About the Report
Base Year 2024Malaysia Specialty Injectable Generics Market Overview
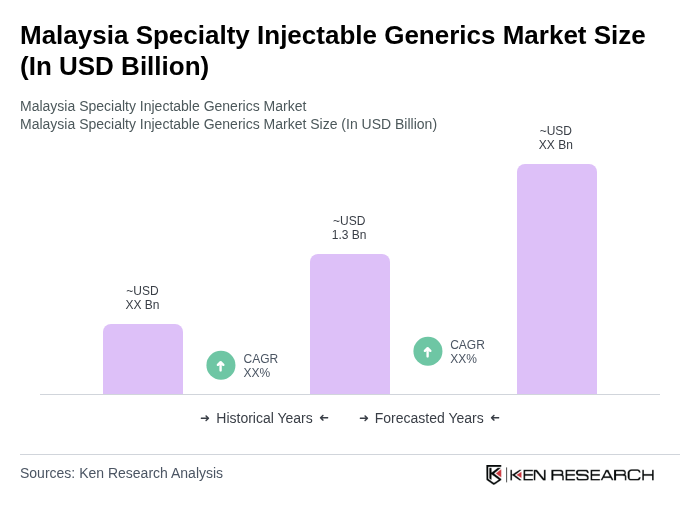
- The Malaysia Specialty Injectable Generics Market is valued at USD 1.3 billion, based on a five-year historical analysis of the overall Malaysian generic drugs market and the significant contribution of parenteral and hospital-focused therapies within that mix. This growth is primarily driven by the increasing prevalence of chronic diseases such as cardiovascular disease, diabetes, cancer, and inflammatory conditions, the rising demand for cost-effective treatment options, and advancements in pharmaceutical manufacturing technologies, including sterile injectables and biosimilar production. The market is also supported by a growing healthcare infrastructure, strong public hospital utilization, and an increasing number of healthcare facilities and specialist centers across the country.
- Key players in this market are concentrated in urban centers such as Kuala Lumpur, Selangor, and Penang. These regions dominate due to their advanced healthcare facilities, higher patient populations, and proximity to pharmaceutical manufacturing hubs and logistics infrastructure. The presence of research institutions and universities in these areas, such as University of Malaya and Universiti Sains Malaysia, supports clinical research, bioequivalence studies, and formulation development, thereby fostering innovation and development in the specialty injectable generics sector.
- Malaysia’s pharmaceutical and generics landscape is guided by the National Medicines Policy (Dasar Ubat Nasional, DUNas) issued by the Ministry of Health Malaysia, with its latest iteration emphasizing quality, safety, efficacy, affordability, and rational use of medicines, including injectables. In addition, the Medication Substitution Policy approved by the National Pharmaceutical Regulatory Agency in 2021 allows pharmacists to substitute branded medicines with generic equivalents under defined conditions, supporting accessibility and affordability of essential medicines and indirectly encouraging uptake of hospital-administered injectable generics. These policy measures, together with NPRA’s Good Manufacturing Practice and product registration requirements for sterile products, promote local production, innovation, and compliance with international quality standards, thereby supporting growth of the specialty injectable generics market.
