About the Report
Base Year 2024Indonesia Orphan Drugs Market Overview
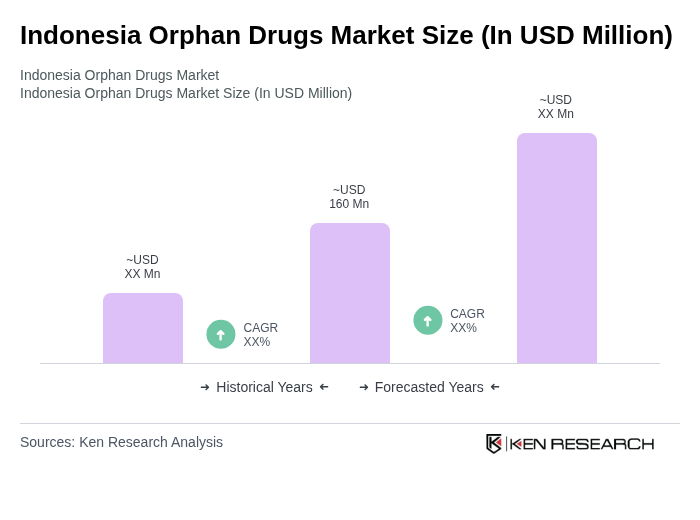
- The Indonesia Orphan Drugs Market is valued at USD 160 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of rare diseases, advancements in biotechnology and precision medicine, and supportive government policies aimed at improving healthcare access for patients with rare conditions.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their robust healthcare infrastructure, concentration of pharmaceutical companies, and higher patient populations. These urban centers are pivotal in driving research, development, and distribution of orphan drugs, making them critical hubs for market growth.
- The Minister of Health Regulation No. 4 of 2019 issued by the Ministry of Health requires pharmaceutical companies to register orphan drugs through a special pathway, ensuring expedited review processes and market exclusivity for up to five years to facilitate availability for rare disease treatments. This initiative aims to stimulate innovation and ensure that patients with rare diseases have access to necessary treatments.
Indonesia Orphan Drugs Market Segmentation
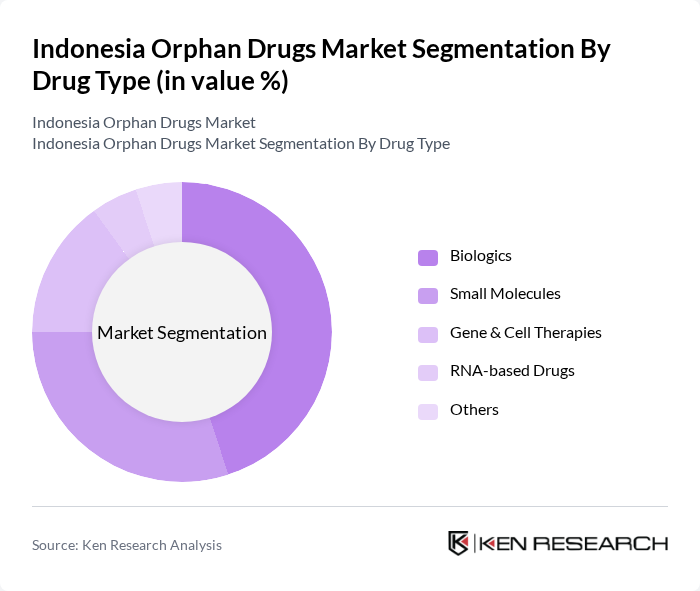
By Drug Type:The orphan drugs market can be segmented into various drug types, including Biologics, Small Molecules, Gene & Cell Therapies, RNA-based Drugs, and Others. Each of these subsegments plays a crucial role in addressing the unique needs of patients with rare diseases. Biologics are particularly dominant due to their effectiveness in treating complex conditions, while small molecules are widely used for their versatility and ease of administration.
