About the Report
Base Year 2024Middle East Primary Biliary Cholangitis Therapeutics Market Overview
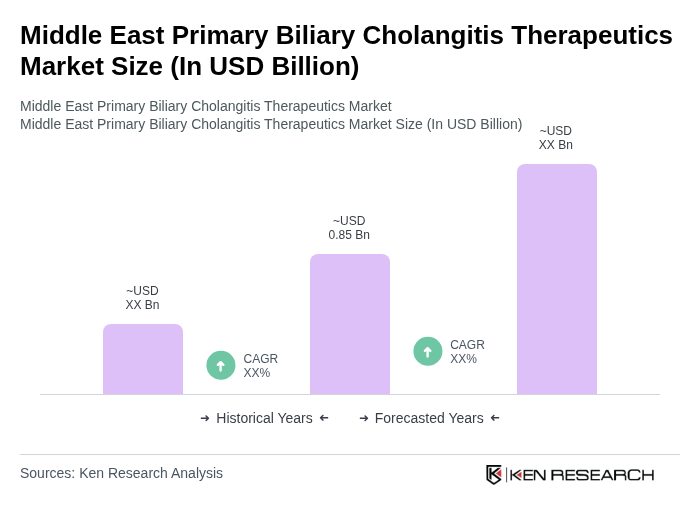
- The Middle East Primary Biliary Cholangitis Therapeutics Market is valued at USD 0.85 billion, based on a five-year historical analysis. This growth is primarily driven by increasing healthcare spending, enhanced diagnostic services, and rising awareness of autoimmune liver conditions. The introduction of specialized liver disease programs and improvements in first-line screening are also contributing to the demand for PBC therapies.
- Key countries dominating the market include Saudi Arabia and the UAE, which have advanced tertiary care infrastructure and a growing emphasis on specialist access. These nations are leading the way in the adoption of PBC therapeutics, supported by government initiatives and investments in healthcare technology.
- In 2024, the UAE government announced a strategic initiative to enhance access to rare disease treatments, including the formal inclusion of innovative therapies into insurance networks. This initiative aims to streamline reimbursement processes and improve patient access to cutting-edge liver care options.
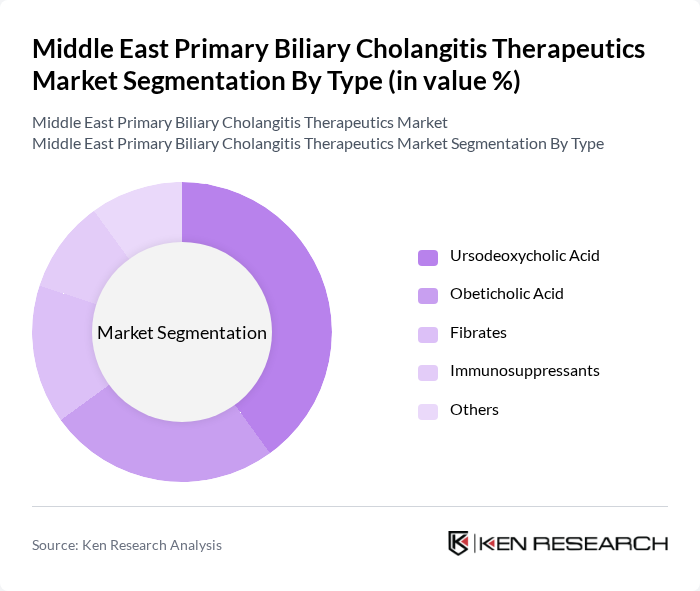
Middle East Primary Biliary Cholangitis Therapeutics Market Segmentation
By Type:The market is segmented into various types of therapeutics, including Ursodeoxycholic Acid, Obeticholic Acid, Fibrates, Immunosuppressants, and Others. Among these, Ursodeoxycholic Acid is the leading sub-segment due to its established efficacy in treating PBC and its widespread acceptance among healthcare providers. The growing prevalence of PBC and the increasing number of patients seeking treatment are driving the demand for these therapies.
