Region:Asia
Author(s):Geetanshi
Product Code:KRAA6139
Pages:98
Published On:January 2026
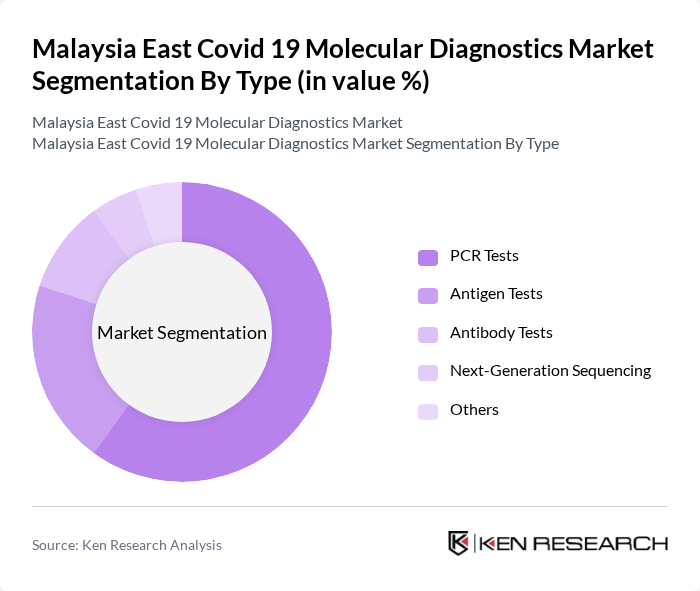
By Type:The market is segmented into various types of diagnostic tests, including PCR Tests, Antigen Tests, Antibody Tests, Next-Generation Sequencing, and Others. Among these, PCR Tests are the most widely used due to their high accuracy and reliability in detecting the virus. Antigen Tests are gaining traction for their rapid results, while Antibody Tests are utilized for seroprevalence studies. Next-Generation Sequencing is emerging as a critical tool for variant detection and epidemiological studies.
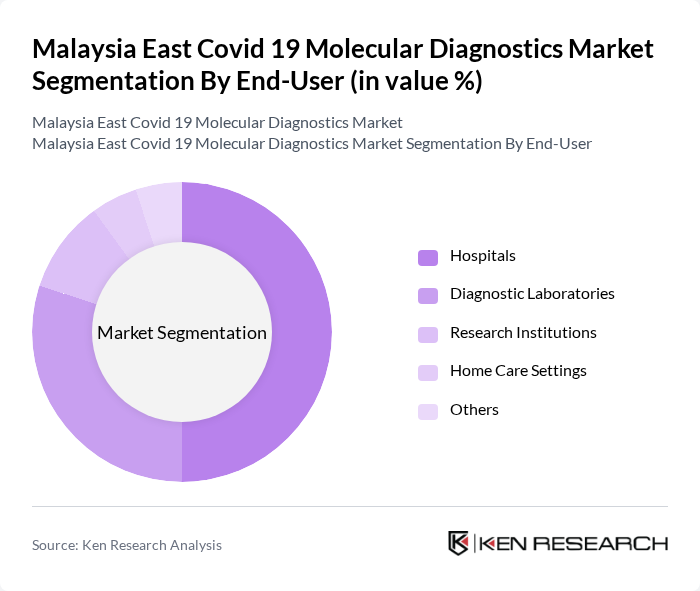
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Hospitals are the primary consumers of molecular diagnostics due to their need for accurate and timely testing for patient management. Diagnostic Laboratories follow closely, providing testing services to both hospitals and the public. Research Institutions utilize these diagnostics for studies and vaccine development, while Home Care Settings are increasingly adopting rapid testing solutions for convenience.
The Malaysia East Covid 19 Molecular Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as BioMérieux, Roche Diagnostics, Abbott Laboratories, Thermo Fisher Scientific, Cepheid, Hologic, Siemens Healthineers, Qiagen, BGI Genomics, GenMark Diagnostics, PerkinElmer, Agilent Technologies, Mylab Discovery Solutions, F. Hoffmann-La Roche AG, Danaher Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Covid-19 molecular diagnostics market in Malaysia East appears promising, driven by ongoing technological advancements and increased government support. As the healthcare sector continues to evolve, the integration of artificial intelligence and machine learning in diagnostics is expected to enhance testing accuracy and efficiency. Additionally, the growing trend towards telehealth services will likely facilitate remote testing and consultations, making diagnostics more accessible to the population, particularly in underserved areas.
| Segment | Sub-Segments |
|---|---|
| By Type | PCR Tests Antigen Tests Antibody Tests Next-Generation Sequencing Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Sample Type | Nasopharyngeal Swabs Saliva Samples Blood Samples Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Sabah Sarawak Labuan Others |
| By Testing Method | Molecular Testing Rapid Testing Others |
| By Technology | Real-Time PCR Digital PCR Microarray Technology Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Laboratories | 100 | Laboratory Directors, Public Health Officials |
| Private Diagnostic Centers | 80 | Clinical Pathologists, Operations Managers |
| Healthcare Providers | 75 | General Practitioners, Infectious Disease Specialists |
| Government Health Agencies | 60 | Policy Makers, Epidemiologists |
| Research Institutions | 50 | Research Scientists, Academic Professors |
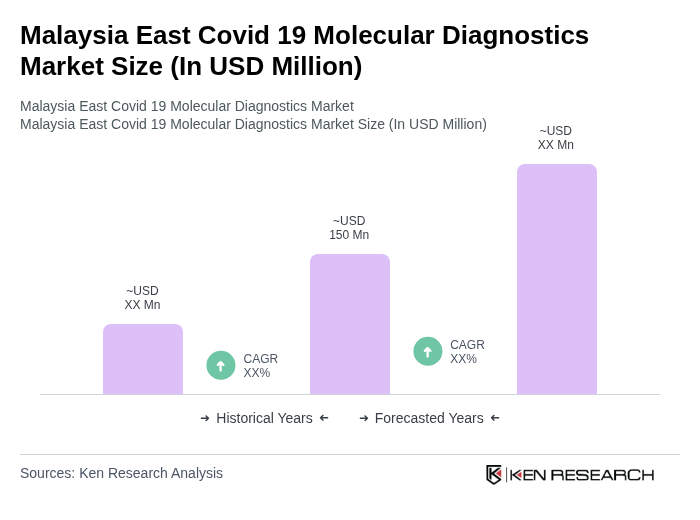
The Malaysia East Covid 19 Molecular Diagnostics Market is valued at approximately USD 150 million, reflecting a significant increase driven by the demand for accurate and rapid testing solutions during the ongoing pandemic.