About the Report
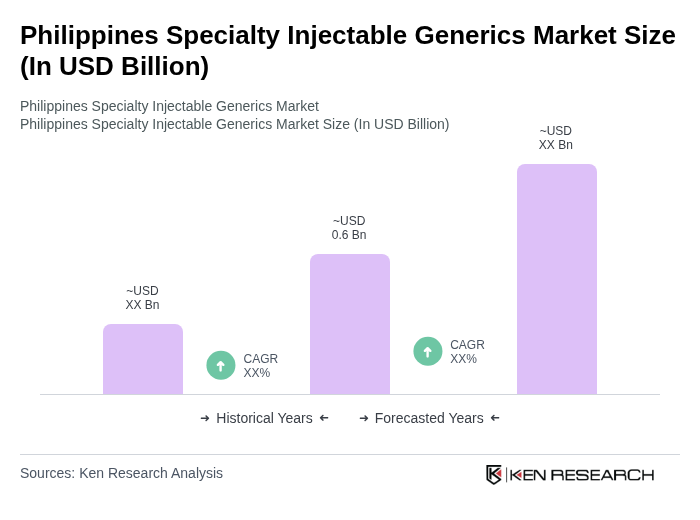
Base Year 2024Philippines Specialty Injectable Generics Market Overview
- The Philippines Specialty Injectable Generics Market is valued at USD 0.6 billion, based on a five-year historical analysis of the broader injectable drug solutions segment and its specialty generic component within the national pharmaceutical market. This growth is primarily driven by the increasing prevalence of chronic and complex diseases such as cancer, autoimmune disorders, and infectious diseases, rising healthcare expenditure, expansion of hospital and specialist care capacity, and the growing demand for affordable, high?value therapies including biosimilar injectables. The market is further supported by government initiatives to promote the use of generics under national medicines and reimbursement policies, which has contributed to greater availability of specialty injectable products in public and private hospital formularies.
- Metro Manila, Cebu, and Davao are the dominant regions in the Philippines Specialty Injectable Generics Market. Metro Manila, being the capital and the largest metropolitan region, has a higher concentration of tertiary and specialty hospitals, specialist clinics, and pharmaceutical distributors, as well as access to advanced sterile?manufacturing and logistics infrastructure. Cebu and Davao act as key hubs for distribution across the Visayas and Mindanao, respectively, leveraging their strategic port and airport connectivity, growing urbanization, and expanding healthcare infrastructure, which collectively strengthen their role as regional centers for injectable drug supply and utilization.
- The Philippines government enacted Republic Act No. 11223, the Universal Health Care (UHC) Act, in 2019, with phased nationwide implementation by the Department of Health (DOH) and the Philippine Health Insurance Corporation (PhilHealth). This law aims to provide all Filipinos with equitable access to quality and affordable healthcare services and explicitly strengthens the role of essential and generic medicines within the national formulary and benefit packages, including hospital?based and specialty injectable therapies. Under the UHC framework, PhilHealth’s National Health Insurance Program integrates the Philippine National Formulary and generic prescribing and dispensing policies, thereby supporting wider availability and financial coverage of generic and biosimilar injectables in accredited facilities.
