About the Report
Base Year 2024Oman Specialty Injectable Generics Market Overview
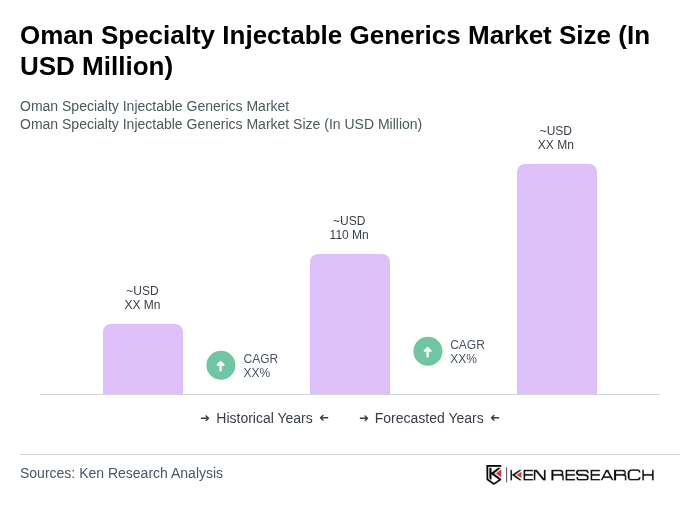
- The Oman Specialty Injectable Generics Market is valued at USD 110 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases such as diabetes, cardiovascular disease, and cancer in Oman, alongside rising demand for cost-effective treatment options and the ongoing expansion of healthcare infrastructure. The market is also supported by strong government focus on medicine affordability, the growing acceptance of generics among healthcare providers and patients, and wider availability of injectable generics in hospital and retail channels, all contributing to overall market expansion.
- Muscat and Dhofar are the dominant regions in the Oman Specialty Injectable Generics Market due to their advanced healthcare facilities and relatively higher population density compared to other governorates. Muscat, being the capital and the principal urban and economic center, has a concentration of tertiary hospitals, specialty clinics, and private healthcare providers that extensively utilize specialty injectables, while Dhofar (with Salalah as the main city) benefits from sustained government initiatives to enhance healthcare access and infrastructure in the southern region. These factors create a conducive environment for market growth in these regions, with Muscat and Dhofar typically cited as key regional clusters in Oman’s broader specialty generics landscape.
- In 2023, the Omani government implemented more stringent quality and regulatory oversight for generic medicines, including injectables, under the framework of Ministerial Decision No. 41/2023 issuing the Executive Regulations of the Pharmaceutical Products Law, by the Ministry of Health. These regulations require that all pharmaceutical products, including specialty injectable generics, undergo rigorous quality, safety, and efficacy assessment and obtain marketing authorization from the Directorate General of Pharmaceutical Affairs and Drug Control before being placed on the market, covering aspects such as bioequivalence, Good Manufacturing Practice (GMP) compliance, and pharmacovigilance obligations. This framework aims to enhance patient safety and ensure that only high-quality products are available, thereby reinforcing trust among healthcare providers and patients in the use of generic injectable therapies.
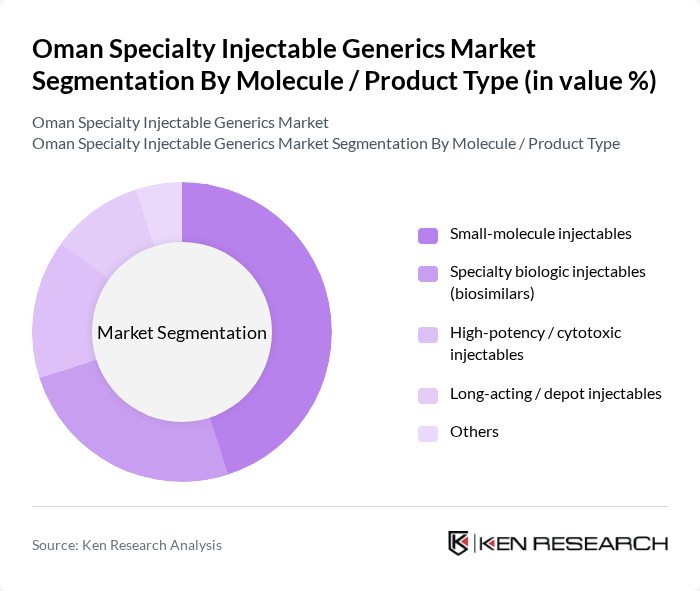
Oman Specialty Injectable Generics Market Segmentation
By Molecule / Product Type:The market is segmented into various product types, including small-molecule injectables, specialty biologic injectables (biosimilars), high-potency/cytotoxic injectables, long-acting/depot injectables, and others. This structure is consistent with global specialty injectable generics categorization, where injectable generics and biosimilars are treated as distinct but closely related segments. Among these, small-molecule injectables are expected to account for the largest share in Oman, reflecting their widespread use in anti-infectives, anesthesia, cardiovascular care, and supportive oncology therapies, in line with the broader specialty generics pattern in which injectables form the leading type segment. The affordability, established efficacy, and familiarity of these injectables in hospital formularies make them a preferred choice for healthcare providers, while the biosimilar and high-potency cytotoxic segments are gradually expanding as oncology and autoimmune treatment needs grow.