Malaysia Specialty Injectable Generics Market Segmentation
By Molecule Type:
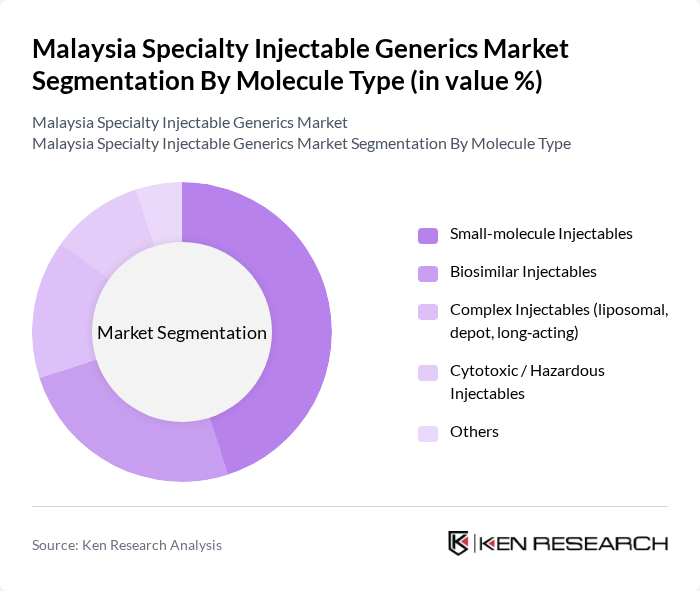
The molecule type segmentation includes various subsegments such as small-molecule injectables, biosimilar injectables, complex injectables (liposomal, depot, long-acting), cytotoxic/hazardous injectables, and others. This structure is aligned with global specialty injectable generics categorizations, where injectables are split between traditional small-molecule generics and biologic/biosimilar-based therapies, as well as complex formulations. Among these, small-molecule injectables dominate the market due to their widespread use in treating infections, cardiovascular conditions, perioperative care, and chronic diseases, supported by established manufacturing processes and lower development costs relative to biologics. Additionally, the increasing demand for biosimilars is notable, driven by the need for cost-effective alternatives to expensive biologics in oncology, autoimmune, and endocrine indications, in line with broader Asia Pacific and global specialty generics trends.
By Therapeutic Area:
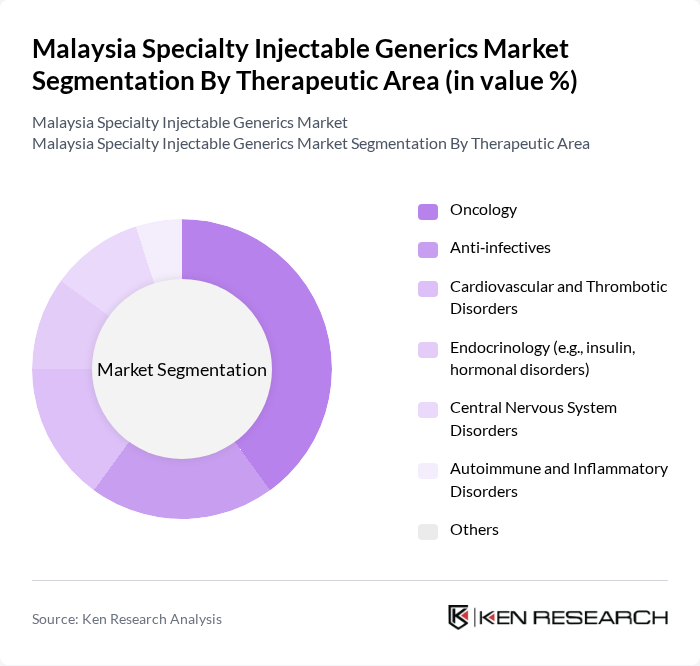
This segmentation includes oncology, anti-infectives, cardiovascular and thrombotic disorders, endocrinology (e.g., insulin, hormonal disorders), central nervous system disorders, autoimmune and inflammatory disorders, and others. This breakdown is consistent with the therapeutic mix of generic and specialty drugs in Malaysia, where cardiovascular diseases, diabetes, neurology, oncology, and anti-inflammatory diseases are key application areas for generics, including parenteral products used predominantly in hospitals. Oncology injectables lead the market in value terms due to the rising incidence of cancer and increasing adoption of high-cost biologics and targeted therapies for which biosimilar and complex generic injectables provide more affordable alternatives. The demand for anti-infectives is also significant, driven by the ongoing burden of infectious diseases and hospital-acquired infections requiring intravenous antibiotics and antifungals. The focus on chronic disease management in Malaysia’s health system further boosts the demand for injectables in cardiovascular, endocrine (notably insulin), and autoimmune therapies, with hospital pharmacies and public sector procurement playing a central role.
