Region:Asia
Author(s):Dev
Product Code:KRAC0917
Pages:91
Published On:December 2025
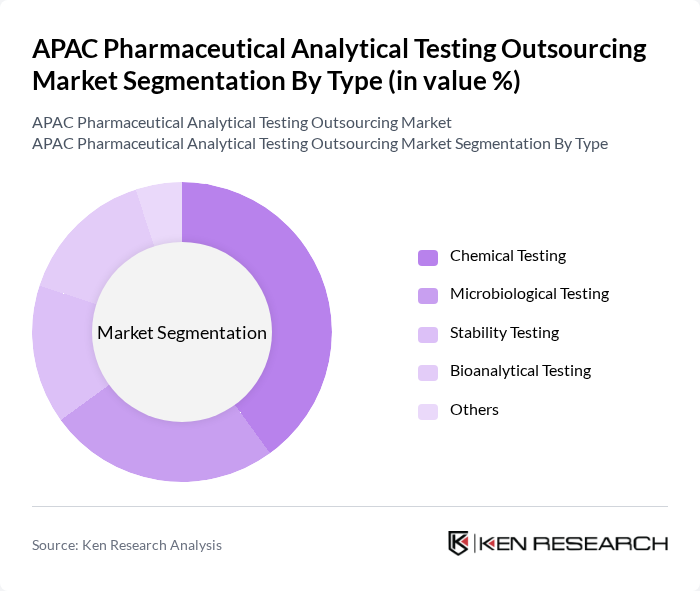
By Type:The market is segmented into various types of analytical testing services, including Chemical Testing, Microbiological Testing, Stability Testing, Bioanalytical Testing, and Others. Among these, Chemical Testing is the most dominant segment due to its critical role in ensuring the safety and efficacy of pharmaceutical products. The increasing demand for chemical analysis in drug formulation and quality control drives this segment's growth.
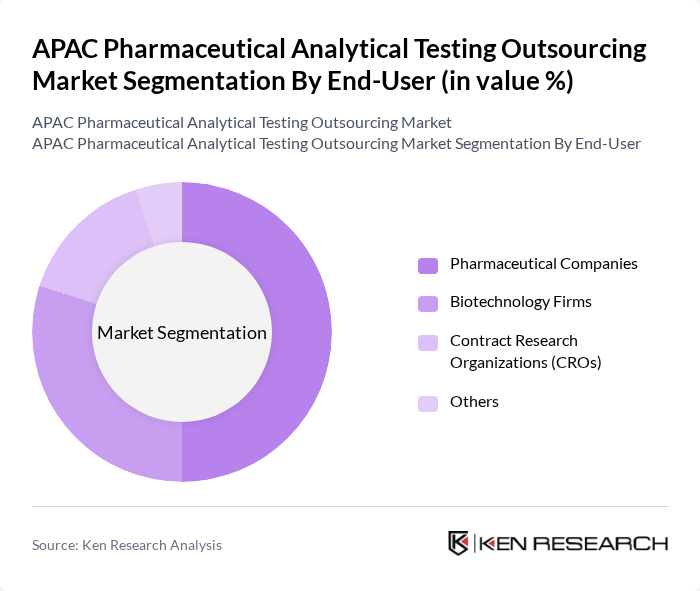
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), and Others. Pharmaceutical Companies dominate this segment, driven by the need for rigorous testing to comply with regulatory standards and ensure product safety. The increasing number of drug approvals and the growing focus on R&D in the pharmaceutical sector further bolster this segment's growth.
The APAC Pharmaceutical Analytical Testing Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as **SGS S.A.**, **Eurofins Scientific**, **Charles River Laboratories**, **Pace Analytical Services**, **Intertek Group plc**, **LabCorp**, **Covance Inc.**, **WuXi AppTec**, **Medpace Holdings, Inc.**, **Bureau Veritas**, **TÜV SÜD**, **Mérieux NutriSciences**, **RSSL**, **Kemin Industries**, **Apex Laboratories** contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC pharmaceutical analytical testing outsourcing market is poised for significant transformation, driven by advancements in digital technologies and a growing emphasis on specialized testing services. The integration of artificial intelligence and automation will enhance testing accuracy and efficiency, while the demand for niche testing services, such as biosafety and cell-based assays, will expand. As infrastructure investments in emerging markets increase, the region will likely become a pivotal hub for pharmaceutical R&D and analytical testing outsourcing, attracting global partnerships and investments.
| Segment | Sub-Segments |
|---|---|
| By Type | **Chemical Testing** **Microbiological Testing** **Stability Testing** **Bioanalytical Testing** **Others** |
| By End-User | **Pharmaceutical Companies** **Biotechnology Firms** **Contract Research Organizations (CROs)** **Others** |
| By Service Type | **Testing Services** **Consulting Services** **Regulatory Services** **Others** |
| By Geography | **China** **India** **Japan** **Australia** **Others** |
| By Analytical Method | **Chromatography** **Spectroscopy** **Electrophoresis** **Others** |
| By Sample Type | **Solid Samples** **Liquid Samples** **Gas Samples** **Others** |
| By Project Size | **Small Scale Projects** **Medium Scale Projects** **Large Scale Projects** **Others** |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Quality Control Testing | 150 | Quality Control Managers, Laboratory Directors |
| Bioanalytical Testing Services | 100 | Clinical Research Coordinators, Regulatory Affairs Specialists |
| Stability Testing Services | 80 | Product Development Managers, Compliance Officers |
| Contract Research Organizations (CROs) | 120 | Business Development Managers, Operations Directors |
| Market Access and Regulatory Consulting | 90 | Market Access Managers, Regulatory Consultants |
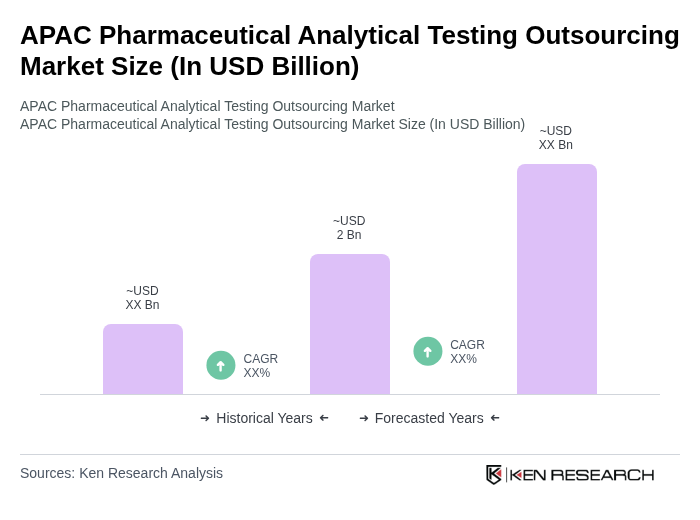
The APAC Pharmaceutical Analytical Testing Outsourcing Market is valued at approximately USD 2 billion, reflecting a five-year historical analysis. This growth is driven by the increasing complexity of drug development and the need for specialized analytical capabilities.