Region:Middle East
Author(s):Geetanshi
Product Code:KRAA6163
Pages:80
Published On:January 2026
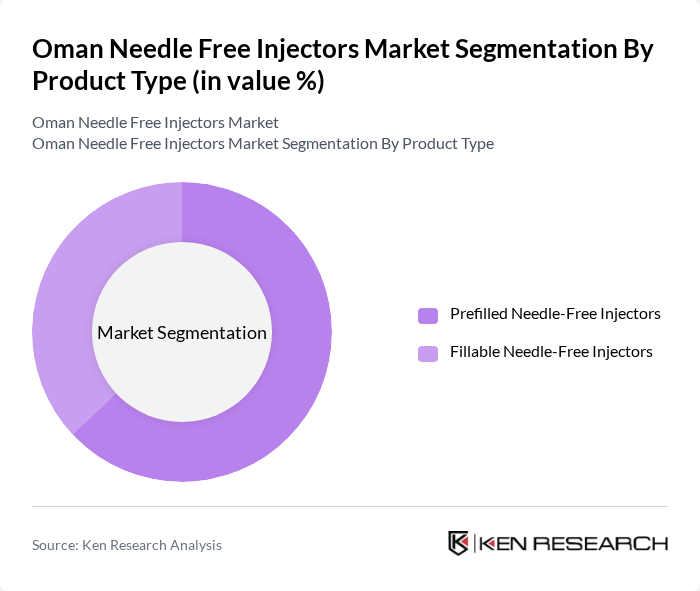
By Product Type:The product type segmentation includes prefilled needle-free injectors and fillable needle-free injectors. Prefilled needle-free injectors dominate the market, accounting for approximately 63% of the segment, driven by their convenience, dosage accuracy, and reduced preparation time in clinical and home care environments. These formats minimize contamination risks and ensure consistent drug delivery, making them ideal for vaccines, insulin, and biologics. Fillable needle-free injectors remain popular among healthcare professionals who prefer customizable dosing options for various medications.
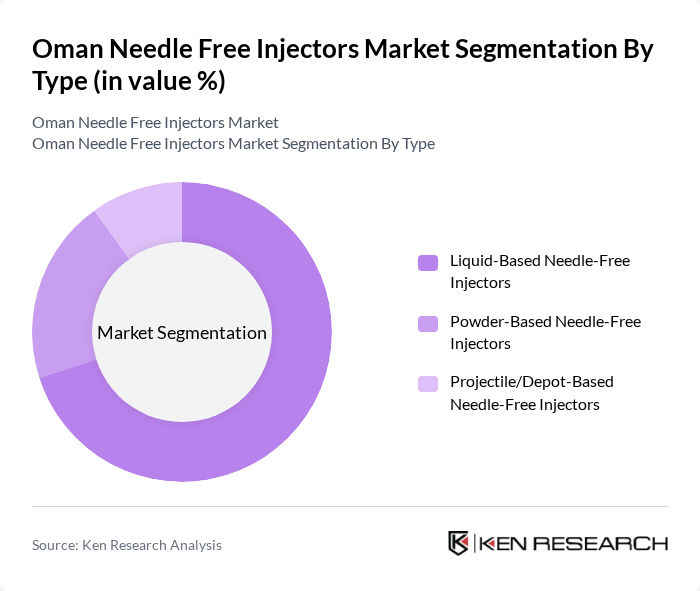
By Type:The type segmentation encompasses liquid-based needle-free injectors, powder-based needle-free injectors, and projectile/depot-based needle-free injectors. Liquid-based injectors dominate the market due to their widespread application in vaccine delivery and insulin administration. Powder-based injectors are emerging as a viable alternative for certain medications, while projectile/depot-based injectors are gaining attention for their potential in long-acting drug delivery.
The Oman Needle Free Injectors Market is characterized by a dynamic mix of regional and international players. Leading participants such as PharmaJet, Zogenix, MedImmune, West Pharmaceutical Services, Injex Pharma, Bioject Medical Technologies, AptarGroup, Vaxart, Enable Injections, Becton, Dickinson and Company, Eitan Medical, Acelity, Terumo Corporation, Medtronic, and 3M contribute to innovation, geographic expansion, and service delivery in this space.
The future of the needle-free injectors market in Oman appears promising, driven by increasing healthcare investments and a growing emphasis on patient comfort. As the healthcare infrastructure expands, more facilities are likely to adopt innovative drug delivery technologies. Additionally, the integration of digital health solutions will enhance the efficiency of needle-free injectors, making them more appealing to both healthcare providers and patients. This trend is expected to foster a more robust market environment in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Prefilled Needle-Free Injectors Fillable Needle-Free Injectors |
| By Type | Liquid-Based Needle-Free Injectors Powder-Based Needle-Free Injectors Projectile/Depot-Based Needle-Free Injectors |
| By Technology | Jet-Based Needle-Free Injectors Spring-Based Needle-Free Injectors Gas-Powered Needle-Free Injectors Others |
| By Site of Delivery | Subcutaneous Injectors Intramuscular Injectors Intradermal Injectors |
| By Usability | Reusable Needle-Free Injectors Disposable Needle-Free Injectors |
| By Application | Vaccine Delivery Insulin Delivery Pain Management Others |
| By End-User | Hospitals Clinics Home Care Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Doctors, Nurses, Pharmacists |
| Pharmaceutical Companies | 45 | Product Managers, R&D Directors |
| Patients Using Needle-Free Injectors | 80 | Chronic Disease Patients, General Public |
| Healthcare Administrators | 65 | Hospital Administrators, Procurement Officers |
| Regulatory Bodies | 40 | Health Policy Makers, Compliance Officers |
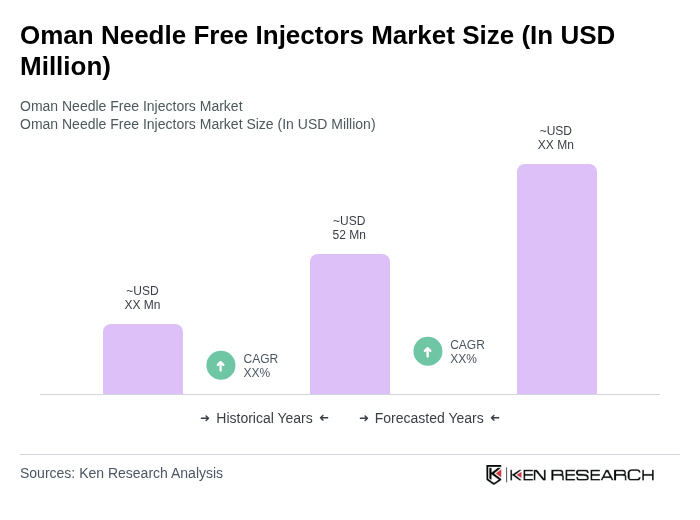
The Oman Needle Free Injectors Market is valued at approximately USD 52 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases and advancements in needle-free technology that enhance patient comfort and compliance.