Malaysia Specialty Injectable Generics Market Competitive Landscape
The Malaysia Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pharmaniaga Berhad, Duopharma Biotech Berhad, Hovid Berhad, Kotra Pharma (M) Sdn Bhd, Apex Healthcare Berhad, Y.S.P. Southeast Asia Holding Berhad, CCM Duopharma Biotech Berhad, Viatris Inc. (formerly Mylan N.V.), Sandoz AG, Fresenius Kabi AG, Pfizer Inc., Merck & Co., Inc., Teva Pharmaceutical Industries Ltd., B. Braun Melsungen AG, Hikma Pharmaceuticals PLC contribute to innovation, geographic expansion, and service delivery in this space.
Malaysia Specialty Injectable Generics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is significant, with Malaysia reporting an estimated 3.6 million adults living with diabetes in recent national health surveys. This growing patient population drives the demand for injectable generics, as healthcare systems seek affordable treatment options. The World Health Organization estimates that noncommunicable diseases account for about 74% of all deaths in Malaysia, highlighting the urgent need for effective and accessible healthcare solutions.
- Rising Demand for Cost-Effective Treatment Options:With current health expenditure in Malaysia amounting to around MYR 77 billion in the latest available national accounts, the demand for cost-effective treatment options is paramount. Specialty injectable generics offer a more affordable alternative to branded drugs, which can be up to 80% more expensive. This financial pressure on both patients and healthcare providers is driving the adoption of generics, as they help alleviate the burden on national healthcare budgets while ensuring patient access to essential medications.
- Expanding Healthcare Infrastructure:Malaysia's healthcare infrastructure is projected to grow significantly, with government expenditure on health currently above MYR 30 billion and expected to increase further in future in line with policy priorities and demographic change. This expansion includes the establishment of new hospitals and clinics, which will enhance the distribution and availability of specialty injectable generics. Improved healthcare facilities will facilitate better access to these medications, ultimately supporting the market's growth as more patients receive timely and effective treatments.
Market Challenges
- Stringent Regulatory Requirements:The Malaysian pharmaceutical sector faces rigorous regulatory scrutiny, with the National Pharmaceutical Regulatory Agency (NPRA) enforcing strict guidelines for the approval of generics. The average time for regulatory approval can exceed 12 months, delaying market entry for new products. This lengthy process can hinder the ability of manufacturers to respond quickly to market demands, ultimately affecting the availability of specialty injectable generics in the healthcare system.
- Competition from Branded Pharmaceuticals:The presence of established branded pharmaceuticals poses a significant challenge to the specialty injectable generics market. Branded and originator medicines account for a majority share of medicine spending in Malaysia’s pharmaceutical market, although precise published shares vary and are not consistently reported. This dominance creates a competitive landscape where generics must not only match the efficacy of branded products but also overcome brand loyalty among healthcare providers and patients, complicating market penetration efforts.
Malaysia Specialty Injectable Generics Market Future Outlook
The future of the Malaysia specialty injectable generics market appears promising, driven by increasing healthcare investments and a growing focus on chronic disease management. As the government continues to promote generics through various initiatives, including financial incentives for local manufacturers, the market is expected to witness enhanced competition and innovation. Additionally, the integration of digital health solutions will likely streamline processes, improve patient engagement, and facilitate better access to injectable generics, ultimately benefiting the healthcare landscape.
Market Opportunities
- Growth in the Aging Population:Malaysia's aging population is projected to reach about 15% of the total population in future as the country transitions towards aged-nation status, according to United Nations and national projections. This demographic shift will necessitate increased healthcare services and medications tailored to age-related conditions, presenting a lucrative opportunity for generics manufacturers to expand their product offerings and cater to this growing segment.
