About the Report
Base Year 2024Middle East Pacific Medical Injection Molding Market Overview
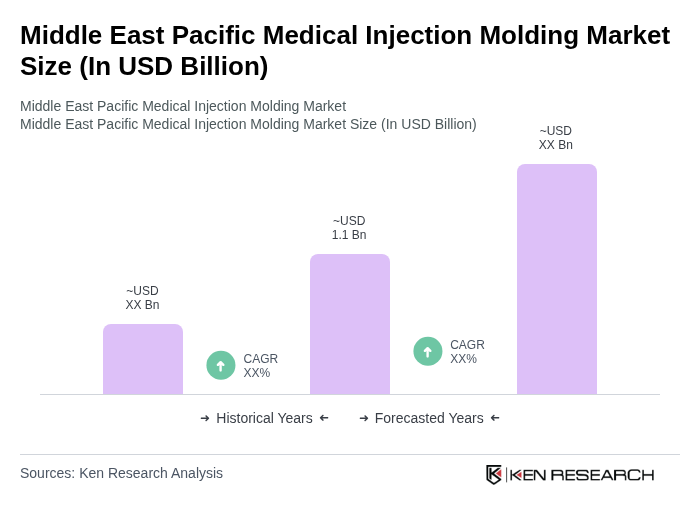
- The Middle East Pacific Medical Injection Molding Market is valued at approximately USD 1.1 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for medical devices and components, coupled with advancements in manufacturing technologies that enhance production efficiency and quality. The rising prevalence of chronic diseases and the need for innovative healthcare solutions, including single-use consumables, diagnostic components, and minimally invasive device parts, further fuel market expansion as healthcare providers focus on infection control, cost-effective care, and localized manufacturing.
- Key players in this market include countries like the United Arab Emirates, Saudi Arabia, and Israel, which dominate due to their robust healthcare infrastructure, significant investments in medical technology, and a growing population that demands high-quality medical services. These nations are also home to leading medical device manufacturers and contract manufacturers serving regional hubs, supported by strong injection molding capabilities for plastics used in consumables, patient aids, and orthopedic and dental products, contributing to their market leadership.
- In the United Arab Emirates, medical device manufacturing is governed by a comprehensive framework that requires conformity with internationally recognized quality and safety standards. The key binding instrument is the Medical Devices Regulation issued under Cabinet Resolution No. 40 of 2016 by the UAE Cabinet, administered operationally by the Ministry of Health and Prevention (MOHAP) and other competent authorities. This regulation mandates that medical devices, including injection-molded components, comply with quality management standards such as ISO 13485 and relevant ISO material and biocompatibility standards, and it sets requirements for registration, licensing of manufacturers and importers, technical documentation, labeling, and post-market surveillance. Compliance with these requirements is crucial for manufacturers seeking to market medical injection?molded products in the UAE and, by extension, to use the country as a regional distribution base.
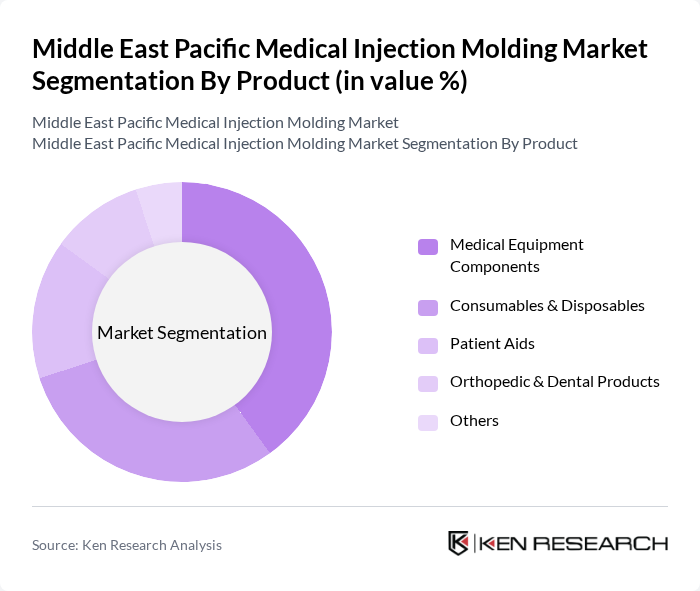
Middle East Pacific Medical Injection Molding Market Segmentation
By Product:The product segmentation of the market includes various categories such as Medical Equipment Components, Consumables & Disposables, Patient Aids, Orthopedic & Dental Products, and Others. Among these, Medical Equipment Components are leading due to their essential role in the production of critical medical devices and diagnostic systems, which are increasingly in demand as healthcare systems adopt more complex imaging, monitoring, and minimally invasive therapy solutions. Consumables and disposables also account for a substantial share, driven by infection-prevention protocols, higher procedure volumes, and the regional shift toward single-use products in hospitals and outpatient settings.
