Region:Global
Author(s):Geetanshi
Product Code:KRAE2030
Pages:99
Published On:February 2026
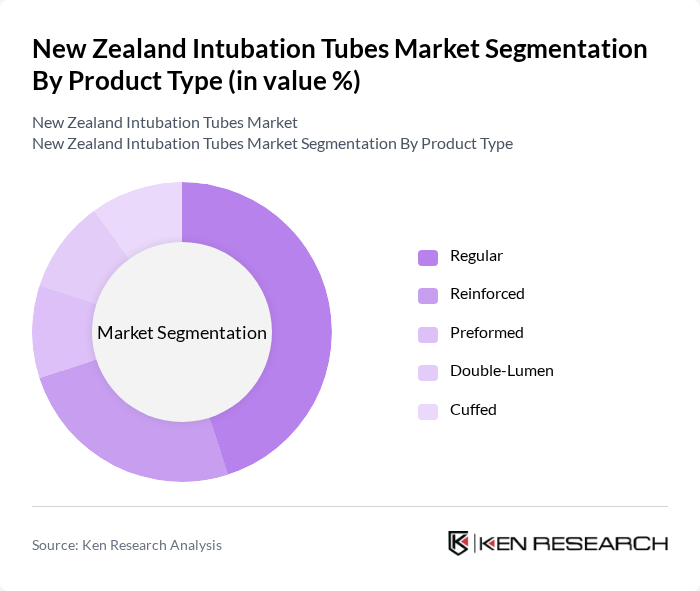
By Product Type:The product type segmentation includes Regular, Reinforced, Preformed, Double-Lumen, and Cuffed intubation tubes. Among these, Regular intubation tubes dominate the market due to their widespread use in various medical settings. Their versatility and cost-effectiveness make them the preferred choice for healthcare providers. Reinforced tubes are also gaining traction, particularly in cases requiring increased stability and resistance to kinking.
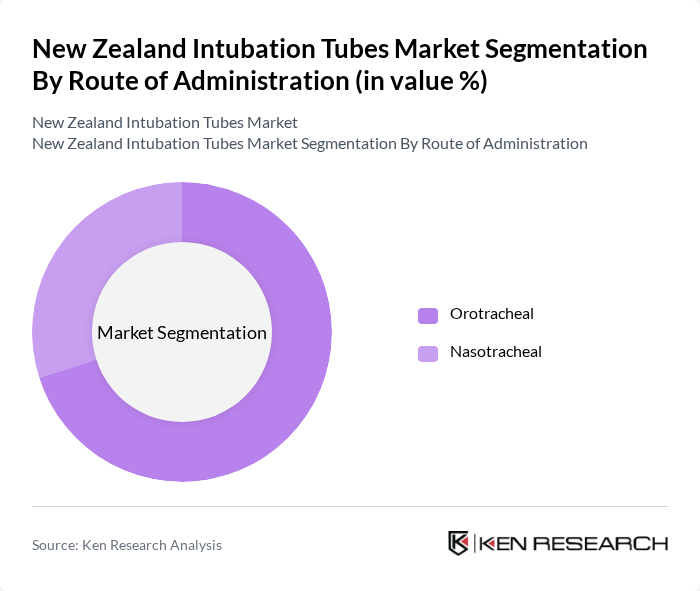
By Route of Administration:The route of administration segmentation includes Orotracheal and Nasotracheal intubation. Orotracheal intubation is the leading method due to its effectiveness in securing the airway in emergency situations and during surgeries. It is preferred by healthcare professionals for its ease of use and reliability. Nasotracheal intubation, while less common, is utilized in specific cases where oro-tracheal access is not feasible.
The New Zealand Intubation Tubes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Teleflex Incorporated, Smiths Medical, Boston Scientific, ConvaTec, Halyard Health, Vyaire Medical, Cook Medical, Becton, Dickinson and Company, Ambu A/S, Medline Industries, Airway Management Solutions, Intersurgical, Flexicare Medical, Armstrong Medical contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand intubation tubes market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centered care. As healthcare providers continue to prioritize safety and efficiency, the integration of smart technology into intubation devices is expected to gain traction. Additionally, the expansion of healthcare infrastructure, particularly in rural areas, will likely enhance access to advanced airway management solutions, further supporting market growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Regular Reinforced Preformed Double-Lumen Cuffed |
| By Route of Administration | Orotracheal Nasotracheal |
| By End-User | Hospitals & Clinics Ambulatory Surgical Centers Others |
| By Application | Emergency Treatment Therapy Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Anesthesia Departments | 45 | Anesthesiologists, Nurse Anesthetists |
| Emergency Medical Services | 40 | Paramedics, Emergency Room Physicians |
| Medical Device Distributors | 40 | Sales Managers, Product Specialists |
| Healthcare Procurement Departments | 45 | Procurement Officers, Supply Chain Managers |
| Clinical Research Organizations | 40 | Clinical Researchers, Regulatory Affairs Specialists |
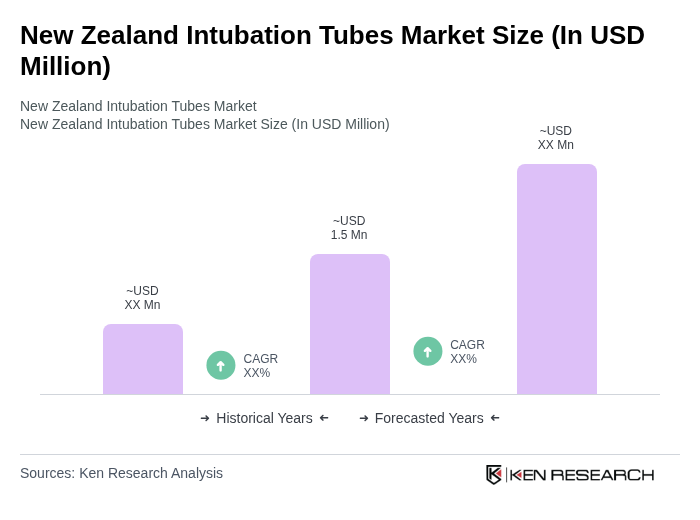
The New Zealand Intubation Tubes Market is valued at approximately USD 1.5 million, reflecting a five-year historical analysis. This growth is driven by factors such as the increasing prevalence of respiratory diseases and advancements in medical technology.