- Expansion of Telemedicine Services:The rise of telemedicine in Malaysia, supported by Ministry of Health virtual health initiatives and increasing digital health platform usage, presents a unique opportunity for specialty injectable generics, although specific national consultation volumes are not consistently published. As healthcare providers increasingly adopt telehealth solutions, the demand for injectable medications that can be prescribed remotely will grow, allowing generics to reach a broader patient base and improve treatment adherence.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Small-molecule Injectables Biosimilar Injectables Complex Injectables (liposomal, depot, long?acting) Cytotoxic / Hazardous Injectables Others |
| By Therapeutic Area | Oncology Anti?infectives Cardiovascular and Thrombotic Disorders Endocrinology (e.g., insulin, hormonal disorders) Central Nervous System Disorders Autoimmune and Inflammatory Disorders Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Intrathecal / Epidural Others |
| By End-User | Public Hospitals Private Hospitals Specialist Clinics and Day-care Centres Retail & Chain Pharmacies Homecare / Ambulatory Care Settings Others |
| By Formulation & Delivery | Ready-to-use (RTU) Injectables Lyophilized Injectables Pre-filled Syringes and Pens Large-volume Parenterals (LVP) Others |
| By Packaging Type | Vials Ampoules Pre-filled Syringes Cartridges Others |
| By Region | Central Region (Kuala Lumpur, Selangor, Putrajaya) Northern Region Southern Region East Coast Region East Malaysia (Sabah & Sarawak) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health Malaysia, National Pharmaceutical Regulatory Agency)
Manufacturers and Producers
Distributors and Wholesalers
Pharmaceutical Retail Chains
Healthcare Providers and Hospitals
Industry Associations (e.g., Malaysian Pharmaceutical Society)
Financial Institutions and Banks
Players Mentioned in the Report:
Pharmaniaga Berhad
Duopharma Biotech Berhad
Hovid Berhad
Kotra Pharma (M) Sdn Bhd
Apex Healthcare Berhad
Y.S.P. Southeast Asia Holding Berhad
CCM Duopharma Biotech Berhad
Viatris Inc. (formerly Mylan N.V.)
Sandoz AG
Fresenius Kabi AG
Pfizer Inc.
Merck & Co., Inc.
Teva Pharmaceutical Industries Ltd.
B. Braun Melsungen AG
Hikma Pharmaceuticals PLC
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Malaysia Specialty Injectable Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Malaysia Specialty Injectable Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Malaysia Specialty Injectable Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for cost-effective treatment options
3.1.3 Expanding healthcare infrastructure
3.1.4 Government initiatives to promote generics
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 Competition from branded pharmaceuticals
3.2.3 Limited awareness among healthcare providers
3.2.4 Supply chain complexities
3.3 Market Opportunities
3.3.1 Growth in the aging population
3.3.2 Expansion of telemedicine services
3.3.3 Increasing investment in R&D
3.3.4 Collaborations with healthcare providers
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Adoption of advanced manufacturing technologies
3.4.3 Growing focus on sustainability in production
3.4.4 Integration of digital health solutions
3.5 Government Regulation
3.5.1 Implementation of price controls
3.5.2 Regulatory approval processes for generics
3.5.3 Quality assurance standards
3.5.4 Incentives for local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Malaysia Specialty Injectable Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Malaysia Specialty Injectable Generics Market Segmentation
8.1 By Molecule Type
8.1.1 Small-molecule Injectables
8.1.2 Biosimilar Injectables
8.1.3 Complex Injectables (liposomal, depot, long?acting)
8.1.4 Cytotoxic / Hazardous Injectables
8.1.5 Others
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Anti?infectives
8.2.3 Cardiovascular and Thrombotic Disorders
8.2.4 Endocrinology (e.g., insulin, hormonal disorders)
8.2.5 Central Nervous System Disorders
8.2.6 Autoimmune and Inflammatory Disorders
8.2.7 Others
8.3 By Route of Administration
8.3.1 Intravenous (IV)
8.3.2 Intramuscular (IM)
8.3.3 Subcutaneous (SC)
8.3.4 Intrathecal / Epidural
8.3.5 Others
8.4 By End-User
8.4.1 Public Hospitals
8.4.2 Private Hospitals
8.4.3 Specialist Clinics and Day-care Centres
