Region:Asia
Author(s):Geetanshi
Product Code:KRAA6138
Pages:91
Published On:January 2026
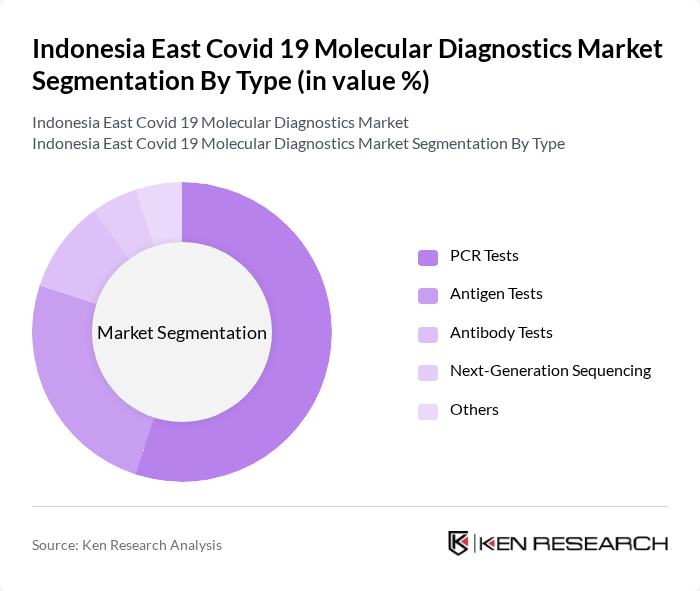
By Type:The market is segmented into various types of molecular diagnostic tests, including PCR Tests, Antigen Tests, Antibody Tests, Next-Generation Sequencing, and Others. Among these, PCR Tests are the most widely used due to their high sensitivity and specificity, making them the gold standard for Covid-19 diagnosis. The demand for Antigen Tests has also surged due to their rapid results, which are crucial for timely decision-making in clinical settings. Antibody Tests, while less frequently used for acute diagnosis, play a significant role in seroprevalence studies and understanding population immunity.
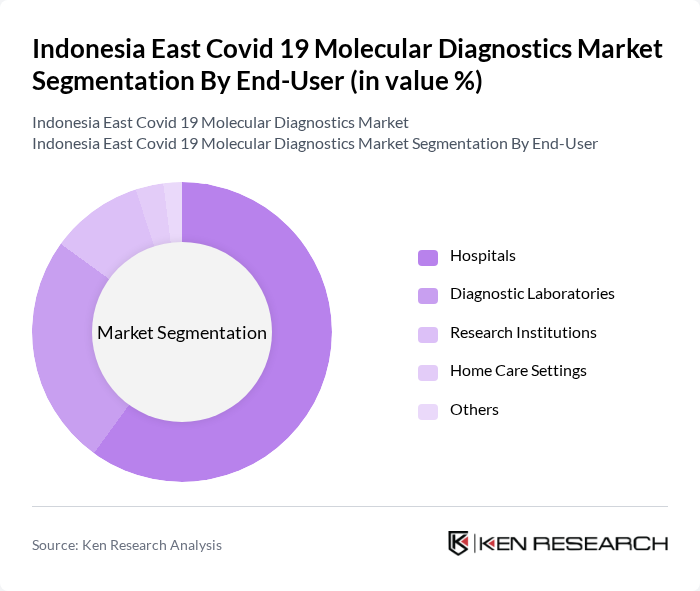
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Hospitals are the leading end-users of molecular diagnostics, driven by the need for accurate and timely testing in patient management. Diagnostic Laboratories also play a crucial role, as they are equipped with advanced technologies to perform a high volume of tests. Research Institutions utilize these diagnostics for epidemiological studies and vaccine development, while Home Care Settings are increasingly adopting testing kits for convenience and accessibility.
The Indonesia East Covid 19 Molecular Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bio-Rad Laboratories, Roche Diagnostics, Abbott Laboratories, Cepheid, Thermo Fisher Scientific, Hologic, Siemens Healthineers, BGI Genomics, Genomik Solidaritas, Medisafe Technologies, Genetika Science, Eijkelamp, Labcorp, PerkinElmer, Agilent Technologies contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Covid-19 molecular diagnostics market in Indonesia East appears promising, driven by technological advancements and increased healthcare funding. The integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, while the expansion of telehealth services will facilitate remote testing and consultations. As the government continues to invest in healthcare infrastructure, the market is poised for growth, with a focus on improving accessibility and affordability of testing solutions for all demographics.
| Segment | Sub-Segments |
|---|---|
| By Type | PCR Tests Antigen Tests Antibody Tests Next-Generation Sequencing Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Region | Java Sumatra Bali Kalimantan Others |
| By Technology | Real-Time PCR Digital PCR Microarray Technology Others |
| By Application | Clinical Diagnostics Research Applications Public Health Surveillance Others |
| By Investment Source | Government Funding Private Investments International Aid Others |
| By Policy Support | Subsidies for Testing Kits Tax Incentives for Manufacturers Grants for Research and Development Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Molecular Diagnostics Laboratories | 150 | Laboratory Directors, Chief Technologists |
| Healthcare Providers (Hospitals & Clinics) | 100 | Infectious Disease Specialists, Medical Directors |
| Government Health Officials | 80 | Public Health Officers, Policy Makers |
| Diagnostic Equipment Manufacturers | 70 | Sales Managers, Product Development Leads |
| Research Institutions & Universities | 60 | Research Scientists, Academic Professors |
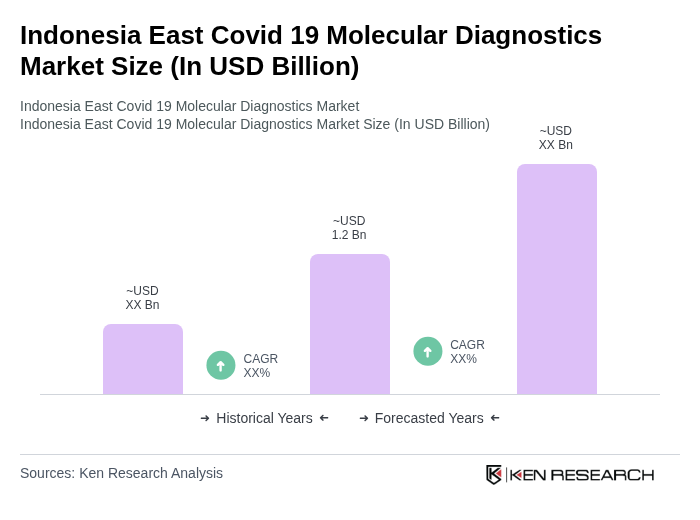
The Indonesia East Covid 19 Molecular Diagnostics Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by the demand for accurate and rapid testing solutions during the ongoing pandemic.