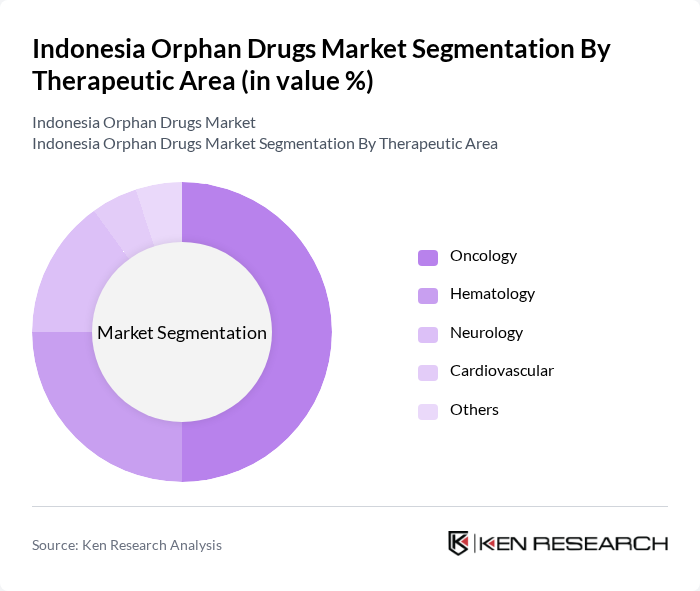
By Therapeutic Area:The therapeutic areas for orphan drugs include Oncology, Hematology, Neurology, Cardiovascular, and Others. Oncology is the leading segment, driven by the high prevalence of rare cancers and the increasing number of targeted therapies. Hematology follows closely, with a growing focus on blood disorders, while Neurology is gaining traction due to advancements in treatments for rare neurological conditions.
Indonesia Orphan Drugs Market Competitive Landscape
The Indonesia Orphan Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Indofarma Tbk, PT Merck Indonesia, PT Sanofi Indonesia, PT Novartis Indonesia, PT Pfizer Indonesia, PT Roche Indonesia, PT AstraZeneca Indonesia, PT Amgen Indonesia, PT Takeda Indonesia, PT GSK Indonesia, PT Bayer Indonesia, PT Biogen Indonesia, PT Elysium Healthcare contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Orphan Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Rare Diseases:The prevalence of rare diseases in Indonesia is estimated to affect approximately 7,000 individuals per 100,000 population, translating to around 1.9 million people. This growing patient population drives demand for orphan drugs, as healthcare providers seek effective treatments. The World Health Organization (WHO) emphasizes the need for targeted therapies, which further propels the market. As awareness increases, more patients are diagnosed, leading to a higher demand for specialized medications.
- Government Initiatives for Orphan Drug Development:The Indonesian government has implemented several initiatives to support orphan drug development, including the establishment of a regulatory framework that encourages research and innovation. In future, the government allocated approximately IDR 1.6 trillion (around USD 105 million) to fund research projects focused on rare diseases. These initiatives aim to enhance local production capabilities and reduce dependency on imported drugs, fostering a more sustainable healthcare environment for rare disease patients.
- Rising Healthcare Expenditure:Indonesia's healthcare expenditure is projected to reach IDR 1,300 trillion (approximately USD 85 billion) in future, reflecting a significant increase in government and private sector investment. This rise in spending is crucial for improving healthcare infrastructure and access to orphan drugs. Enhanced funding allows for better diagnostic tools and treatment options, ultimately benefiting patients with rare diseases. Increased healthcare budgets also facilitate the establishment of specialized centers for rare disease management.
Market Challenges
- High Cost of Orphan Drugs:The average cost of orphan drugs in Indonesia can exceed IDR 600 million (around USD 40,000) per patient annually, creating significant financial barriers for many families. This high cost limits access to essential treatments, particularly in a country where the average annual income is approximately IDR 70 million (USD 4,600). Consequently, many patients may forgo necessary therapies, leading to poorer health outcomes and increased long-term healthcare costs.
- Regulatory Hurdles and Lengthy Approval Processes:The regulatory landscape for orphan drugs in Indonesia is complex, with approval processes often taking over 20 months. This delay can hinder timely access to critical therapies for patients. The Indonesian Food and Drug Authority (BPOM) has been working to streamline these processes, but challenges remain. Lengthy approvals can discourage pharmaceutical companies from investing in orphan drug development, limiting the availability of new treatments in the market.
Indonesia Orphan Drugs Market Future Outlook
The future of the orphan drugs market in Indonesia appears promising, driven by increasing government support and a growing focus on personalized medicine. As healthcare expenditure rises, more resources will be allocated to research and development, fostering innovation in rare disease treatments. Additionally, the expansion of telemedicine and digital health solutions will enhance patient access to specialized care, improving diagnosis and treatment outcomes. These trends indicate a robust growth trajectory for the orphan drugs sector in the coming years.
Market Opportunities
- Expansion of Telemedicine and Digital Health Solutions:The rise of telemedicine in Indonesia presents a significant opportunity for orphan drug providers. By leveraging digital platforms, healthcare professionals can reach patients in remote areas, facilitating timely diagnosis and treatment. In future, the telemedicine market is expected to grow to IDR 12 trillion (approximately USD 800 million), enhancing access to rare disease therapies and improving patient outcomes.
