Region:Middle East
Author(s):Geetanshi
Product Code:KRAD4833
Pages:91
Published On:December 2025
By Molecule Type:The molecule type segmentation of the market includes various subsegments that cater to different therapeutic needs. Among these, cytotoxic payload peptide-drug conjugates are leading due to their effectiveness in targeting cancer cells while minimizing damage to healthy tissues, in line with global trends where therapeutic peptide drug conjugates are predominantly developed for oncology indications. Radioligand peptide-drug conjugates are also gaining traction, particularly in oncology, as radiolabeled peptides such as lutetium-177–based agents have demonstrated strong clinical utility in neuroendocrine tumors and prostate cancer through targeted delivery to tumor receptors. The increasing focus on personalized medicine and precision oncology, supported by biomarker-driven patient selection, is driving the demand for these advanced therapeutic options across the Middle East, including Kuwait.

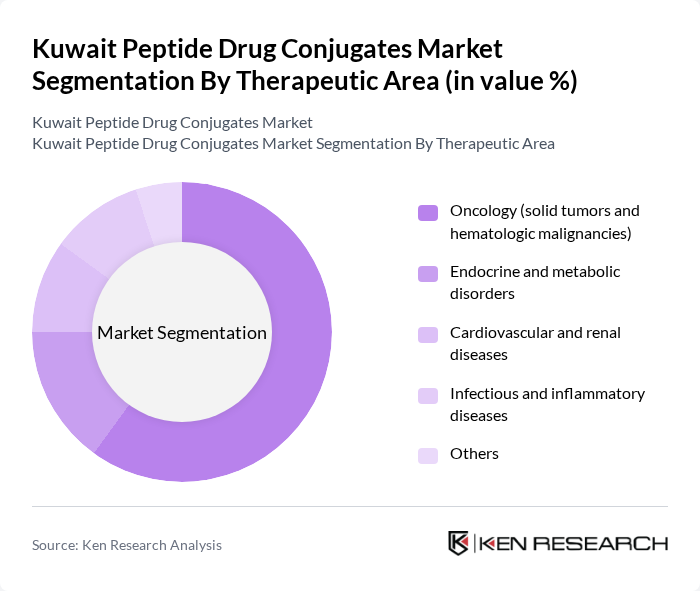
By Therapeutic Area:The therapeutic area segmentation highlights the diverse applications of peptide drug conjugates. Oncology remains the leading therapeutic area, driven by the increasing incidence of cancer and the need for targeted therapies that improve tumor selectivity and reduce systemic toxicity. The focus on solid tumors and hematologic malignancies is particularly pronounced, as these areas present significant unmet medical needs and are the primary focus of most peptide drug conjugate pipelines and approved products worldwide. Other therapeutic areas, such as endocrine and metabolic disorders, cardiovascular and renal diseases, and infectious and inflammatory diseases, are also witnessing growth as research expands into non-oncology indications, but oncology continues to dominate due to the high demand for effective cancer treatments and the strong alignment of peptide drug conjugate mechanisms with tumor-targeted delivery.
The Kuwait Peptide Drug Conjugates Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, F. Hoffmann-La Roche Ltd, Takeda Pharmaceutical Company Limited, Ipsen Pharma, RayzeBio, Inc., Oncopeptides AB, Bicycle Therapeutics plc, CordenPharma International GmbH, WuXi STA (WuXi AppTec Co., Ltd.), Bachem Holding AG, PolyPeptide Group AG, Ajinomoto Bio-Pharma Services, Kuwait Cancer Control Center (KCCC), Ministry of Health – Kuwait (MoH Kuwait), Kuwait University – Faculty of Medicine & Health Sciences Research Center contribute to innovation, geographic expansion, and service delivery in this space.
The future of the peptide drug conjugates market in Kuwait appears promising, driven by ongoing advancements in biotechnology and a growing emphasis on personalized medicine. As healthcare infrastructure continues to expand, the integration of innovative therapies is expected to accelerate. Additionally, increased collaboration between pharmaceutical companies and research institutions will likely enhance the development of novel peptide-based therapies, addressing unmet medical needs and improving patient outcomes in the region.
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Cytotoxic payload peptide-drug conjugates Radioligand peptide-drug conjugates Peptide–siRNA/oligonucleotide conjugates Peptide–polymer conjugates Others |
| By Therapeutic Area | Oncology (solid tumors and hematologic malignancies) Endocrine and metabolic disorders Cardiovascular and renal diseases Infectious and inflammatory diseases Others |
| By Development Stage | Preclinical candidates Phase I clinical candidates Phase II–III clinical candidates Commercially approved products |
| By End-User | Tertiary care and cancer specialty hospitals Contract research organizations (CROs) Biopharmaceutical and biotech companies Academic and government research institutes Others |
| By Distribution Channel | Hospital pharmacies Specialty and oncology pharmacies Direct tender and institutional sales Others |
| By Manufacturing Approach | In-house GMP manufacturing Contract development and manufacturing organizations (CDMOs) Hybrid and partnered manufacturing models |
| By Kuwait Health System Cluster | Capital Region (Kuwait City and surrounding areas) Hawalli and Farwaniya Governorates Ahmadi and Mubarak Al-Kabeer Governorates Northern and border governorates |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Peptide Drug Conjugates | 100 | Oncologists, Clinical Researchers |
| Cardiovascular Peptide Applications | 80 | Cardiologists, Pharmacologists |
| Neurological Disorders Treatments | 70 | Neurologists, Biotech R&D Managers |
| Regulatory Affairs in Peptide Development | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Market Access and Pricing Strategies | 90 | Market Access Managers, Health Economists |
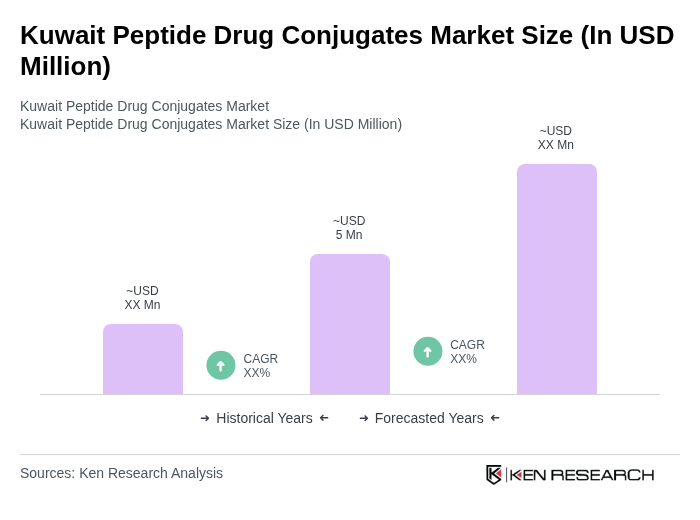
The Kuwait Peptide Drug Conjugates Market is valued at approximately USD 5 million, reflecting a five-year historical analysis. This valuation is influenced by advancements in targeted therapies and increasing investments in biopharmaceutical research.