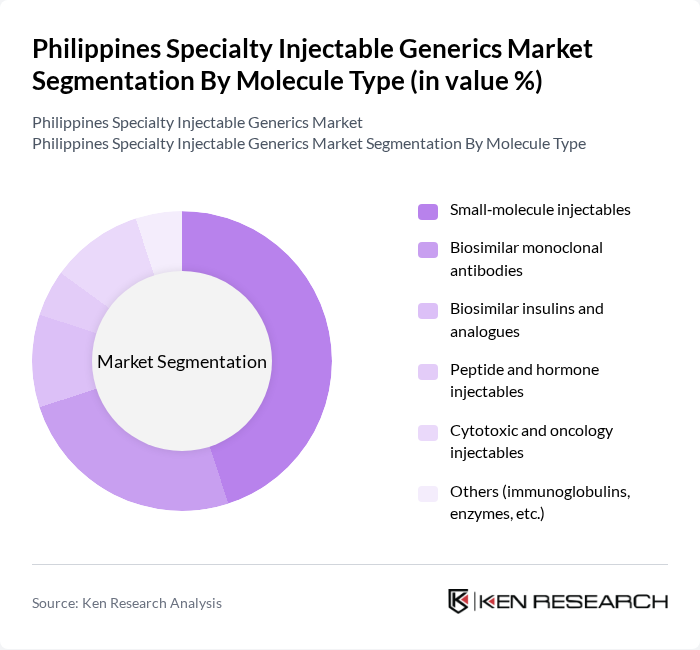
Philippines Specialty Injectable Generics Market Segmentation
By Molecule Type:The market is segmented into various molecule types, including small-molecule injectables, biosimilar monoclonal antibodies, biosimilar insulins and analogues, peptide and hormone injectables, cytotoxic and oncology injectables, and others such as immunoglobulins and enzymes. Within the broader injectable solutions market in the Philippines, biologic and biosimilar injectables (such as monoclonal antibodies and other complex biologics) are gaining a growing share due to the rising burden of oncology and autoimmune diseases and the global shift toward high?value biologic therapies. Small-molecule injectables, however, continue to account for a substantial portion of hospital and critical?care use because of their established role across multiple therapeutic areas (anti?infectives, anesthesia, cardiovascular and critical care) and their comparatively lower cost. The increasing demand for biosimilars in oncology, autoimmune conditions, and diabetes care reflects payer and provider focus on cost?effective alternatives to originator biologics, enhancing uptake of biosimilar monoclonal antibodies and biosimilar insulins and analogues in tertiary hospitals and government procurement.
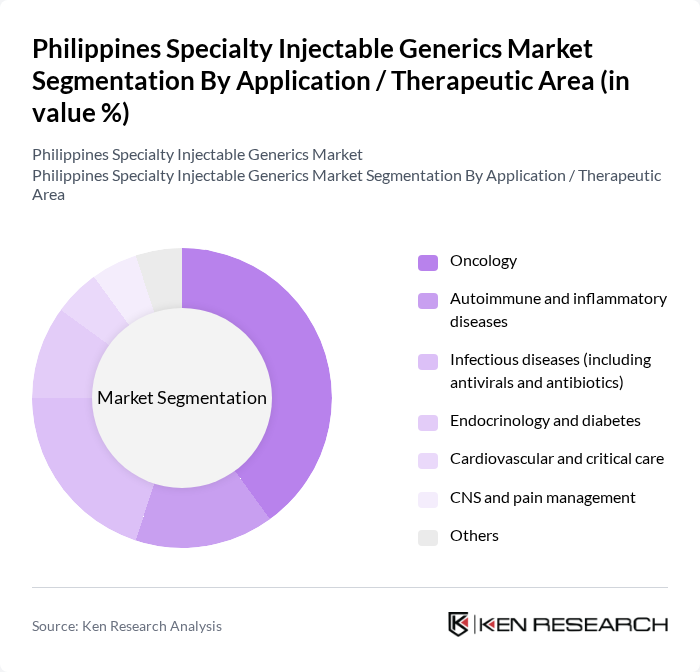
By Application / Therapeutic Area:The market is further segmented by application into oncology, autoimmune and inflammatory diseases, infectious diseases (including antivirals and antibiotics), endocrinology and diabetes, cardiovascular and critical care, CNS and pain management, and others. Oncology injectables lead the market in value terms, aligned with the high and growing cancer burden in the Philippines and the increasing use of targeted biologic and biosimilar therapies in tertiary hospitals and cancer centers. Demand for injectables in infectious diseases remains strong due to the continued need for parenteral antibiotics and antivirals in hospital settings, while endocrinology and diabetes care is seeing rising use of insulin and GLP?1 analog injectables in both public and private sectors. Cardiovascular and critical care, CNS and pain management, and other specialties also contribute significantly to utilization, reflecting the role of injectable formulations in emergency, intensive care, and perioperative management.