- Collaborations with International Pharmaceutical Companies:Partnerships with global pharmaceutical firms can enhance local capabilities in orphan drug development. Collaborations can lead to knowledge transfer, improved research methodologies, and access to advanced technologies. In future, Indonesia aims to attract foreign direct investment of around USD 1.2 billion in the healthcare sector, which could significantly boost the orphan drugs market and improve treatment availability for rare diseases.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Type | Biologics Small Molecules Gene & Cell Therapies RNA-based Drugs Others |
| By Therapeutic Area | Oncology Hematology Neurology Cardiovascular Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Specialty Pharmacies Others |
| By Patient Demographics | Pediatric patients Adult patients Geriatric patients Others |
| By Region | Java Sumatra Bali Kalimantan Others |
| By Treatment Setting | Inpatient Outpatient Home care Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Pharmaceutical Manufacturers and Producers
Biotechnology Companies
Healthcare Providers and Hospitals
Patient Advocacy Groups
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Health Insurance Companies
Players Mentioned in the Report:
PT Kalbe Farma Tbk
PT Kimia Farma Tbk
PT Indofarma Tbk
PT Merck Indonesia
PT Sanofi Indonesia
PT Novartis Indonesia
PT Pfizer Indonesia
PT Roche Indonesia
PT AstraZeneca Indonesia
PT Amgen Indonesia
PT Takeda Indonesia
PT GSK Indonesia
PT Bayer Indonesia
PT Biogen Indonesia
PT Elysium Healthcare
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Orphan Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Orphan Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Orphan Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of rare diseases
3.1.2 Government initiatives for orphan drug development
3.1.3 Rising healthcare expenditure
3.1.4 Enhanced awareness and diagnosis of rare diseases
3.2 Market Challenges
3.2.1 High cost of orphan drugs
3.2.2 Limited market access and distribution channels
3.2.3 Regulatory hurdles and lengthy approval processes
3.2.4 Lack of awareness among healthcare professionals
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and digital health solutions
3.3.2 Collaborations with international pharmaceutical companies
3.3.3 Development of biosimilars for orphan drugs
3.3.4 Increasing investment in R&D for rare diseases
3.4 Market Trends
3.4.1 Growing focus on personalized medicine
3.4.2 Rise in patient advocacy groups
3.4.3 Shift towards value-based healthcare models
3.4.4 Increased use of real-world evidence in drug development
3.5 Government Regulation
3.5.1 Orphan drug designation and incentives
3.5.2 Pricing regulations for rare disease treatments
3.5.3 Clinical trial regulations specific to orphan drugs
3.5.4 Health technology assessment requirements
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Orphan Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Orphan Drugs Market Segmentation
8.1 By Drug Type
8.1.1 Biologics
8.1.2 Small Molecules
8.1.3 Gene & Cell Therapies
8.1.4 RNA-based Drugs
8.1.5 Others
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Hematology
8.2.3 Neurology
8.2.4 Cardiovascular
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Hospital Pharmacies
8.3.2 Retail Pharmacies
8.3.3 Online Pharmacies
8.3.4 Specialty Pharmacies
8.3.5 Others
8.4 By Patient Demographics
8.4.1 Pediatric patients
8.4.2 Adult patients
8.4.3 Geriatric patients
8.4.4 Others
8.5 By Region
8.5.1 Java
8.5.2 Sumatra
8.5.3 Bali
8.5.4 Kalimantan
8.5.5 Others
8.6 By Treatment Setting
8.6.1 Inpatient
8.6.2 Outpatient
8.6.3 Home care
8.6.4 Others
9. Indonesia Orphan Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Capitalization (USD Million)
9.2.3 Revenue Growth Rate (CAGR %)
9.2.4 Market Penetration Rate (%)
9.2.5 Orphan Drug Portfolio Size (Number of Approved Drugs)
9.2.6 Pricing Strategy (Premium/Mid-tier/Value)
9.2.7 R&D Investment as % of Revenue
9.2.8 Regulatory Approval Success Rate (%)
9.2.9 Distribution Network Coverage (%)
9.2.10 Brand Recognition Score (1-10)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Kalbe Farma Tbk
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Indofarma Tbk
9.5.4 PT Merck Indonesia
9.5.5 PT Sanofi Indonesia
9.5.6 PT Novartis Indonesia
9.5.7 PT Pfizer Indonesia
9.5.8 PT Roche Indonesia
9.5.9 PT AstraZeneca Indonesia
9.5.10 PT Amgen Indonesia
9.5.11 PT Takeda Indonesia
9.5.12 PT GSK Indonesia
9.5.13 PT Bayer Indonesia
9.5.14 PT Biogen Indonesia
9.5.15 PT Elysium Healthcare
10. Indonesia Orphan Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Social Affairs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for orphan drug research
10.2.3 Partnerships with NGOs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to treatment
10.3.2 Affordability of drugs
10.3.3 Availability of specialized care
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of orphan drugs
10.4.2 Training for healthcare providers
