About the Report
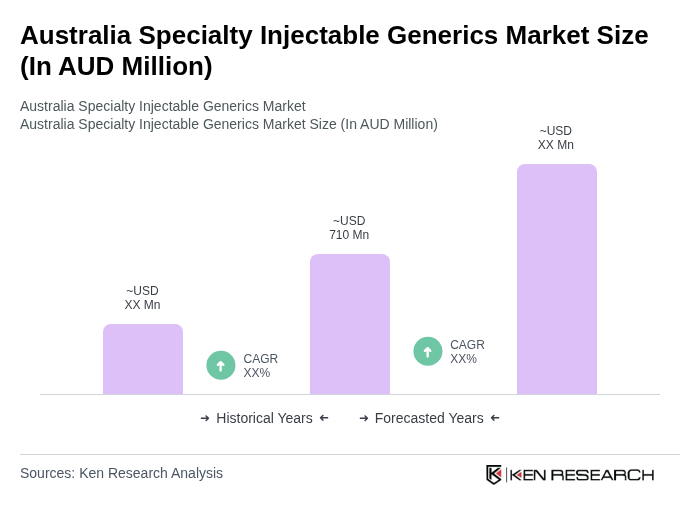
Base Year 2024Australia Specialty Injectable Generics Market Overview
- The Australia Specialty Injectable Generics Market is valued at approximately AUD 710 million, based on a five-year historical analysis of the national generic injectables segment and its specialty share. This growth is primarily driven by the increasing prevalence of chronic diseases, the rising demand for cost-effective treatment options, and the expansion of healthcare infrastructure across both public and private sectors. The market is also supported by the growing acceptance of generics among healthcare providers and patients, with generic medicines accounting for a substantial majority of prescriptions under the Pharmaceutical Benefits Scheme (PBS), which enhances accessibility to essential medications.
- Key players in this market are concentrated in major cities such as Sydney, Melbourne, and Brisbane. These cities dominate due to their advanced tertiary and quaternary healthcare facilities, established research institutions, and a high concentration of multinational and domestic pharmaceutical companies. The presence of a robust regulatory framework administered by the Therapeutic Goods Administration (TGA), PBS pricing and reimbursement mechanisms, and government support for innovation and clinical research further strengthens their position in the specialty injectable generics market.
- In 2023, the Australian government endorsed an updated National Medicines Policy, which aims to ensure that all Australians have access to affordable and effective medicines, underpinned by a sustainable pharmaceutical system. This policy emphasizes the importance of generics and biosimilars in the healthcare system, promoting their use to enhance patient access and reduce healthcare costs through PBS listing and prescribing guidance. The initiative aligns with the Therapeutic Goods Act 1989 and the National Health Act 1953, including PBS pricing and statutory price-reduction provisions administered by the Department of Health and Aged Care, which streamline market entry and support timely approval and uptake of new generics and biosimilars, thereby fostering competition in the market.