Philippines Specialty Injectable Generics Market Competitive Landscape
The Philippines Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer, Inc. (Philippines), Novartis AG / Sandoz (Philippines), Teva Pharmaceutical Industries Ltd., Mylan N.V. (Viatris), Fresenius Kabi Philippines, Inc., Hikma Pharmaceuticals PLC, Baxter International Inc., Lupin Limited, Cipla Limited, Dr. Reddy's Laboratories Ltd., Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Local Philippine generic manufacturers active in injectables, Regional ASEAN injectable generic players present in the Philippines, Emerging biosimilar players targeting the Philippines market contribute to innovation, geographic expansion, and service delivery in this space.
Philippines Specialty Injectable Generics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The Philippines is experiencing a significant rise in chronic diseases, with the Department of Health reporting that non-communicable diseases account for 68% of total deaths in future. This trend is expected to continue, with an estimated 1.2 million Filipinos diagnosed with diabetes in future. The growing patient population drives demand for specialty injectable generics, as these treatments are often more affordable than branded alternatives, making them accessible to a larger segment of the population.
- Rising Demand for Cost-Effective Treatment Options:With healthcare expenditures in the Philippines reaching approximately PHP 1.2 trillion in future, the need for cost-effective treatment options is paramount. Specialty injectable generics offer a viable solution, as they can reduce treatment costs by up to 30% compared to branded drugs. This financial incentive is crucial for both healthcare providers and patients, particularly in a country where out-of-pocket expenses account for 54% of total health spending, according to the World Bank.
- Expansion of Healthcare Infrastructure:The Philippine government has committed to enhancing healthcare infrastructure, with a budget allocation of PHP 100 billion for health facilities in future. This investment aims to improve access to healthcare services, particularly in rural areas. As healthcare facilities expand, the demand for specialty injectable generics is expected to rise, as these products are essential for treating various conditions, including cancer and autoimmune diseases, which require specialized care.
Market Challenges
- Regulatory Hurdles and Compliance Issues:The regulatory landscape for specialty injectable generics in the Philippines is complex, with the Food and Drug Administration (FDA) imposing stringent compliance requirements. In future, over 40% of generic drug applications faced delays due to regulatory challenges. These hurdles can hinder market entry for new products, limiting competition and potentially affecting the availability of affordable treatment options for patients.
- Competition from Branded Pharmaceuticals:The presence of established branded pharmaceuticals poses a significant challenge to the specialty injectable generics market. In future, branded drugs accounted for 75% of the total pharmaceutical market share in the Philippines. This dominance is attributed to brand loyalty and perceived efficacy, making it difficult for generics to penetrate the market despite their cost advantages. The challenge lies in convincing healthcare providers and patients to consider generics as viable alternatives.
Philippines Specialty Injectable Generics Market Future Outlook
The future of the specialty injectable generics market in the Philippines appears promising, driven by increasing healthcare investments and a growing focus on affordable treatment options. As the government continues to enhance healthcare infrastructure, access to specialty generics is expected to improve significantly. Additionally, the rising prevalence of chronic diseases will further fuel demand. However, addressing regulatory challenges and increasing awareness among healthcare providers will be crucial for maximizing market potential and ensuring patient access to these essential medications.
Market Opportunities
- Growth in the Aging Population:The Philippines is witnessing a demographic shift, with the elderly population projected to reach 10 million in future. This demographic change will increase the demand for specialty injectable generics, as older adults often require chronic disease management. Targeting this segment presents a significant opportunity for manufacturers to develop tailored products that meet the specific needs of aging patients.
- Partnerships with Local Healthcare Providers:Collaborating with local healthcare providers can enhance market penetration for specialty injectable generics. By forming strategic partnerships, manufacturers can leverage local knowledge and distribution networks, improving access to these products. Such collaborations can also facilitate educational initiatives aimed at increasing awareness and acceptance of generics among healthcare professionals and patients alike.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Molecule Type | Small?molecule injectables Biosimilar monoclonal antibodies Biosimilar insulins and analogues Peptide and hormone injectables Cytotoxic and oncology injectables Others (immunoglobulins, enzymes, etc.) |
| By Application / Therapeutic Area | Oncology Autoimmune and inflammatory diseases Infectious diseases (including antivirals and antibiotics) Endocrinology and diabetes Cardiovascular and critical care CNS and pain management Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Intrathecal and other parenteral routes |
| By Packaging Format | Single?dose vials Multi?dose vials Ampoules Prefilled syringes and pens Cartridges and others |
| By End?Use Setting | Public hospitals (DOH and LGU facilities) Private hospitals and specialty centers Hospital and specialty pharmacies Retail and chain pharmacies Home?care and ambulatory care providers |
| By Distribution Channel | Direct sales to institutions National and regional distributors Tender?based government procurement Others |
| By Region | Luzon Visayas Mindanao |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Department of Health)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Institutions
Industry Associations (e.g., Pharmaceutical and Healthcare Association of the Philippines)
Financial Institutions
Players Mentioned in the Report:
Pfizer, Inc. (Philippines)
Novartis AG / Sandoz (Philippines)
Teva Pharmaceutical Industries Ltd.
