Region:Middle East
Author(s):Rebecca
Product Code:KRAD5034
Pages:81
Published On:December 2025
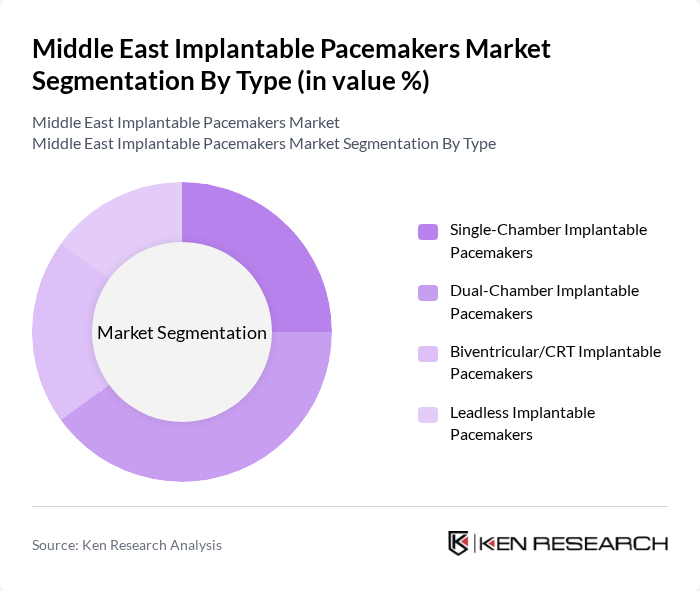
By Type:The market is segmented into various types of implantable pacemakers, including Single-Chamber, Dual-Chamber, Biventricular/CRT, and Leadless Implantable Pacemakers. At the global and regional level, single- and dual-chamber pacemakers represent the largest volume segments, with implantable/internal pacemakers accounting for the majority share of the overall pacemaker market. Dual-Chamber Implantable Pacemakers are widely adopted due to their ability to provide more physiologic heart rate management and improved patient outcomes in many bradyarrhythmia indications. The increasing prevalence of arrhythmias, expanding indications for device therapy, and the growing preference for advanced, programmable devices are driving the demand for Dual-Chamber models. Additionally, technological advancements such as MRI-compatible systems, longer battery life, miniaturization, and the introduction of leadless pacemakers are contributing to the increasing clinical preference for advanced pacemaker types across Middle Eastern cardiac centers.
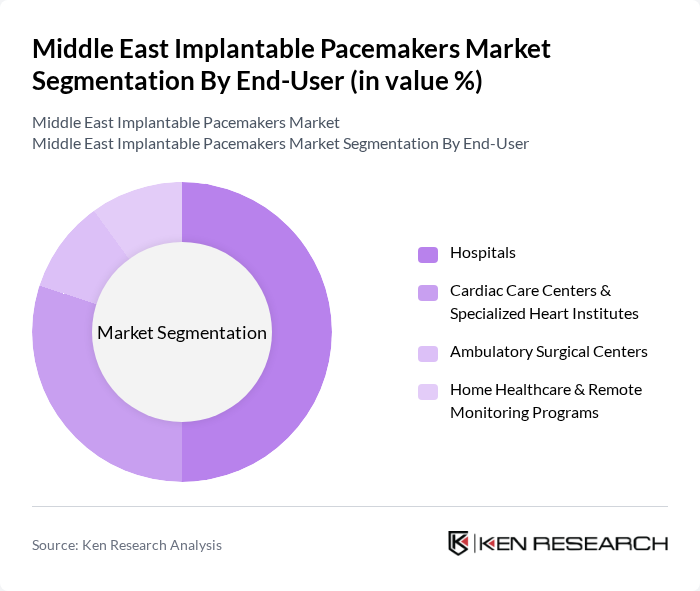
By End-User:The end-user segmentation includes Hospitals, Cardiac Care Centers & Specialized Heart Institutes, Ambulatory Surgical Centers, and Home Healthcare & Remote Monitoring Programs. Hospitals are the dominant end-user segment, accounting for a significant share of pacemaker implant procedures globally and in the Middle East, as most device implantations and follow?up interrogations are conducted in hospital-based cardiology and electrophysiology units. This is primarily due to the high volume of cardiac procedures performed in hospitals, the availability of catheterization laboratories and operating rooms, and access to multidisciplinary teams and advanced imaging technologies. Furthermore, the increasing number of cardiac surgeries, expansion of specialized heart institutes, and wider use of remote device interrogation and telemonitoring programs anchored in hospital or cardiac center networks contribute to the strong demand for implantable pacemakers in hospital and specialized cardiac care settings across the Middle East.
