacies
8.3.4 Specialty Pharmacies
8.3.5 Others
8.4 By Patient Demographics
8.4.1 Pediatric Patients
8.4.2 Adult Patients
8.4.3 Geriatric Patients
8.4.4 Others
8.5 By Region
8.5.1 Capital Governorate
8.5.2 Hawalli Governorate
8.5.3 Al Ahmadi Governorate
8.5.4 Farwaniya Governorate
8.5.5 Others
8.6 By Pricing Strategy
8.6.1 Premium Pricing
8.6.2 Competitive Pricing
8.6.3 Value-Based Pricing
8.6.4 Others
8.7 By Market Access Strategy
8.7.1 Direct Access
8.7.2 Indirect Access
8.7.3 Partnership Models
8.7.4 Others
9. Kuwait Orphan Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Development Cycle Time
9.2.8 Regulatory Approval Success Rate
9.2.9 Distribution Efficiency
9.2.10 Brand Recognition Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pfizer Inc.
9.5.2 Novartis AG
9.5.3 Sanofi S.A.
9.5.4 Roche Holding AG
9.5.5 Amgen Inc.
9.5.6 Vertex Pharmaceuticals Incorporated
9.5.7 BioMarin Pharmaceutical Inc.
9.5.8 Alexion Pharmaceuticals, Inc.
9.5.9 Takeda Pharmaceutical Company Limited
9.5.10 Regeneron Pharmaceuticals, Inc.
9.5.11 Gilead Sciences, Inc.
9.5.12 CSL Behring
9.5.13 Incyte Corporation
9.5.14 Ultragenyx Pharmaceutical Inc.
9.5.15 Horizon Therapeutics plc
10. Kuwait Orphan Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Social Affairs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Pharmaceutical R&D Expenditure
10.2.3 Public Health Initiatives
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Healthcare Providers
10.3.2 Patients and Caregivers
10.3.3 Pharmaceutical Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Support Needs
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcomes Improvement
10.5.3 Market Expansion Opportunities
10.5.4 Others
11. Kuwait Orphan Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing market reports and publications on orphan drugs in Kuwait
- Review of government health policies and regulations affecting orphan drug approvals
- Examination of demographic data and disease prevalence statistics relevant to orphan drug usage
Primary Research
- Interviews with healthcare professionals specializing in rare diseases
- Surveys conducted with pharmaceutical companies involved in orphan drug development
- Focus groups with patient advocacy organizations and caregivers of patients with rare diseases
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including academic journals and industry reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks through expert panels comprising healthcare economists and industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Kuwait allocated to orphan drugs
- Analysis of the market share of orphan drugs within the broader pharmaceutical market
- Incorporation of government funding and subsidies for orphan drug development
Bottom-up Modeling
- Collection of sales data from key orphan drug manufacturers operating in Kuwait
- Estimation of patient population sizes for specific rare diseases treated by orphan drugs
- Calculation of average treatment costs and frequency of drug administration
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as healthcare policy changes and market access
- Scenario modeling based on potential new drug approvals and market entry of generics
- Baseline, optimistic, and pessimistic forecasts for orphan drug market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals in Rare Diseases | 120 | Oncologists, Geneticists, Rare Disease Specialists |
| Pharmaceutical Companies in Orphan Drug Development | 45 | Product Managers, Regulatory Affairs Specialists |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Caregivers, Patient Representatives |
| Health Economists and Policy Makers | 50 | Health Policy Analysts, Economic Researchers |
| Insurance Providers and Payers | 70 | Claims Analysts, Pharmacy Benefit Managers |
Frequently Asked Questions
What is the current value of the Kuwait Orphan Drugs Market?
The Kuwait Orphan Drugs Market is valued at approximately USD 140 million, reflecting a five-year historical analysis. This growth is attributed to the rising prevalence of rare diseases and advancements in biotechnology, alongside increased awareness among healthcare professionals and patients.