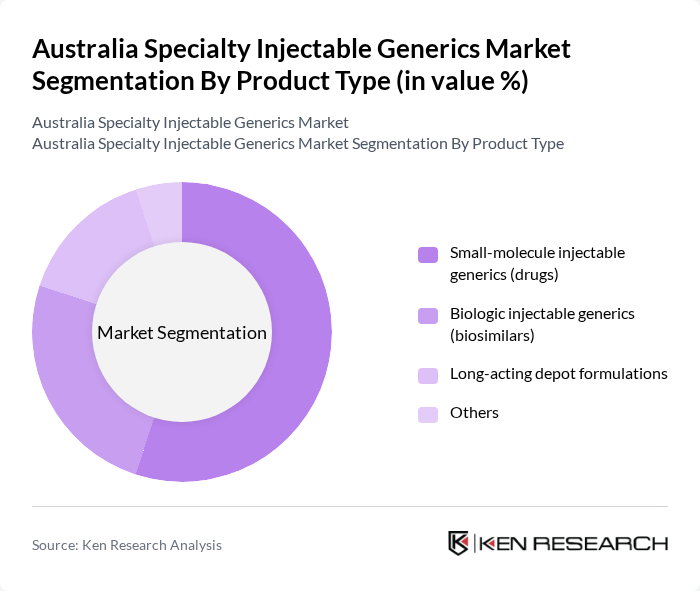
Australia Specialty Injectable Generics Market Segmentation
By Product Type:The product type segmentation includes various categories such as small-molecule injectable generics, biologic injectable generics, long-acting depot formulations, and others. Among these, small-molecule injectable generics are leading the market due to their widespread use in hospital and outpatient settings for treating conditions including infections, cardiovascular diseases, oncology indications, and perioperative care. The affordability and availability of these generics under PBS listing and hospital tenders have made them the preferred choice for healthcare providers and patients alike. Biologic injectable generics, or biosimilars, are also gaining traction in Australia, supported by TGA biosimilar approval pathways and PBS reimbursement, as they offer similar efficacy and safety profiles to their reference products and are increasingly used in oncology, immunology, and rheumatology, further driving market growth.
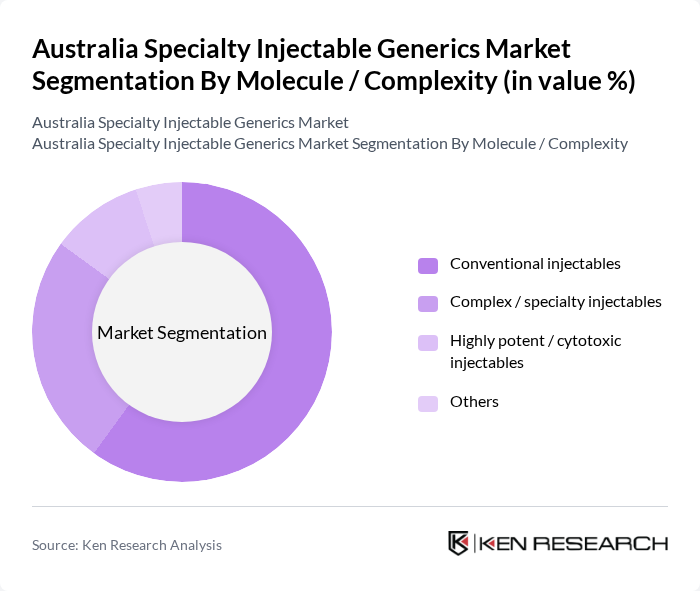
By Molecule / Complexity:This segmentation includes conventional injectables, complex/specialty injectables, highly potent/cytotoxic injectables, and others. Conventional injectables dominate the market due to their established use across high-volume therapeutic areas such as anesthesia, anti-infectives, cardiovascular care, and parenteral nutrition, where numerous molecules have long-standing generic competition. The relative simplicity of their formulation, established clinical guidelines, and familiarity among prescribers and hospital pharmacists contribute to their widespread acceptance. Complex and specialty injectables, including liposomal formulations, depot injections, and certain biologic-derived products, are also gaining ground, driven by advancements in biotechnology, the growing burden of oncology and autoimmune diseases, and a rising number of specialty brands coming off patent that are being targeted for generic and biosimilar development.
Australia Specialty Injectable Generics Market Competitive Landscape
The Australia Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Australia Pty Ltd, Amgen Australia Pty Ltd, Sandoz (a Novartis division) – Australia, Viatris (including legacy Mylan) Australia, Fresenius Kabi Australia Pty Ltd, Teva Pharma Australia Pty Ltd, Baxter Healthcare Pty Ltd (Australia), Merck Sharp & Dohme (MSD) Australia, Biocon Biologics / Biocon Limited – Australia operations, Hikma Pharmaceuticals PLC – Australia presence, Bausch Health Companies Inc. – Australia, Eli Lilly Australia Pty Ltd, Novartis Pharmaceuticals Australia Pty Ltd, Sanofi-Aventis Australia Pty Ltd, GlaxoSmithKline (GSK) Australia contribute to innovation, geographic expansion, and service delivery in this space, leveraging their global injectable and biosimilar portfolios to participate in PBS listings and hospital tenders in Australia.