The Middle East Implantable Pacemakers Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Abbott Laboratories (Abbott Cardiovascular), Boston Scientific Corporation, BIOTRONIK SE & Co. KG, MicroPort Scientific Corporation (Cardiac Rhythm Management), LivaNova PLC, Shree Pacetronix Ltd., Lepu Medical Technology (Beijing) Co., Ltd., Osypka Medical GmbH, Medico S.p.A., Cook Medical LLC, ELA Medical (Historical Sorin Group / LivaNova Brand), Regional Distributors and Importers in GCC (e.g., Gulf Medical Co., Tamer Group), Emerging Local & Regional Manufacturers in MEA Active Implantable Devices, Other Global Cardiac Rhythm Management Players Active in the Region contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East implantable pacemakers market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centric care. As healthcare systems invest in expanding infrastructure and integrating telehealth services, the accessibility of cardiac care is expected to improve. Additionally, the rise of minimally invasive procedures will likely enhance patient outcomes, further stimulating demand for implantable pacemakers in the region, fostering a more robust healthcare environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Single-Chamber Implantable Pacemakers Dual-Chamber Implantable Pacemakers Biventricular/CRT Implantable Pacemakers Leadless Implantable Pacemakers |
| By End-User | Hospitals Cardiac Care Centers & Specialized Heart Institutes Ambulatory Surgical Centers Home Healthcare & Remote Monitoring Programs |
| By Patient Demographics | Pediatric Patients Adult Patients (18–64 Years) Geriatric Patients (65+ Years) High-Risk & Comorbid Patient Groups |
| By Distribution Channel | Direct Tenders to Hospitals and Health Ministries Regional Distributors and Importers Group Purchasing Organizations (GPOs) E-Procurement and Online Hospital Purchasing Portals |
| By Geography | GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Oman, Bahrain) Levant Region (Jordan, Lebanon, Iraq, Others) Rest of Middle East (Iran, Israel, Yemen, Others) North Africa (Egypt and Neighboring Markets with MEA Clusters) |
| By Technology | Conventional Implantable Pacemakers MRI-Compatible Implantable Pacemakers Leadless Implantable Pacemakers Remote Monitoring-Enabled & Connected Pacemakers |
| By Application | Arrhythmia and Bradycardia Management Heart Failure and Cardiac Resynchronization Therapy (CRT) Atrioventricular Block and Conduction Disorders Others (Syncope, Post-Myocardial Infarction Indications) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 120 | Cardiologists, Electrophysiologists |
| Medical Device Distributors | 90 | Sales Managers, Product Specialists |
| Healthcare Procurement Managers | 70 | Procurement Officers, Supply Chain Managers |
| Patient Advocacy Groups | 50 | Patient Representatives, Health Advocates |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
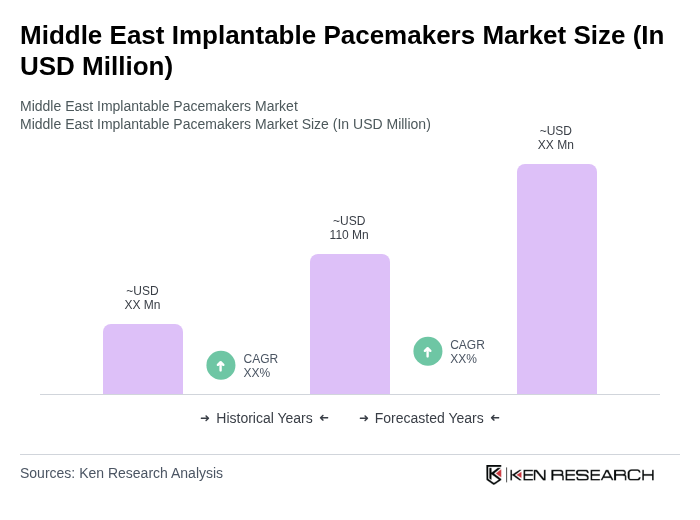
The Middle East Implantable Pacemakers Market is valued at approximately USD 110 million, reflecting significant growth driven by the rising prevalence of cardiovascular diseases and advancements in medical technology.