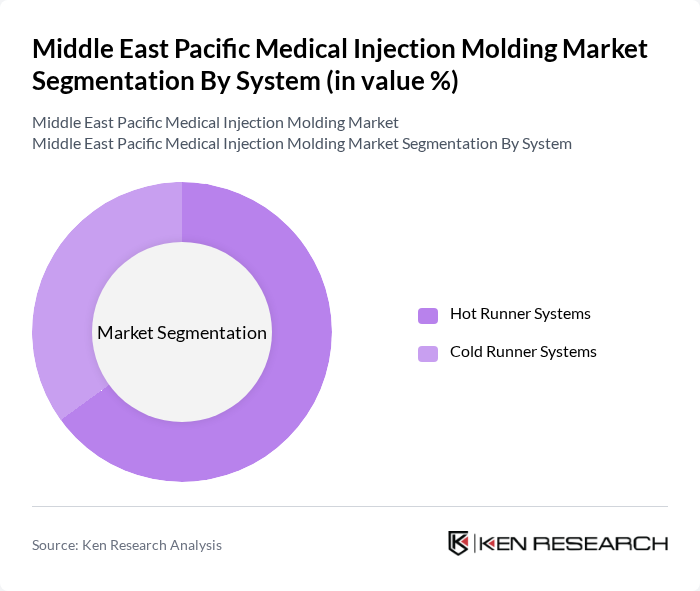
By System:The market is also segmented by system types, including Hot Runner Systems and Cold Runner Systems. Hot Runner Systems are currently dominating the market due to their efficiency in reducing material waste and improving cycle times, which is crucial for manufacturers aiming to optimize production processes and reduce costs in high-volume medical device and consumable manufacturing. These systems support precise molding of complex geometries required in medtech applications and are increasingly favored in regional facilities that supply both local hospitals and export markets.
Middle East Pacific Medical Injection Molding Market Competitive Landscape
The Middle East Pacific Medical Injection Molding Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (including Ethicon), Becton, Dickinson and Company (BD), Stryker Corporation, Boston Scientific Corporation, Thermo Fisher Scientific Inc., 3M Company, Baxter International Inc., GE HealthCare Technologies Inc., Philips Healthcare (Koninklijke Philips N.V.), Siemens Healthineers AG, Canon Medical Systems Corporation, FUJIFILM Healthcare, Olympus Corporation, Hitachi Medical Systems contribute to innovation, geographic expansion, and service delivery in this space, particularly through localized distribution, regional manufacturing partnerships, and the use of advanced injection molding technologies for device components and consumables.
Middle East Pacific Medical Injection Molding Market Industry Analysis
Growth Drivers
- Increasing Demand for Medical Devices:The Middle East is witnessing a surge in demand for medical devices, driven by a growing population and rising prevalence of chronic diseases. In future, the region's healthcare market is projected to reach approximately $144 billion, with medical devices accounting for approximately $20–25 billion. This growth is fueled by advancements in technology and increased healthcare access, leading to a higher consumption of medical devices, which in turn drives the demand for injection molding services.
- Technological Advancements in Injection Molding:The injection molding sector is experiencing rapid technological advancements, enhancing production efficiency and product quality. In future, investments in advanced molding technologies, such as multi-material and 3D printing, in the Middle East are expected to reach more than $1 billion. These innovations enable manufacturers to produce complex medical components with precision, thereby meeting the stringent requirements of the healthcare industry and driving market growth.
- Rising Healthcare Expenditure:Healthcare expenditure in the Middle East is projected to reach around $200 billion in future, driven primarily by government initiatives to improve healthcare infrastructure and services. As healthcare spending rises, the demand for high-quality medical devices and components manufactured through injection molding is expected to grow, further propelling the market forward.