Mylan N.V. (Viatris)
Fresenius Kabi Philippines, Inc.
Hikma Pharmaceuticals PLC
Baxter International Inc.
Lupin Limited
Cipla Limited
Dr. Reddy's Laboratories Ltd.
Sun Pharmaceutical Industries Ltd.
Aurobindo Pharma Ltd.
Local Philippine generic manufacturers active in injectables
Regional ASEAN injectable generic players present in the Philippines
Emerging biosimilar players targeting the Philippines market
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Philippines Specialty Injectable Generics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Philippines Specialty Injectable Generics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Philippines Specialty Injectable Generics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for cost-effective treatment options
3.1.3 Expansion of healthcare infrastructure
3.1.4 Government initiatives to promote generics
3.2 Market Challenges
3.2.1 Regulatory hurdles and compliance issues
3.2.2 Competition from branded pharmaceuticals
3.2.3 Limited awareness among healthcare providers
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Growth in the aging population
3.3.2 Increasing investment in R&D for generics
3.3.3 Expansion into rural healthcare markets
3.3.4 Partnerships with local healthcare providers
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Adoption of digital health technologies
3.4.3 Increasing focus on sustainability in production
3.4.4 Growth of telemedicine and remote patient monitoring
3.5 Government Regulation
3.5.1 Implementation of the Generics Act
3.5.2 Price control measures for essential medicines
3.5.3 Regulatory guidelines for biosimilars
3.5.4 Quality assurance standards for injectable generics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Philippines Specialty Injectable Generics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Philippines Specialty Injectable Generics Market Segmentation
8.1 By Molecule Type
8.1.1 Small?molecule injectables
8.1.2 Biosimilar monoclonal antibodies
8.1.3 Biosimilar insulins and analogues
8.1.4 Peptide and hormone injectables
8.1.5 Cytotoxic and oncology injectables
8.1.6 Others (immunoglobulins, enzymes, etc.)
8.2 By Application / Therapeutic Area
8.2.1 Oncology
8.2.2 Autoimmune and inflammatory diseases
8.2.3 Infectious diseases (including antivirals and antibiotics)
8.2.4 Endocrinology and diabetes
8.2.5 Cardiovascular and critical care
8.2.6 CNS and pain management
8.2.7 Others
8.3 By Route of Administration
8.3.1 Intravenous (IV)
8.3.2 Intramuscular (IM)
8.3.3 Subcutaneous (SC)
8.3.4 Intrathecal and other parenteral routes
8.4 By Packaging Format
8.4.1 Single?dose vials
8.4.2 Multi?dose vials
8.4.3 Ampoules
8.4.4 Prefilled syringes and pens
8.4.5 Cartridges and others
8.5 By End?Use Setting
8.5.1 Public hospitals (DOH and LGU facilities)
8.5.2 Private hospitals and specialty centers
8.5.3 Hospital and specialty pharmacies
8.5.4 Retail and chain pharmacies
8.5.5 Home?care and ambulatory care providers
8.6 By Distribution Channel
8.6.1 Direct sales to institutions
8.6.2 National and regional distributors
8.6.3 Tender?based government procurement
8.6.4 Others
8.7 By Region
8.7.1 Luzon
8.7.2 Visayas
8.7.3 Mindanao
9. Philippines Specialty Injectable Generics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Ownership type (local, multinational affiliate, regional generic player)
9.2.3 Philippines specialty injectable generics revenue (latest year, US$ or PHP)
9.2.4 3?year revenue CAGR in specialty injectable generics (%)
9.2.5 Philippines market share in specialty injectable generics (%)
9.2.6 Share of injectables in total Philippines revenues (%)
9.2.7 Portfolio breadth (number of SKUs / molecules in injectables)
9.2.8 Biosimilar and complex generics share of portfolio (%)
9.2.9 Average realized price vs market benchmark (%)
9.2.10 EBITDA margin for Philippines operations (%)
9.2.11 Capacity utilization of sterile injectable lines (%)
9.2.12 Share of institutional / tender sales in total revenues (%)
9.2.13 Sales force / KAM coverage (number of hospital accounts covered)
9.2.14 Regulatory track record (approvals, warning letters, recalls – 3?year view)
9.2.15 R&D and filing pipeline (number of pending injectable ANDAs / dossiers)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Pfizer, Inc. (Philippines)
