Region:Global
Author(s):Shubham
Product Code:KRAD3057
Pages:92
Published On:January 2026
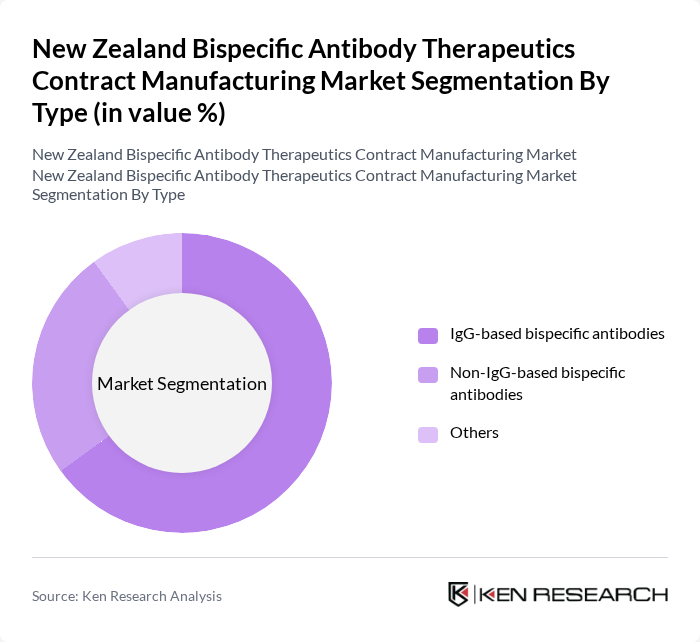
By Type:The market is segmented into three main types: IgG-based bispecific antibodies, Non-IgG-based bispecific antibodies, and Others. IgG-based bispecific antibodies are currently leading the market due to their established efficacy and safety profiles, making them the preferred choice for therapeutic applications. Non-IgG-based bispecific antibodies are gaining traction, particularly in niche therapeutic areas, while the 'Others' category includes emerging technologies that are still in developmental stages.
By End-User:The end-user segmentation includes Pharmaceutical companies, Biotechnology firms, Research institutions, and Others. Pharmaceutical companies dominate the market as they have the resources and infrastructure to develop and manufacture bispecific antibodies at scale. Biotechnology firms are also significant players, focusing on innovative therapies, while research institutions contribute to the development of new technologies and applications in this field.

The New Zealand Bispecific Antibody Therapeutics Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fisher BioServices, GenScript, Lonza Group, WuXi AppTec, Catalent, Samsung Biologics, AbbVie Contract Manufacturing, Boehringer Ingelheim, Merck KGaA, Amgen, Takeda Pharmaceutical Company, Pfizer, Roche, Novartis, Regeneron Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space.
The future of the bispecific antibody therapeutics contract manufacturing market in New Zealand appears promising, driven by ongoing advancements in biotechnology and a growing emphasis on personalized medicine. As the healthcare landscape evolves, manufacturers are likely to adopt modular and continuous manufacturing processes to enhance efficiency. Furthermore, the integration of artificial intelligence in drug development is expected to streamline R&D efforts, enabling faster and more cost-effective production of innovative therapies tailored to patient needs in future.
| Segment | Sub-Segments |
|---|---|
| By Type | IgG-based bispecific antibodies Non-IgG-based bispecific antibodies Others |
| By End-User | Pharmaceutical companies Biotechnology firms Research institutions Others |
| By Therapeutic Area | Oncology Autoimmune diseases Infectious diseases Others |
| By Manufacturing Process | Cell culture-based production Protein purification techniques Others |
| By Distribution Channel | Direct sales Distributors Online platforms Others |
| By Region | North Island South Island |
| By Policy Support | Government grants Tax incentives Research funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical R&D Departments | 45 | R&D Directors, Lead Scientists |
| Contract Manufacturing Organizations | 35 | Operations Managers, Quality Assurance Heads |
| Regulatory Affairs in Biotech | 30 | Regulatory Affairs Managers, Compliance Officers |
| Healthcare Providers and Institutions | 25 | Oncologists, Immunologists |
| Market Analysts and Consultants | 15 | Market Research Analysts, Industry Consultants |
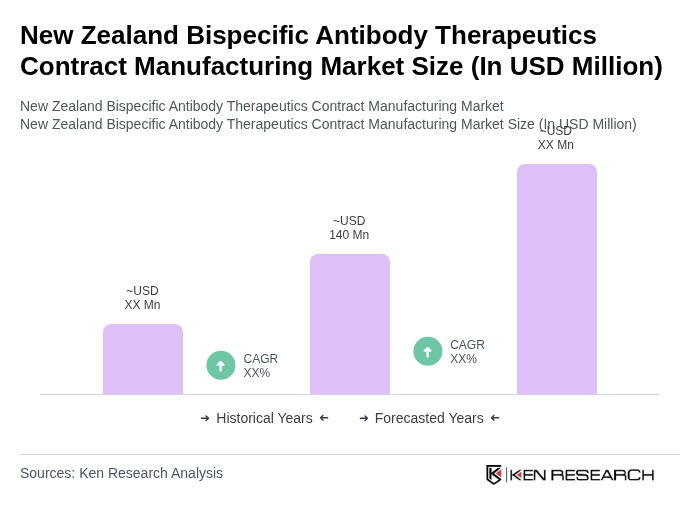
The New Zealand Bispecific Antibody Therapeutics Contract Manufacturing Market is valued at approximately USD 140 million, reflecting a five-year historical analysis driven by chronic disease prevalence, biotechnology advancements, and personalized medicine demand.