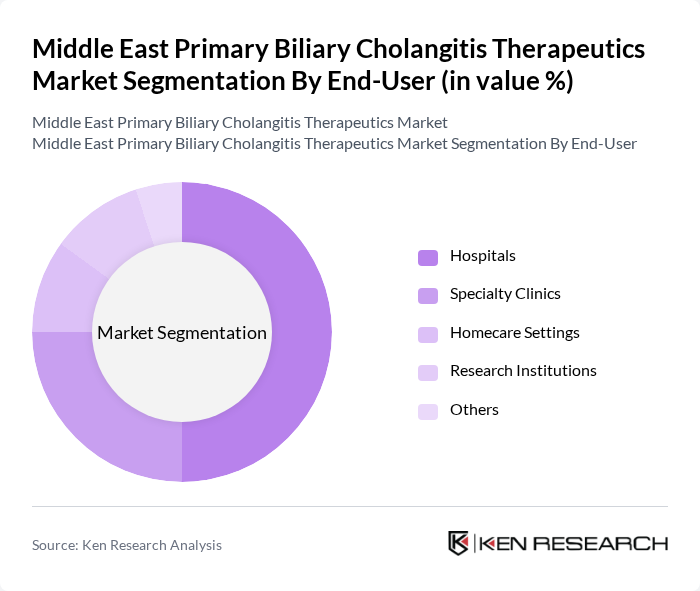
By End-User:The end-user segmentation includes Hospitals, Specialty Clinics, Homecare Settings, Research Institutions, and Others. Hospitals are the dominant end-user segment, primarily due to their capacity to provide comprehensive care and access to advanced diagnostic and therapeutic options. The increasing number of patients being diagnosed with PBC is leading to higher hospital admissions, thereby boosting the demand for therapeutics.
Middle East Primary Biliary Cholangitis Therapeutics Market Competitive Landscape
The Middle East Primary Biliary Cholangitis Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Intercept Pharmaceuticals, Zydus Cadila, GSK (GlaxoSmithKline), Novartis, AbbVie, Sanofi, Genfit, Mirum Pharmaceuticals, Mallinckrodt Pharmaceuticals, Amgen, Pfizer, Takeda Pharmaceutical Company, Eiger BioPharmaceuticals, Horizon Therapeutics, Sobi (Swedish Orphan Biovitrum) contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Primary Biliary Cholangitis Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Primary Biliary Cholangitis:The Middle East has witnessed a notable rise in the prevalence of Primary Biliary Cholangitis (PBC), with estimates indicating that approximately 22,000 cases are diagnosed annually. This increase is attributed to factors such as lifestyle changes and environmental influences. The World Health Organization reported that liver diseases, including PBC, are responsible for 2.5% of total deaths in the region, highlighting the urgent need for effective therapeutic interventions.
- Advancements in Therapeutic Options:Recent advancements in therapeutic options for PBC have significantly improved patient outcomes. For instance, the introduction of novel medications such as obeticholic acid has shown a 50% improvement in liver function tests among patients. Additionally, the Middle East is experiencing a surge in clinical trials, with over 18 ongoing studies aimed at evaluating new therapies, which is expected to enhance treatment accessibility and efficacy in the coming years.
- Rising Awareness and Diagnosis Rates:Increased awareness of PBC among healthcare professionals and the general public has led to higher diagnosis rates. In future, the number of diagnosed cases is expected to rise by 35% compared to previous years, driven by educational campaigns and improved screening protocols. The region's healthcare systems are investing in training programs, resulting in a 30% increase in the number of specialists capable of diagnosing and managing PBC, further supporting market growth.
Market Challenges
- High Cost of Treatment:The cost of PBC treatment remains a significant barrier in the Middle East, with annual treatment expenses averaging around $32,000 per patient. This high financial burden limits access to necessary therapies, particularly in lower-income populations. The economic impact of PBC is substantial, with the total annual healthcare expenditure for liver diseases in the region estimated at $1.3 billion, underscoring the need for more affordable treatment options.
- Limited Access to Healthcare Facilities:Access to specialized healthcare facilities for PBC treatment is limited in many Middle Eastern countries. For example, only 42% of patients in rural areas have access to liver specialists, which hampers timely diagnosis and treatment. The World Bank reported that healthcare infrastructure in the region is underdeveloped, with only 2.7 hospital beds per 1,000 people, exacerbating the challenges faced by PBC patients seeking care.