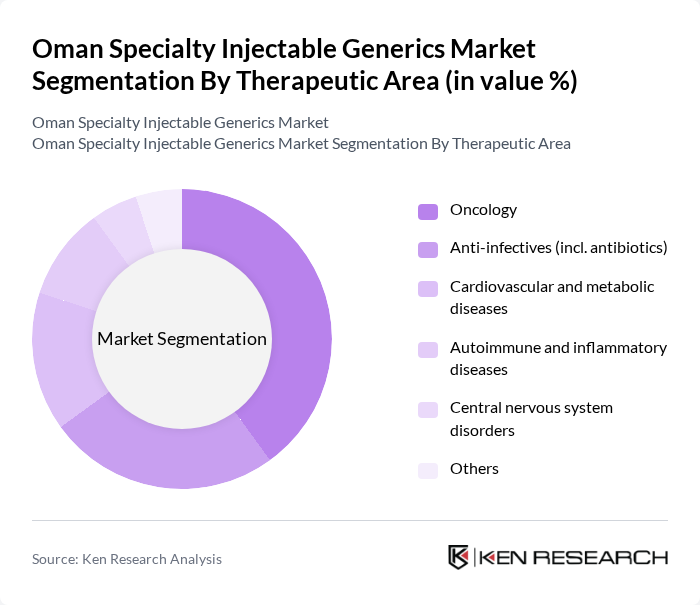
By Therapeutic Area:The therapeutic areas covered include oncology, anti-infectives (including antibiotics), cardiovascular and metabolic diseases, autoimmune and inflammatory diseases, central nervous system disorders, and others. This aligns with the main application areas of specialty injectable generics globally, where oncology, infectious diseases, and inflammatory conditions represent the core demand clusters. Oncology injectables are particularly prominent due to the rising incidence of cancer in Oman and the wider Gulf region, which has been documented as a key driver for both specialty generics and generic injectables demand. The focus on more advanced oncology regimens, greater use of supportive care injectables, and progressive introduction of biosimilar oncology products is driving significant investments and formulary inclusion in this segment, while anti-infectives and cardiovascular/metabolic therapies remain essential high-volume categories.
Oman Specialty Injectable Generics Market Competitive Landscape
The Oman Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oman Pharmaceutical Products Co. LLC, Gulf Pharmaceutical Industries (Julphar), National Pharmaceutical Industries Co. SAOG, Dhofar Pharmaceutical Industries Co. SAOC, Muscat Pharmacy & Stores LLC, Aster DM Healthcare, Badr Al Samaa Group of Hospitals, Apex Medical Group, Pharmazone LLC, Life Pharmacy Oman, Al Hayat International Hospital, NMC Healthcare (Oman operations), Al Raffah Hospital, Starcare Hospital, Others (regional injectable generic manufacturers and distributors) contribute to innovation, geographic expansion, and service delivery in this space.
Oman Specialty Injectable Generics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the Oman specialty injectable generics market. In future, it is estimated that over 500,000 Omanis will be living with diabetes, reflecting an increase from previous periods. This growing patient population necessitates effective and affordable treatment options, thereby boosting the demand for specialty injectables, which are crucial for managing these conditions effectively.
- Rising Demand for Cost-Effective Treatment Options:With healthcare costs escalating, there is a marked shift towards cost-effective treatment alternatives. In future, the healthcare expenditure in Oman is projected to reach approximately USD 4 billion, with a significant portion allocated to pharmaceuticals. This trend is driving healthcare providers and patients to seek affordable injectable generics, which offer similar efficacy to branded drugs at a fraction of the cost, thus expanding market opportunities.
- Advancements in Pharmaceutical Manufacturing Technologies:Technological innovations in pharmaceutical manufacturing are enhancing the production of specialty injectables. In future, investments in advanced manufacturing technologies are expected to exceed $500 million in Oman. These advancements not only improve production efficiency but also ensure higher quality and compliance with international standards, thereby fostering growth in the specialty injectable generics market as manufacturers can meet increasing demand effectively.
Market Challenges
- Stringent Regulatory Requirements:The regulatory landscape for pharmaceuticals in Oman is characterized by stringent requirements that can hinder market entry for new products. In future, the Ministry of Health is expected to maintain rigorous standards for drug approvals, which may delay the introduction of new specialty injectables. This regulatory burden can limit the ability of manufacturers to respond swiftly to market needs, posing a significant challenge to growth.