8.4.4 Retail & Chain Pharmacies
8.4.5 Homecare / Ambulatory Care Settings
8.4.6 Others
8.5 By Formulation & Delivery
8.5.1 Ready-to-use (RTU) Injectables
8.5.2 Lyophilized Injectables
8.5.3 Pre-filled Syringes and Pens
8.5.4 Large-volume Parenterals (LVP)
8.5.5 Others
8.6 By Packaging Type
8.6.1 Vials
8.6.2 Ampoules
8.6.3 Pre-filled Syringes
8.6.4 Cartridges
8.6.5 Others
8.7 By Region
8.7.1 Central Region (Kuala Lumpur, Selangor, Putrajaya)
8.7.2 Northern Region
8.7.3 Southern Region
8.7.4 East Coast Region
8.7.5 East Malaysia (Sabah & Sarawak)
9. Malaysia Specialty Injectable Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Type (Local, Regional, Multinational)
9.2.3 Malaysia Specialty Injectable Generics Revenue (Latest Year, USD Mn)
9.2.4 3-year CAGR in Malaysia Specialty Injectables Revenue
9.2.5 Market Share in Malaysia Specialty Injectable Generics (%)
9.2.6 Portfolio Breadth Index (No. of SKUs / ATC classes)
9.2.7 Share of Oncology & High-value Therapies in Portfolio (%)
9.2.8 Average Realized Price vs Market Benchmark (%)
9.2.9 Gross Margin from Specialty Injectables (%)
9.2.10 Capex Intensity (Capex / Sales %) in Injectables
9.2.11 Regulatory & GMP Compliance Track Record (Inspections, observations)
9.2.12 Manufacturing Capacity Utilization (%) – Injectables
9.2.13 R&D Intensity (% of Sales) in Complex / Biosimilar Injectables
9.2.14 New Product Launches in Last 3 Years (No.)
9.2.15 Export Revenue Share of Malaysia-made Injectables (%)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pharmaniaga Berhad
9.5.2 Duopharma Biotech Berhad
9.5.3 Hovid Berhad
9.5.4 Kotra Pharma (M) Sdn Bhd
9.5.5 Apex Healthcare Berhad
9.5.6 Y.S.P. Southeast Asia Holding Berhad
9.5.7 CCM Duopharma Biotech Berhad
9.5.8 Viatris Inc. (formerly Mylan N.V.)
9.5.9 Sandoz AG
9.5.10 Fresenius Kabi AG
9.5.11 Pfizer Inc.
9.5.12 Merck & Co., Inc.
9.5.13 Teva Pharmaceutical Industries Ltd.
9.5.14 B. Braun Melsungen AG
9.5.15 Hikma Pharmaceuticals PLC
10. Malaysia Specialty Injectable Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 Technology Upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Healthcare Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvements
10.5.3 Scalability of Solutions
10.5.4 Others
11. Malaysia Specialty Injectable Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from Malaysian pharmaceutical associations and health ministries
- Review of published articles in medical journals focusing on injectable generics
- Examination of regulatory frameworks and guidelines from the National Pharmaceutical Regulatory Agency (NPRA)
Primary Research
- Interviews with key opinion leaders in the pharmaceutical industry, including pharmacists and healthcare professionals
- Surveys conducted with hospital procurement managers to understand purchasing behaviors
- Field interviews with representatives from specialty injectable generic manufacturers
Validation & Triangulation
- Cross-validation of data through multiple sources, including industry reports and expert interviews
- Triangulation of market size estimates using sales data and regulatory insights
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and pharmaceutical spending
- Segmentation of the market by therapeutic areas and types of injectable generics
- Incorporation of government healthcare initiatives promoting the use of generics
Bottom-up Modeling
- Volume estimates derived from sales data of leading injectable generic products
- Cost analysis based on pricing strategies of manufacturers and distributors
- Calculation of market size using volume x price methodology for various segments
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating demographic trends and healthcare access
- Scenario analysis based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 100 | Procurement Managers, Supply Chain Coordinators |
| Pharmacy Chains | 80 | Pharmacy Managers, Inventory Specialists |
| Specialty Clinics | 70 | Clinic Administrators, Medical Directors |
| Healthcare Distributors | 90 | Sales Representatives, Distribution Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Malaysia Specialty Injectable Generics Market?
The Malaysia Specialty Injectable Generics Market is valued at approximately USD 1.3 billion, reflecting significant growth driven by the increasing prevalence of chronic diseases and advancements in pharmaceutical manufacturing technologies.