Middle East Primary Biliary Cholangitis Therapeutics Market Future Outlook
The future of the Middle East Primary Biliary Cholangitis therapeutics market appears promising, driven by ongoing advancements in treatment options and increased healthcare investments. As governments prioritize healthcare infrastructure, the region is expected to see a rise in specialized clinics and telemedicine services, enhancing patient access. Furthermore, the integration of digital health technologies will facilitate better patient management and monitoring, ultimately improving treatment outcomes and patient satisfaction in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Middle East is investing heavily in healthcare infrastructure, with over $55 billion allocated for new hospitals and clinics by 2025. This expansion will improve access to PBC treatments and enhance patient care, creating a favorable environment for market growth and innovation in therapeutic options.
- Development of Novel Therapies:There is a significant opportunity for the development of novel therapies targeting PBC, with over 25 new drug candidates currently in the pipeline. Collaborations between pharmaceutical companies and research institutions are expected to accelerate the introduction of innovative treatments, addressing unmet medical needs and improving patient outcomes in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Ursodeoxycholic Acid Obeticholic Acid Fibrates Immunosuppressants Others |
| By End-User | Hospitals Specialty Clinics Homecare Settings Research Institutions Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Direct Sales Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Patient Demographics | Age Group (Adults, Elderly) Gender (Male, Female) Socioeconomic Status (Low, Middle, High) Others |
| By Treatment Stage | Early Stage Advanced Stage Palliative Care Others |
| By Research and Development Focus | Clinical Trials Drug Development Patient Registries Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration of
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Health Insurance Companies
Clinical Research Organizations
Pharmaceutical Distributors
Players Mentioned in the Report:
Intercept Pharmaceuticals
Zydus Cadila
GSK (GlaxoSmithKline)
Novartis
AbbVie
Sanofi
Genfit
Mirum Pharmaceuticals
Mallinckrodt Pharmaceuticals
Amgen
Pfizer
Takeda Pharmaceutical Company
Eiger BioPharmaceuticals
Horizon Therapeutics
Sobi (Swedish Orphan Biovitrum)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Primary Biliary Cholangitis Therapeutics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Primary Biliary Cholangitis Therapeutics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Primary Biliary Cholangitis Therapeutics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Primary Biliary Cholangitis
3.1.2 Advancements in therapeutic options
3.1.3 Rising awareness and diagnosis rates
3.1.4 Supportive government initiatives
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited access to healthcare facilities
3.2.3 Regulatory hurdles
3.2.4 Lack of specialized healthcare professionals
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Development of novel therapies
3.3.3 Collaborations with research institutions
3.3.4 Increasing investment in healthcare R&D
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth in telemedicine services
3.4.3 Rising demand for patient-centric care
3.4.4 Integration of digital health technologies
3.5 Government Regulation
3.5.1 Drug approval processes
3.5.2 Pricing regulations
3.5.3 Reimbursement policies
3.5.4 Clinical trial regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Primary Biliary Cholangitis Therapeutics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Primary Biliary Cholangitis Therapeutics Market Segmentation
8.1 By Type
8.1.1 Ursodeoxycholic Acid
8.1.2 Obeticholic Acid
8.1.3 Fibrates
8.1.4 Immunosuppressants
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty Clinics
8.2.3 Homecare Settings
8.2.4 Research Institutions
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Hospital Pharmacies
8.3.2 Retail Pharmacies
8.3.3 Online Pharmacies
8.3.4 Direct Sales
8.3.5 Others
8.4 By Region
8.4.1 GCC Countries
8.4.2 Levant Region
8.4.3 North Africa
8.4.4 Others
8.5 By Patient Demographics
8.5.1 Age Group (Adults, Elderly)
8.5.2 Gender (Male, Female)
8.5.3 Socioeconomic Status (Low, Middle, High)
8.5.4 Others
8.6 By Treatment Stage
8.6.1 Early Stage
8.6.2 Advanced Stage
8.6.3 Palliative Care
8.6.4 Others
8.7 By Research and Development Focus
8.7.1 Clinical Trials
8.7.2 Drug Development
8.7.3 Patient Registries
8.7.4 Others
9. Middle East Primary Biliary Cholangitis Therapeutics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Diversification Index
9.2.8 Research & Development Investment
9.2.9 Distribution Efficiency
9.2.10 Brand Recognition Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Intercept Pharmaceuticals
9.5.2 Zydus Cadila
9.5.3 GSK (GlaxoSmithKline)
9.5.4 Novartis
9.5.5 AbbVie
9.5.6 Sanofi
9.5.7 Genfit
9.5.8 Mirum Pharmaceuticals
9.5.9 Mallinckrodt Pharmaceuticals
9.5.10 Amgen
9.5.11 Pfizer
9.5.12 Takeda Pharmaceutical Company