Market Challenges
- High Initial Investment Costs:The medical injection molding industry faces significant barriers due to high initial investment costs, which can exceed $2 million for advanced machinery and setup. This financial burden can deter new entrants and limit the expansion of existing manufacturers. As a result, many companies struggle to adopt the latest technologies, hindering their competitiveness in a rapidly evolving market.
- Stringent Regulatory Compliance:Compliance with stringent regulatory standards poses a significant challenge for manufacturers in the Middle East. The region's medical device regulations require extensive testing and certification processes, which can take up to 18 months and incur costs of approximately $500,000. These regulatory hurdles can delay product launches and increase operational costs, impacting the overall market growth.
Middle East Pacific Medical Injection Molding Market Future Outlook
The future of the Middle East Pacific medical injection molding market appears promising, driven by ongoing technological advancements and increasing healthcare investments. As manufacturers adopt automation and smart manufacturing practices, production efficiency is expected to improve significantly. Additionally, the growing emphasis on sustainability will likely lead to the development of eco-friendly materials and processes, aligning with global trends. This evolution will create a dynamic landscape for innovation and growth in the medical injection molding sector.
Market Opportunities
- Growth in Biodegradable Materials:The rising demand for environmentally friendly products presents a significant opportunity for the injection molding market. In future, the biodegradable plastics market in the Middle East is expected to be worth around $6 billion, encouraging manufacturers to innovate and incorporate sustainable materials into their production processes, thus attracting eco-conscious clients.
- Increasing Adoption of Automation:The shift towards automation in manufacturing processes is set to enhance productivity and reduce labor costs. In future, investments in automation technologies in the Middle East are projected to exceed $1 billion. This trend will enable manufacturers to streamline operations, improve product quality, and respond more effectively to market demands, creating a competitive edge.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product | Medical Equipment Components Consumables & Disposables Patient Aids Orthopedic & Dental Products Others |
| By System | Hot Runner Systems Cold Runner Systems |
| By Material | Plastics Metals Others |
| By Class of Device | Class I Class II Class III |
| By Application | Surgical Instruments Diagnostic Devices Drug Delivery Systems Pharmaceutical & Medical Packaging Others |
| By End User | Medical Device OEMs Pharmaceutical & Biotech Companies Contract Manufacturing Organizations (CMOs) Others |
| By Region | GCC Countries Levant Region North Africa Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Medical Device Manufacturers
Pharmaceutical Companies
Healthcare Providers and Hospitals
Supply Chain and Logistics Companies
Industry Associations (e.g., Medical Device Manufacturers Association)
Insurance Companies and Healthcare Payers
Players Mentioned in the Report:
Medtronic plc
Johnson & Johnson (including Ethicon)
Becton, Dickinson and Company (BD)
Stryker Corporation
Boston Scientific Corporation
Thermo Fisher Scientific Inc.
3M Company
Baxter International Inc.
GE HealthCare Technologies Inc.
Philips Healthcare (Koninklijke Philips N.V.)
Siemens Healthineers AG
Canon Medical Systems Corporation
FUJIFILM Healthcare
Olympus Corporation
Hitachi Medical Systems
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Pacific Medical Injection Molding Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Pacific Medical Injection Molding Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Pacific Medical Injection Molding Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Medical Devices
3.1.2 Technological Advancements in Injection Molding
3.1.3 Rising Healthcare Expenditure
3.1.4 Expansion of Healthcare Infrastructure
3.2 Market Challenges
3.2.1 High Initial Investment Costs
3.2.2 Stringent Regulatory Compliance
3.2.3 Competition from Low-Cost Manufacturers
3.2.4 Supply Chain Disruptions
3.3 Market Opportunities
3.3.1 Growth in Biodegradable Materials
3.3.2 Increasing Adoption of Automation
3.3.3 Expansion into Emerging Markets
3.3.4 Development of Custom Medical Solutions
3.4 Market Trends
3.4.1 Shift Towards Sustainable Manufacturing
3.4.2 Integration of IoT in Manufacturing Processes
3.4.3 Customization and Personalization of Medical Products
3.4.4 Focus on Quality and Safety Standards
3.5 Government Regulation
3.5.1 Medical Device Regulations