9.5.2 Novartis AG / Sandoz (Philippines)
9.5.3 Teva Pharmaceutical Industries Ltd.
9.5.4 Mylan N.V. (Viatris)
9.5.5 Fresenius Kabi Philippines, Inc.
9.5.6 Hikma Pharmaceuticals PLC
9.5.7 Baxter International Inc.
9.5.8 Lupin Limited
9.5.9 Cipla Limited
9.5.10 Dr. Reddy's Laboratories Ltd.
9.5.11 Sun Pharmaceutical Industries Ltd.
9.5.12 Aurobindo Pharma Ltd.
9.5.13 Local Philippine generic manufacturers active in injectables
9.5.14 Regional ASEAN injectable generic players present in the Philippines
9.5.15 Emerging biosimilar players targeting the Philippines market
10. Philippines Specialty Injectable Generics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Healthcare
10.1.2 Procurement Processes and Regulations
10.1.3 Preferred Suppliers and Partnerships
10.1.4 Evaluation Criteria for Bids
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Spending on Medical Equipment
10.2.3 Budget for Injectable Generics
10.2.4 Trends in Corporate Healthcare Spending
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Affordable Medications
10.3.2 Quality Assurance Concerns
10.3.3 Supply Chain Reliability
10.3.4 Training and Education Needs
10.4 User Readiness for Adoption
10.4.1 Awareness of Injectable Generics
10.4.2 Training Programs for Healthcare Providers
10.4.3 Infrastructure Readiness
10.4.4 Patient Acceptance Levels
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Cost Savings
10.5.2 Impact on Patient Outcomes
10.5.3 Opportunities for Product Line Expansion
10.5.4 Long-term Sustainability of Use Cases
11. Philippines Specialty Injectable Generics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from pharmaceutical associations in the Philippines
- Review of government publications on healthcare spending and drug approvals
- Examination of academic journals and articles focusing on injectable generics
Primary Research
- Interviews with key opinion leaders in the pharmaceutical industry
- Surveys with healthcare professionals regarding prescribing habits
- Focus groups with patients to understand preferences for injectable generics
Validation & Triangulation
- Cross-validation of data from multiple sources including industry reports and expert interviews
- Triangulation of market trends with sales data from pharmacies and hospitals
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size in the Philippines
- Segmentation of the injectable generics market by therapeutic area
- Analysis of historical growth rates and market trends
Bottom-up Modeling
- Collection of sales data from leading injectable generics manufacturers
- Estimation of market share based on production volumes and pricing
- Calculation of growth potential based on new product launches and market entry
Forecasting & Scenario Analysis
- Development of forecasts based on demographic trends and healthcare policies
- Scenario analysis considering regulatory changes and market dynamics
- Projections of market growth under different economic conditions through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Distributors | 60 | Distribution Managers, Sales Directors |
| Healthcare Providers | 120 | Doctors, Pharmacists, Hospital Administrators |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Patients Using Injectable Generics | 80 | Patients, Caregivers, Patient Advocacy Group Members |
| Market Analysts | 50 | Market Researchers, Industry Analysts |
Frequently Asked Questions
What is the current value of the Philippines Specialty Injectable Generics Market?
The Philippines Specialty Injectable Generics Market is valued at approximately USD 0.6 billion, reflecting a significant component of the broader injectable drug solutions segment within the national pharmaceutical market.