Region:Middle East
Author(s):Geetanshi
Product Code:KRAC3094
Pages:87
Published On:October 2025
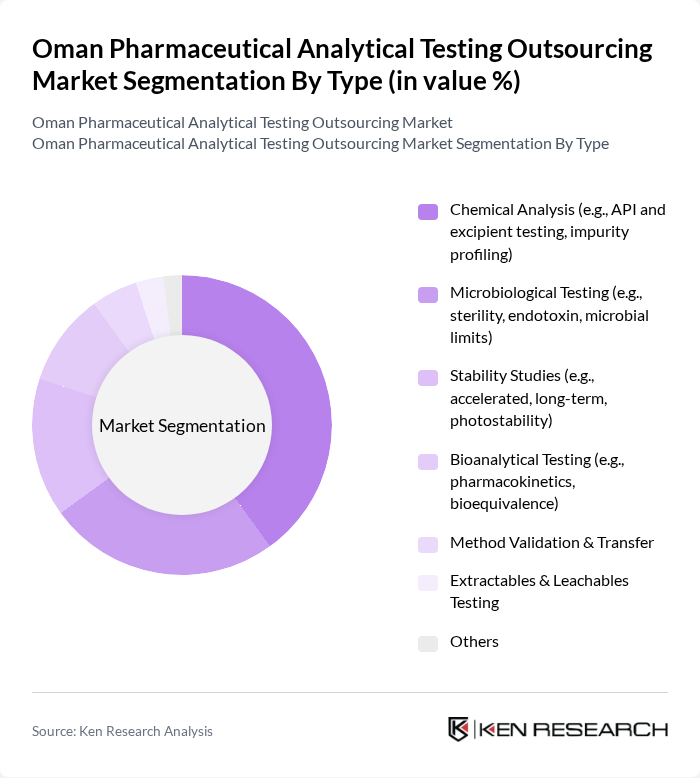
By Type:The market is segmented into various types of analytical testing services, including Chemical Analysis, Microbiological Testing, Stability Studies, Bioanalytical Testing, Method Validation & Transfer, Extractables & Leachables Testing, and Others. Among these, Chemical Analysis remains the leading sub-segment due to its critical role in ensuring the quality and safety of active pharmaceutical ingredients (APIs) and excipients. The increasing complexity of drug formulations, stricter impurity profiling standards, and the need for advanced analytical instrumentation further drive the demand for chemical analysis services .
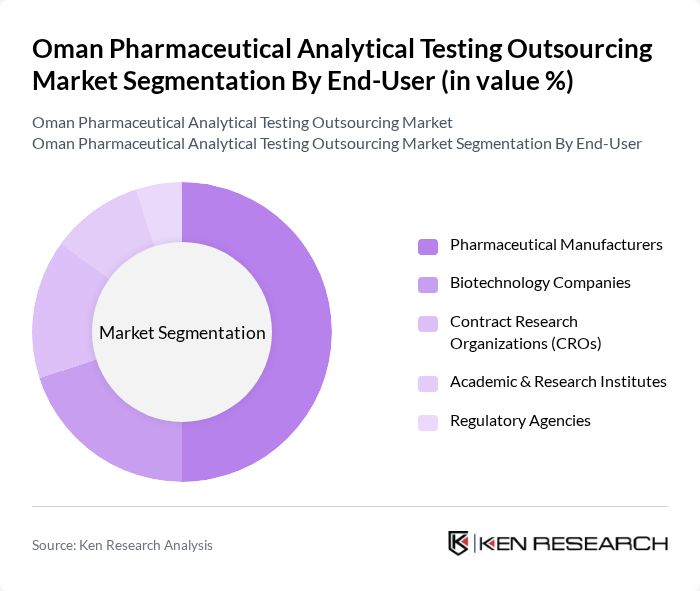
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Biotechnology Companies, Contract Research Organizations (CROs), Academic & Research Institutes, and Regulatory Agencies. Pharmaceutical Manufacturers are the dominant end-users, driven by the need for compliance with stringent regulatory standards and the increasing complexity of drug formulations. Their reliance on outsourcing analytical testing services to ensure product quality and safety significantly contributes to the market's growth. Biotechnology companies and CROs are also increasing their share, reflecting the rise in biologics and biosimilars development and clinical trial activity in Oman .
The Oman Pharmaceutical Analytical Testing Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oman Pharmaceutical Products Co. LLC, Gulf Pharmaceutical Industries (Julphar), Muscat Pharmacy & Stores LLC, Dhofar Pharmaceutical Industries LLC, Al Nahda Pharmaceuticals LLC, United Pharmaceutical Manufacturing Co. LLC, Oman Medical Supplies & Services Co. LLC, Al Ameen Pharmaceutical LLC, Al Mufeed Pharmaceuticals LLC, Al Muna Pharmaceuticals LLC, National Pharmaceutical Industries Co. LLC, Pharmazone LLC, Aster DM Healthcare, Eurofins Scientific (regional operations), SGS Oman (SGS S.A. regional analytical services) contribute to innovation, geographic expansion, and service delivery in this space .
The future of the Oman pharmaceutical analytical testing outsourcing market appears promising, driven by ongoing advancements in technology and increasing regulatory scrutiny. As the demand for personalized medicine grows, pharmaceutical companies are likely to seek innovative testing solutions that can accommodate complex drug formulations. Additionally, the trend towards automation in testing processes is expected to enhance efficiency and reduce turnaround times, making outsourcing an attractive option for local manufacturers.
| Segment | Sub-Segments |
|---|---|
| By Type | Chemical Analysis (e.g., API and excipient testing, impurity profiling) Microbiological Testing (e.g., sterility, endotoxin, microbial limits) Stability Studies (e.g., accelerated, long-term, photostability) Bioanalytical Testing (e.g., pharmacokinetics, bioequivalence) Method Validation & Transfer Extractables & Leachables Testing Others |
| By End-User | Pharmaceutical Manufacturers Biotechnology Companies Contract Research Organizations (CROs) Academic & Research Institutes Regulatory Agencies |
| By Service Type | Analytical Testing Services Method Development & Validation Stability Testing Services Regulatory Compliance & Consulting Quality Control & Release Testing |
| By Sample Type | Raw Materials Intermediates Finished Products Packaging Materials |
| By Testing Phase | Preclinical Testing Clinical Testing (Phase I-III) Post-Market Surveillance |
| By Geographic Focus | Domestic (Oman) GCC/Regional Market International Market |
| By Pricing Model | Fixed Pricing Variable Pricing Subscription-Based Pricing |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Labs | 60 | Laboratory Managers, Quality Control Analysts |
| Pharmaceutical Manufacturers | 50 | Production Managers, Regulatory Affairs Officers |
| Healthcare Providers | 40 | Pharmacists, Clinical Research Coordinators |
| Regulatory Bodies | 40 | Compliance Officers, Policy Makers |
| Industry Associations | 40 | Executive Directors, Research Analysts |
The Oman Pharmaceutical Analytical Testing Outsourcing Market is valued at approximately USD 45 million, reflecting a five-year historical analysis driven by the demand for high-quality pharmaceutical products and stringent regulatory requirements.