- High Competition from Branded Pharmaceuticals:The presence of established branded pharmaceuticals poses a formidable challenge to the specialty injectable generics market. In future, branded drugs are projected to account for over 70% of the total pharmaceutical market in Oman. This high market share creates intense competition, making it difficult for generic manufacturers to gain market traction and necessitating strategic marketing and differentiation efforts to attract healthcare providers and patients.
Oman Specialty Injectable Generics Market Future Outlook
The future of the Oman specialty injectable generics market appears promising, driven by increasing healthcare investments and a growing focus on chronic disease management. As the healthcare infrastructure expands, particularly in rural areas, access to specialty injectables will improve. Additionally, the integration of digital health technologies is expected to enhance patient engagement and adherence to treatment, further propelling market growth. The emphasis on sustainability in manufacturing practices will also shape the industry's evolution, aligning with global trends towards environmentally responsible healthcare solutions.
Market Opportunities
- Expansion of Healthcare Infrastructure:The ongoing expansion of healthcare facilities in Oman presents a significant opportunity for specialty injectable generics. With the government investing over $1 billion in healthcare infrastructure in future, increased access to hospitals and clinics will facilitate the distribution and use of injectable generics, enhancing market penetration and patient access to essential medications.
- Increasing Investment in R&D for Injectables:The growing focus on research and development in the pharmaceutical sector is creating opportunities for innovation in specialty injectables. In future, R&D spending in Oman is projected to reach $200 million, fostering the development of new formulations and delivery methods. This investment will enable companies to introduce novel products that meet specific patient needs, thereby expanding their market presence.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Molecule / Product Type | Small-molecule injectables Specialty biologic injectables (biosimilars) High-potency / cytotoxic injectables Long-acting / depot injectables Others |
| By Therapeutic Area | Oncology Anti-infectives (incl. antibiotics) Cardiovascular and metabolic diseases Autoimmune and inflammatory diseases Central nervous system disorders Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Others |
| By Container / Packaging Type | Vials Ampoules Pre-filled syringes Premix / ready-to-use bags Others |
| By End-User / Point of Care | Public hospitals Private hospitals Specialty clinics and day-care centers Retail and specialty pharmacies Others |
| By Distribution Channel | Tender-based institutional procurement Direct sales to hospitals and clinics Distributors / wholesalers Retail & online pharmacies Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman Pharmaceutical Association)
Manufacturers and Producers
Distributors and Wholesalers
Pharmaceutical Importers
Healthcare Providers and Hospitals
Industry Associations and Trade Organizations
Financial Institutions and Banks
Players Mentioned in the Report:
Oman Pharmaceutical Products Co. LLC
Gulf Pharmaceutical Industries (Julphar)
National Pharmaceutical Industries Co. SAOG
Dhofar Pharmaceutical Industries Co. SAOC
Muscat Pharmacy & Stores LLC
Aster DM Healthcare
Badr Al Samaa Group of Hospitals
Apex Medical Group
Pharmazone LLC
Life Pharmacy Oman
Al Hayat International Hospital
NMC Healthcare (Oman operations)
Al Raffah Hospital
Starcare Hospital
Others (regional injectable generic manufacturers and distributors)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Specialty Injectable Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Specialty Injectable Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Specialty Injectable Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for cost-effective treatment options
3.1.3 Advancements in pharmaceutical manufacturing technologies
3.1.4 Supportive government policies for generic drugs
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition from branded pharmaceuticals
3.2.3 Limited awareness among healthcare providers
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Growing geriatric population
3.3.3 Increasing investment in R&D for injectables
3.3.4 Potential for export to neighboring markets
3.4 Market Trends
3.4.1 Shift towards biosimilars and biopharmaceuticals
3.4.2 Rise in personalized medicine
3.4.3 Adoption of digital health technologies
3.4.4 Focus on sustainability in manufacturing
3.5 Government Regulation
3.5.1 Regulatory frameworks for drug approval
3.5.2 Pricing regulations for generics
3.5.3 Quality assurance standards
3.5.4 Intellectual property rights enforcement
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Specialty Injectable Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Specialty Injectable Generics Market Segmentation
8.1 By Molecule / Product Type
8.1.1 Small-molecule injectables
8.1.2 Specialty biologic injectables (biosimilars)
8.1.3 High-potency / cytotoxic injectables
8.1.4 Long-acting / depot injectables
8.1.5 Others
8.2 By Therapeutic Area
8.2.1 Oncology