Australia Specialty Injectable Generics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the specialty injectable generics market. In Australia, approximately 1.3 million people are diagnosed with diabetes, and cardiovascular diseases account for about 25% of all deaths. This growing patient population necessitates effective and affordable treatment options, propelling the demand for specialty injectables, which are often more cost-effective than branded alternatives.
- Rising Demand for Cost-Effective Treatment Options:The Australian healthcare system is increasingly focused on cost containment, leading to a surge in demand for cost-effective treatment options. In future, the Australian government is projected to spend AUD 90 billion on pharmaceuticals, with a significant portion allocated to generics. This trend is driven by the need to manage healthcare budgets while ensuring patient access to essential medications, thereby boosting the specialty injectable generics market.
- Advancements in Drug Formulation Technologies:Innovations in drug formulation technologies are enhancing the efficacy and safety of specialty injectables. In future, the Australian pharmaceutical sector is expected to invest AUD 1.5 billion in research and development, focusing on advanced delivery systems and biopharmaceuticals. These advancements not only improve patient outcomes but also expand the range of conditions that can be treated with specialty injectables, driving market growth.
Market Challenges
- Stringent Regulatory Requirements:The specialty injectable generics market faces significant challenges due to stringent regulatory requirements imposed by the Therapeutic Goods Administration (TGA). In future, compliance costs are expected to rise by 10%, impacting smaller manufacturers disproportionately. These regulations, while ensuring safety and efficacy, can delay product launches and increase barriers to entry for new players in the market.
- Competition from Branded Pharmaceuticals:The presence of established branded pharmaceuticals poses a considerable challenge to the specialty injectable generics market. In future, branded drugs are projected to account for 70% of the injectable market share in Australia. This competition often leads to price wars and aggressive marketing strategies, making it difficult for generics to gain market traction despite their cost advantages.
Australia Specialty Injectable Generics Market Future Outlook
The future of the specialty injectable generics market in Australia appears promising, driven by technological advancements and a growing emphasis on personalized medicine. As healthcare providers increasingly adopt digital health solutions, the integration of telehealth and remote monitoring will facilitate better patient management. Furthermore, the shift towards value-based healthcare will encourage the development of innovative generics that meet specific patient needs, enhancing treatment outcomes and market penetration.
Market Opportunities
- Expansion into Emerging Therapeutic Areas:There is a significant opportunity for specialty injectable generics to expand into emerging therapeutic areas such as oncology and autoimmune diseases. In future, with the Australian government investing AUD 2 billion in cancer research, generics that target these high-demand areas can capture substantial market share and improve patient access to essential treatments.