10.4.3 Patient education initiatives
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings from early intervention
10.5.2 Improved patient outcomes
10.5.3 Expansion into new therapeutic areas
10.5.4 Others
11. Indonesia Orphan Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Model Exploration
1.4 Customer Segmentation
1.5 Competitive Landscape Analysis
1.6 Key Partnerships
1.7 Risk Assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Event Marketing
2.7 Feedback Mechanisms
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
3.2 Logistics and Supply Chain Management
3.3 Distribution Partnerships
3.4 Inventory Management
3.5 Channel Optimization
3.6 Performance Metrics
3.7 Risk Mitigation Strategies
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
4.3 Competitor Pricing Analysis
4.4 Customer Willingness to Pay
4.5 Price Sensitivity Analysis
4.6 Value-Based Pricing Strategies
4.7 Pricing Adjustments
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
5.3 Emerging Trends
5.4 Customer Feedback Analysis
5.5 Market Research Insights
5.6 Product Development Opportunities
5.7 Strategic Recommendations
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer Engagement Strategies
6.4 Feedback and Improvement Loops
6.5 Community Building
6.6 Customer Support Systems
6.7 Relationship Management Tools
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
7.5 Competitive Advantages
7.6 Market Differentiation Strategies
7.7 Long-term Value Creation
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
8.4 Market Research
8.5 Training and Development
8.6 Performance Monitoring
8.7 Strategic Partnerships
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.1.4 Marketing approach
9.1.5 Distribution channels
9.1.6 Regulatory considerations
9.1.7 Risk assessment
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
9.2.3 Market entry barriers
9.2.4 Distribution partnerships
9.2.5 Pricing strategies
9.2.6 Marketing strategies
9.2.7 Risk management
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Fit
10.7 Long-term Viability
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
11.3 Funding Sources
11.4 Financial Projections
11.5 Risk Assessment
11.6 Contingency Planning
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
12.3 Long-term Strategy
12.4 Performance Metrics
12.5 Exit Strategies
12.6 Stakeholder Engagement
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Financial Health Indicators
13.4 Market Positioning
13.5 Strategic Recommendations
13.6 Risk Assessment
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
14.4 Strategic Alliances
14.5 Partnership Evaluation Criteria
14.6 Risk Assessment
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
15.2.3 Performance Metrics
15.2.4 Risk Management
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health reports and orphan drug policies in Indonesia
- Review of published market studies and white papers on orphan drugs
- Examination of pharmaceutical industry databases for orphan drug approvals and market trends
Primary Research
- Interviews with healthcare professionals specializing in rare diseases
- Surveys with pharmaceutical companies involved in orphan drug development
- Focus groups with patient advocacy organizations and caregivers
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including academic journals and industry reports
- Triangulation of qualitative insights from interviews with quantitative data from market reports
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Indonesia allocated to orphan drugs
- Analysis of the prevalence of rare diseases and corresponding treatment costs
- Incorporation of government funding and subsidies for orphan drug development
Bottom-up Modeling
- Collection of sales data from leading pharmaceutical companies in the orphan drug sector
- Estimation of patient population sizes for specific rare diseases
- Calculation of average treatment costs based on existing orphan drug pricing
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as healthcare access and economic growth
- Scenario modeling based on potential regulatory changes and market entry of new therapies
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers in Rare Disease Treatment | 100 | Oncologists, Hematologists, Geneticists |
| Pharmaceutical Companies Developing Orphan Drugs | 45 | Product Managers, R&D Directors |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Patient Representatives |
| Regulatory Bodies and Health Authorities | 50 | Policy Makers, Regulatory Affairs Specialists |
| Insurance Providers Covering Orphan Drugs | 70 | Claims Managers, Underwriters |
Frequently Asked Questions
What is the current value of the Indonesia Orphan Drugs Market?
The Indonesia Orphan Drugs Market is valued at approximately USD 160 million, reflecting a significant growth driven by increased awareness of rare diseases, advancements in biotechnology, and supportive government policies aimed at improving healthcare access for patients with rare conditions.