8.2.2 Anti-infectives (incl. antibiotics)
8.2.3 Cardiovascular and metabolic diseases
8.2.4 Autoimmune and inflammatory diseases
8.2.5 Central nervous system disorders
8.2.6 Others
8.3 By Route of Administration
8.3.1 Intravenous (IV)
8.3.2 Intramuscular (IM)
8.3.3 Subcutaneous (SC)
8.3.4 Others
8.4 By Container / Packaging Type
8.4.1 Vials
8.4.2 Ampoules
8.4.3 Pre-filled syringes
8.4.4 Premix / ready-to-use bags
8.4.5 Others
8.5 By End-User / Point of Care
8.5.1 Public hospitals
8.5.2 Private hospitals
8.5.3 Specialty clinics and day-care centers
8.5.4 Retail and specialty pharmacies
8.5.5 Others
8.6 By Distribution Channel
8.6.1 Tender-based institutional procurement
8.6.2 Direct sales to hospitals and clinics
8.6.3 Distributors / wholesalers
8.6.4 Retail & online pharmacies
8.6.5 Others
8.7 By Region
8.7.1 Muscat
8.7.2 Dhofar
8.7.3 Al Batinah
8.7.4 Al Dakhiliyah
8.7.5 Others
9. Oman Specialty Injectable Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Revenue from specialty injectable generics (OMR / USD)
9.2.3 3-year revenue CAGR in Oman specialty injectable generics
9.2.4 Market share in Oman specialty injectable generics (%)
9.2.5 EBITDA margin from Oman specialty injectable generics (%)
9.2.6 R&D intensity (% of revenue allocated to injectables)
9.2.7 Capacity utilization rate for sterile injectable lines (%)
9.2.8 Tender win rate with Oman public institutions (%)
9.2.9 Oman-focused portfolio depth (number of registered injectable SKUs)
9.2.10 Average time-to-market for new injectable generic launches (months)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Oman Pharmaceutical Products Co. LLC
9.5.2 Gulf Pharmaceutical Industries (Julphar)
9.5.3 National Pharmaceutical Industries Co. SAOG
9.5.4 Dhofar Pharmaceutical Industries Co. SAOC
9.5.5 Muscat Pharmacy & Stores LLC
9.5.6 Aster DM Healthcare
9.5.7 Badr Al Samaa Group of Hospitals
9.5.8 Apex Medical Group
9.5.9 Pharmazone LLC
9.5.10 Life Pharmacy Oman
9.5.11 Al Hayat International Hospital
9.5.12 NMC Healthcare (Oman operations)
9.5.13 Al Raffah Hospital
9.5.14 Starcare Hospital
9.5.15 Others (regional injectable generic manufacturers and distributors)
10. Oman Specialty Injectable Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Defense
10.1.3 Ministry of Education
10.1.4 Ministry of Social Development
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare infrastructure investments
10.2.2 Energy-efficient solutions in healthcare
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home healthcare providers
10.4 User Readiness for Adoption
10.4.1 Training and education needs
10.4.2 Technology adoption barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of cost savings
10.5.2 Expansion into new therapeutic areas
11. Oman Specialty Injectable Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Business model development
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV, Greenfield, M&A, Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health authorities and pharmaceutical associations in Oman
- Review of published articles and white papers on specialty injectable generics
- Examination of regulatory frameworks and guidelines from the Ministry of Health in Oman
Primary Research
- Interviews with key opinion leaders in the pharmaceutical industry, including pharmacists and hospital administrators
- Surveys with healthcare professionals to understand prescribing patterns and preferences
- Field interviews with representatives from specialty injectable manufacturers and distributors
Validation & Triangulation
- Cross-validation of data through multiple sources, including industry reports and expert interviews
- Triangulation of findings from primary and secondary research to ensure consistency
- Sanity checks through expert panel reviews to validate market assumptions and projections
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in Oman as a baseline
- Segmentation of the market by therapeutic areas and types of specialty injectables
- Incorporation of growth rates based on historical data and market trends
Bottom-up Modeling
- Collection of sales data from leading specialty injectable manufacturers operating in Oman
- Estimation of market share based on product offerings and distribution channels
- Volume and pricing analysis to derive revenue estimates for each segment
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market growth based on historical trends
- Scenario modeling based on potential regulatory changes and healthcare reforms
- Development of best-case, worst-case, and most-likely scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacy Directors | 45 | Pharmacy Managers, Clinical Pharmacists |
| Oncology Specialty Clinics | 40 | Oncologists, Nurse Practitioners |
| Cardiology Departments | 40 | Cardiologists, Cardiac Nurses |
| Government Health Officials | 40 | Health Policy Makers, Regulatory Affairs Specialists |
| Pharmaceutical Distributors | 45 | Distribution Managers, Sales Representatives |
Frequently Asked Questions
What is the current value of the Oman Specialty Injectable Generics Market?
The Oman Specialty Injectable Generics Market is valued at approximately USD 110 million, reflecting a five-year historical analysis. This growth is driven by the increasing prevalence of chronic diseases and the demand for cost-effective treatment options.