- Development of Biosimilars:The increasing focus on biosimilars presents a lucrative opportunity for the specialty injectable generics market. In future, the Australian biosimilars market is expected to reach AUD 1 billion, driven by the expiration of patents for several biologics. This trend allows generics manufacturers to introduce cost-effective alternatives, enhancing competition and patient access to biologic therapies.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Small-molecule injectable generics (drugs) Biologic injectable generics (biosimilars) Long-acting depot formulations Others |
| By Molecule / Complexity | Conventional injectables Complex / specialty injectables Highly potent / cytotoxic injectables Others |
| By Therapeutic Area | Oncology Cardiovascular and metabolic diseases Infectious diseases Autoimmune and inflammatory disorders Central nervous system disorders Others |
| By End-User | Public hospitals Private hospitals Specialist clinics and day infusion centres Home- and community-based healthcare providers Others |
| By Procurement & Payer Channel | PBS-listed hospital tenders Private hospital group purchasing Retail and specialty pharmacies Direct-to-provider supply Others |
| By Route of Administration | Intravenous (IV) Subcutaneous (SC) Intramuscular (IM) Others |
| By Australian Region | New South Wales (NSW) Victoria (VIC) Queensland (QLD) Western Australia (WA) South Australia (SA) Tasmania (TAS), Australian Capital Territory (ACT) & Northern Territory (NT) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Department of Health)
Manufacturers and Producers
Distributors and Wholesalers
Pharmaceutical Supply Chain Managers
Healthcare Providers and Hospitals
Pharmacy Benefit Managers
Insurance Companies and Payers
Players Mentioned in the Report:
Pfizer Australia Pty Ltd
Amgen Australia Pty Ltd
Sandoz (a Novartis division) Australia
Viatris (including legacy Mylan) Australia
Fresenius Kabi Australia Pty Ltd
Teva Pharma Australia Pty Ltd
Baxter Healthcare Pty Ltd (Australia)
Merck Sharp & Dohme (MSD) Australia
Biocon Biologics / Biocon Limited Australia operations
Hikma Pharmaceuticals PLC Australia presence
Bausch Health Companies Inc. Australia
Eli Lilly Australia Pty Ltd
Novartis Pharmaceuticals Australia Pty Ltd
Sanofi-Aventis Australia Pty Ltd
GlaxoSmithKline (GSK) Australia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Specialty Injectable Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Specialty Injectable Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Specialty Injectable Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for cost-effective treatment options
3.1.3 Advancements in drug formulation technologies
3.1.4 Supportive government policies for generics
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 Competition from branded pharmaceuticals
3.2.3 Supply chain complexities
3.2.4 Limited awareness among healthcare providers
3.3 Market Opportunities
3.3.1 Expansion into emerging therapeutic areas
3.3.2 Collaborations with healthcare providers
3.3.3 Development of biosimilars
3.3.4 Increasing focus on personalized medicine
3.4 Market Trends
3.4.1 Growth of telehealth and remote patient monitoring
3.4.2 Shift towards value-based healthcare
3.4.3 Rising adoption of digital health solutions
3.4.4 Increased focus on sustainability in manufacturing
3.5 Government Regulation
3.5.1 Therapeutic Goods Administration (TGA) guidelines
3.5.2 Pharmaceutical Benefits Scheme (PBS) policies
3.5.3 Pricing and reimbursement regulations
3.5.4 Quality assurance standards for injectables
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Specialty Injectable Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Specialty Injectable Generics Market Segmentation
8.1 By Product Type
8.1.1 Small-molecule injectable generics (drugs)
8.1.2 Biologic injectable generics (biosimilars)
8.1.3 Long-acting depot formulations
8.1.4 Others
8.2 By Molecule / Complexity
8.2.1 Conventional injectables
8.2.2 Complex / specialty injectables
8.2.3 Highly potent / cytotoxic injectables
8.2.4 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Cardiovascular and metabolic diseases
8.3.3 Infectious diseases
8.3.4 Autoimmune and inflammatory disorders
8.3.5 Central nervous system disorders
8.3.6 Others
8.4 By End-User
8.4.1 Public hospitals
8.4.2 Private hospitals
8.4.3 Specialist clinics and day infusion centres
8.4.4 Home- and community-based healthcare providers
8.4.5 Others
8.5 By Procurement & Payer Channel
8.5.1 PBS-listed hospital tenders
8.5.2 Private hospital group purchasing
8.5.3 Retail and specialty pharmacies
8.5.4 Direct-to-provider supply
8.5.5 Others
8.6 By Route of Administration
8.6.1 Intravenous (IV)
8.6.2 Subcutaneous (SC)
8.6.3 Intramuscular (IM)
8.6.4 Others
8.7 By Australian Region
8.7.1 New South Wales (NSW)
8.7.2 Victoria (VIC)
8.7.3 Queensland (QLD)
8.7.4 Western Australia (WA)
8.7.5 South Australia (SA)
8.7.6 Tasmania (TAS), Australian Capital Territory (ACT) & Northern Territory (NT)
9. Australia Specialty Injectable Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Group size (large, medium, or small as per industry convention)
9.2.3 Australia specialty injectable generics revenue (AUD, latest year)
9.2.4 3-year revenue CAGR in specialty injectables (Australia)