9.5.13 Eiger BioPharmaceuticals
9.5.14 Horizon Therapeutics
9.5.15 Sobi (Swedish Orphan Biovitrum)
10. Middle East Primary Biliary Cholangitis Therapeutics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Funding Sources
10.2.3 Project Prioritization
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Treatment Accessibility
10.3.2 Affordability Issues
10.3.3 Quality of Care
10.3.4 Information Gaps
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Technology Acceptance
10.4.4 Support Systems
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Scalability Potential
10.5.4 Long-term Sustainability
11. Middle East Primary Biliary Cholangitis Therapeutics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Competitive Advantage Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Channels
2.5 Marketing Budget Allocation
2.6 Performance Metrics
2.7 Feedback Mechanisms
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Partnership Opportunities
3.6 Logistics and Supply Chain Management
3.7 Distribution Cost Analysis
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Price Sensitivity Assessment
4.6 Discounting Strategies
4.7 Pricing Model Development
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Product Development Opportunities
5.4 Service Gaps Exploration
5.5 Market Entry Barriers
5.6 Customer Pain Points
5.7 Future Trends Anticipation
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Engagement Strategies
6.4 Feedback Collection Methods
6.5 Relationship Management Tools
6.6 Customer Retention Strategies
6.7 Community Building Initiatives
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
7.5 Competitive Differentiation
7.6 Value Delivery Mechanisms
7.7 Long-term Value Creation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Market Research Activities
8.5 Training and Development
8.6 Performance Monitoring
8.7 Stakeholder Engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Control Considerations
10.7 Partnership Opportunities
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Funding Sources
11.4 Financial Projections
11.5 Budget Allocation
11.6 Cost Management Strategies
11.7 Investment Return Expectations
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
12.3 Control Mechanisms
12.4 Partnership Evaluation
12.5 Long-term Sustainability Considerations
12.6 Performance Monitoring
12.7 Exit Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Profit Margin Projections
13.4 Revenue Growth Forecasts
13.5 Cost Structure Analysis
13.6 Financial Health Indicators
13.7 Investment Viability Assessment
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Collaboration Opportunities
14.6 Partnership Evaluation Criteria
14.7 Long-term Relationship Management
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
15.2.3 Performance Evaluation
15.2.4 Adjustment Strategies
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and regulatory bodies in the Middle East
- Review of scientific literature on Primary Biliary Cholangitis (PBC) and its treatment options
- Examination of market reports and industry publications focusing on liver diseases and therapeutics
Primary Research
- Interviews with hepatologists and gastroenterologists specializing in liver diseases
- Surveys with pharmaceutical companies involved in PBC drug development
- Focus groups with patients diagnosed with PBC to understand treatment experiences and preferences
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from clinical trials, market trends, and patient feedback
- Sanity checks through consultations with industry experts and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on prevalence rates of PBC in the Middle East
- Analysis of healthcare expenditure trends related to liver disease treatments
- Incorporation of government health initiatives and funding for liver disease research
Bottom-up Modeling
- Data collection from leading pharmaceutical companies on sales figures for PBC therapeutics
- Estimation of treatment costs based on pricing strategies of existing therapies
- Volume estimates derived from patient population data and treatment adherence rates
Forecasting & Scenario Analysis
- Multi-factor analysis considering demographic changes and healthcare access improvements
- Scenario modeling based on potential new drug approvals and market entry of generics
- Baseline, optimistic, and pessimistic forecasts for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hepatology Clinics | 100 | Hepatologists, Clinic Managers |
| Pharmaceutical Companies | 80 | Product Managers, Market Access Specialists |
| Patient Advocacy Groups | 60 | Patient Representatives, Health Educators |
| Healthcare Payers | 70 | Insurance Analysts, Policy Makers |
| Research Institutions | 50 | Clinical Researchers, Epidemiologists |
Frequently Asked Questions
What is the current value of the Middle East Primary Biliary Cholangitis Therapeutics Market?
The Middle East Primary Biliary Cholangitis Therapeutics Market is valued at approximately USD 0.85 billion, reflecting a five-year historical analysis that highlights growth driven by increased healthcare spending and awareness of autoimmune liver conditions.