Region:Middle East
Author(s):Dev
Product Code:KRAC1293
Pages:87
Published On:December 2025
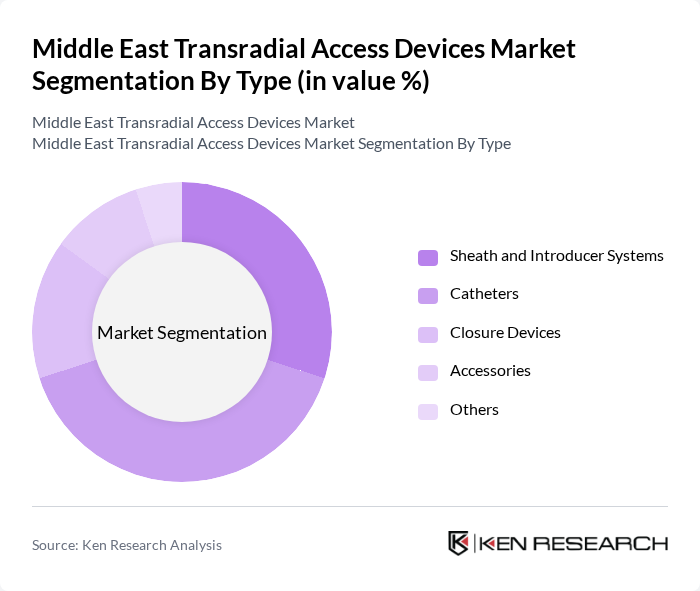
By Type:The transradial access devices market is segmented into various types, including sheath and introducer systems, catheters, closure devices, accessories, and others. Among these, catheters are the leading subsegment due to their critical role in facilitating minimally invasive procedures. The demand for advanced catheter technologies, which enhance procedural efficiency and patient safety, is driving this segment's growth. Sheath and introducer systems also hold significant market share, as they are essential for the successful execution of transradial procedures.
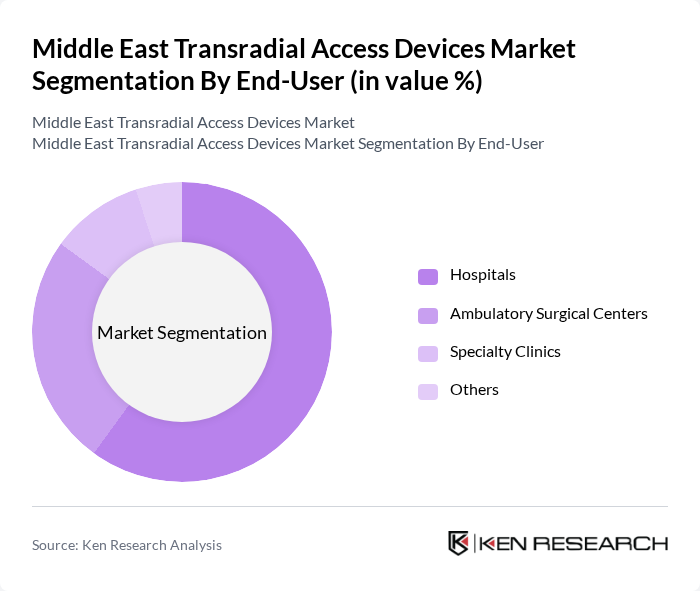
By End-User:The market is categorized based on end-users, including hospitals, ambulatory surgical centers, specialty clinics, and others. Hospitals dominate this segment due to their comprehensive facilities and higher patient volumes, which necessitate the use of transradial access devices for various cardiovascular procedures. Ambulatory surgical centers are also gaining traction as they offer cost-effective and efficient alternatives for outpatient procedures, contributing to the overall market growth.
The Middle East Transradial Access Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Boston Scientific, Abbott Laboratories, Terumo Corporation, Johnson & Johnson, B. Braun Melsungen AG, Cardinal Health, Cook Medical, Merit Medical Systems, Philips Healthcare, Siemens Healthineers, GE Healthcare, Stryker Corporation, Edwards Lifesciences, Asahi Intecc contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East transradial access devices market appears promising, driven by ongoing healthcare infrastructure modernization and a shift towards minimally invasive procedures. As governments continue to invest in healthcare through initiatives like Saudi Vision 2030, the demand for advanced medical technologies is expected to rise. Additionally, the integration of digital health technologies and telemedicine will likely enhance patient outcomes and streamline procedures, further supporting market growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Sheath and Introducer Systems Catheters Closure Devices Accessories Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Application | Diagnostic Procedures Interventional Procedures Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender Others |
| By Technology | Conventional Techniques Advanced Techniques Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 150 | Interventional Cardiologists, Cardiology Department Heads |
| Medical Device Distributors | 100 | Sales Managers, Product Specialists |
| Healthcare Procurement Offices | 80 | Procurement Officers, Supply Chain Managers |
| Clinical Research Organizations | 70 | Clinical Research Coordinators, Medical Affairs Managers |
| Regulatory Bodies and Associations | 60 | Regulatory Affairs Specialists, Policy Advisors |
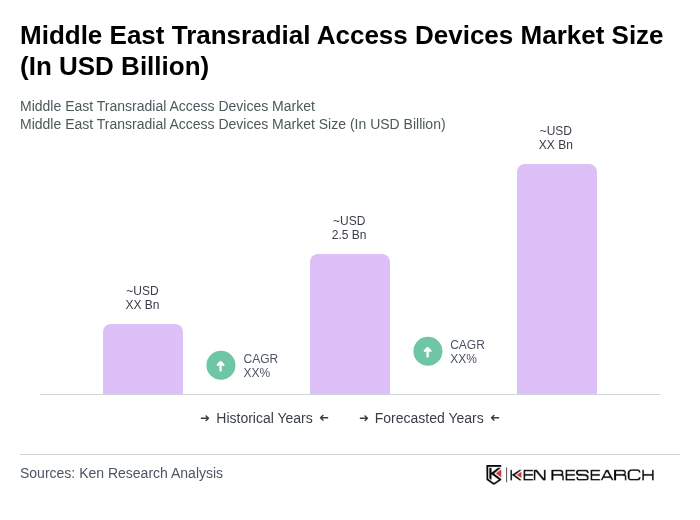
The Middle East Transradial Access Devices Market is valued at approximately USD 2.5 billion, reflecting a robust growth trajectory driven by the increasing prevalence of cardiovascular diseases and the adoption of minimally invasive procedures.