9.2.5 PBS-listed injectable generics portfolio breadth (number of SKUs)
9.2.6 Hospital tender win rate (% of tenders participated)
9.2.7 Average price discount vs reference brands (%)
9.2.8 Market share in key therapeutic areas (oncology, autoimmune, etc.)
9.2.9 Manufacturing footprint in Australia / ANZ (capacity, sites)
9.2.10 Pharmacovigilance and quality compliance metrics (recall rate, deviations)
9.2.11 R&D and pipeline intensity (number of pending TGA filings / biosimilars)
9.2.12 Distribution coverage (hospital, pharmacy, specialty channels)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pfizer Australia Pty Ltd
9.5.2 Amgen Australia Pty Ltd
9.5.3 Sandoz (a Novartis division) – Australia
9.5.4 Viatris (including legacy Mylan) Australia
9.5.5 Fresenius Kabi Australia Pty Ltd
9.5.6 Teva Pharma Australia Pty Ltd
9.5.7 Baxter Healthcare Pty Ltd (Australia)
9.5.8 Merck Sharp & Dohme (MSD) Australia
9.5.9 Biocon Biologics / Biocon Limited – Australia operations
9.5.10 Hikma Pharmaceuticals PLC – Australia presence
9.5.11 Bausch Health Companies Inc. – Australia
9.5.12 Eli Lilly Australia Pty Ltd
9.5.13 Novartis Pharmaceuticals Australia Pty Ltd
9.5.14 Sanofi-Aventis Australia Pty Ltd
9.5.15 GlaxoSmithKline (GSK) Australia
10. Australia Specialty Injectable Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Department of Veterans' Affairs
10.1.3 State Health Departments
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 Technology Upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Healthcare Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcomes Improvement
10.5.3 Scalability of Solutions
10.5.4 Others
11. Australia Specialty Injectable Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Australian pharmaceutical associations and health authorities
- Review of market trends and forecasts from government publications and healthcare journals
- Examination of patent filings and regulatory submissions related to specialty injectable generics
Primary Research
- Interviews with key opinion leaders in the pharmaceutical industry, including pharmacists and healthcare providers
- Surveys targeting procurement managers at hospitals and clinics utilizing specialty injectables
- Focus groups with patients and caregivers to understand usage patterns and preferences
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and market reports
- Triangulation of insights from primary interviews with secondary research findings
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and pharmaceutical spending
- Segmentation of the market by therapeutic areas and types of specialty injectables
- Incorporation of demographic trends and disease prevalence data in Australia
Bottom-up Modeling
- Collection of sales data from leading specialty injectable manufacturers operating in Australia
- Estimation of market share based on volume and revenue from key product categories
- Analysis of pricing strategies and reimbursement rates for specialty injectables
Forecasting & Scenario Analysis
- Development of predictive models using historical growth rates and market drivers
- Scenario analysis based on potential regulatory changes and market entry of new products
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Specialty Injectables | 120 | Oncologists, Pharmacy Directors |
| Cardiovascular Specialty Injectables | 100 | Cardiologists, Clinical Pharmacists |
| Neurology Specialty Injectables | 80 | Neurologists, Hospital Administrators |
| Endocrinology Specialty Injectables | 60 | Endocrinologists, Healthcare Policy Makers |
| Patient Experience with Specialty Injectables | 90 | Patients, Caregivers, Patient Advocacy Groups |
Frequently Asked Questions
What is the current value of the Australia Specialty Injectable Generics Market?
The Australia Specialty Injectable Generics Market is valued at approximately AUD 710 million, driven by the increasing prevalence of chronic diseases and the demand for cost-effective treatment options, alongside the expansion of healthcare infrastructure.