3.5.2 Environmental Compliance Standards
3.5.3 Import and Export Regulations
3.5.4 Labor and Safety Regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Pacific Medical Injection Molding Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Pacific Medical Injection Molding Market Segmentation
8.1 By Product
8.1.1 Medical Equipment Components
8.1.2 Consumables & Disposables
8.1.3 Patient Aids
8.1.4 Orthopedic & Dental Products
8.1.5 Others
8.2 By System
8.2.1 Hot Runner Systems
8.2.2 Cold Runner Systems
8.3 By Material
8.3.1 Plastics
8.3.2 Metals
8.3.3 Others
8.4 By Class of Device
8.4.1 Class I
8.4.2 Class II
8.4.3 Class III
8.5 By Application
8.5.1 Surgical Instruments
8.5.2 Diagnostic Devices
8.5.3 Drug Delivery Systems
8.5.4 Pharmaceutical & Medical Packaging
8.5.5 Others
8.6 By End User
8.6.1 Medical Device OEMs
8.6.2 Pharmaceutical & Biotech Companies
8.6.3 Contract Manufacturing Organizations (CMOs)
8.6.4 Others
8.7 By Region
8.7.1 GCC Countries
8.7.2 Levant Region
8.7.3 North Africa
8.7.4 Others
9. Middle East Pacific Medical Injection Molding Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Revenue (Regional, USD Million)
9.2.3 3-Year Revenue CAGR
9.2.4 EBITDA Margin
9.2.5 Return on Invested Capital (ROIC)
9.2.6 R&D Intensity (% of Revenue)
9.2.7 Capex Intensity (% of Revenue)
9.2.8 Market Share in Target Segment (%)
9.2.9 Average Selling Price Positioning
9.2.10 Capacity Utilization Rate
9.2.11 On?Time Delivery Performance
9.2.12 Customer Retention / Renewal Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Johnson & Johnson (including Ethicon)
9.5.3 Becton, Dickinson and Company (BD)
9.5.4 Stryker Corporation
9.5.5 Boston Scientific Corporation
9.5.6 Thermo Fisher Scientific Inc.
9.5.7 3M Company
9.5.8 Baxter International Inc.
9.5.9 GE HealthCare Technologies Inc.
9.5.10 Philips Healthcare (Koninklijke Philips N.V.)
9.5.11 Siemens Healthineers AG
9.5.12 Canon Medical Systems Corporation
9.5.13 FUJIFILM Healthcare
9.5.14 Olympus Corporation
9.5.15 Hitachi Medical Systems
10. Middle East Pacific Medical Injection Molding Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Procurement Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Medical Infrastructure
10.2.2 Energy Efficiency Initiatives
10.2.3 Budgeting for Medical Equipment
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Clinics
10.3.3 Research Laboratory Needs
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion Opportunities
10.5.3 User Feedback Mechanisms
10.5.4 Others
11. Middle East Pacific Medical Injection Molding Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from regional medical device associations and trade bodies
- Market analysis publications focusing on injection molding technologies in the healthcare sector
- Government publications and healthcare expenditure reports from Middle Eastern countries
Primary Research
- Interviews with key opinion leaders in the medical device manufacturing sector
- Surveys with procurement managers at hospitals and clinics regarding their sourcing practices
- Field interviews with engineers and production managers in injection molding facilities
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert insights
- Triangulation of market size estimates using sales data and production capacity metrics
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of healthcare spending trends across the Middle East to estimate demand for medical devices
- Segmentation of the market by product type, application, and end-user demographics
- Incorporation of regional healthcare initiatives and government funding for medical technology
Bottom-up Modeling
- Volume estimates based on production capacities of leading injection molding manufacturers
- Cost analysis derived from material sourcing and operational expenses in the medical sector
- Estimation of market share based on sales data from key players in the injection molding market
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors such as population growth and aging demographics
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Medical Device Manufacturers | 120 | Production Managers, R&D Directors |
| Healthcare Providers | 90 | Procurement Officers, Hospital Administrators |
| Injection Molding Suppliers | 80 | Sales Managers, Technical Support Engineers |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
| Industry Experts and Consultants | 60 | Market Analysts, Healthcare Consultants |
Frequently Asked Questions
What is the current value of the Middle East Pacific Medical Injection Molding Market?
The Middle East Pacific Medical Injection Molding Market is valued at approximately USD 1.1 billion, driven by the increasing demand for medical devices and advancements in manufacturing technologies that enhance production efficiency and quality.