Region:Asia
Author(s):Dev
Product Code:KRAA8201
Pages:98
Published On:November 2025
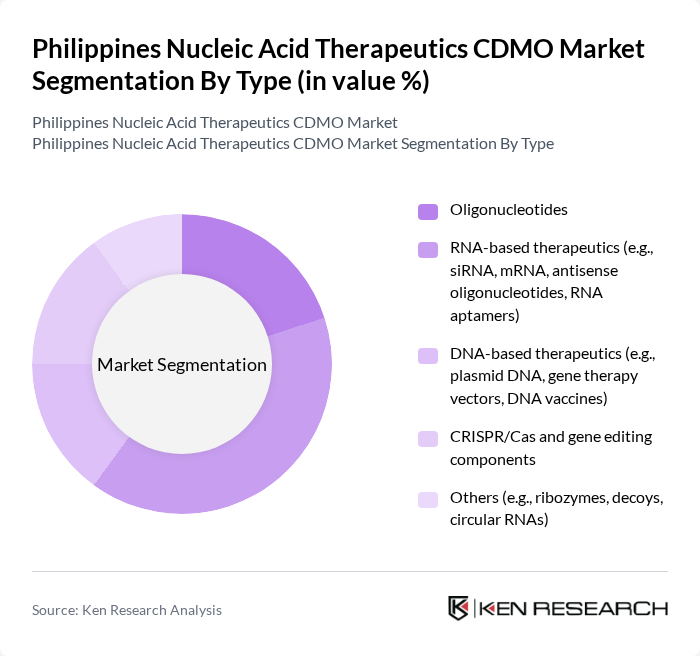
By Type:The market can be segmented into various types of nucleic acid therapeutics, including oligonucleotides, RNA-based therapeutics, DNA-based therapeutics, CRISPR/Cas and gene editing components, and others. Among these,RNA-based therapeuticsare currently leading the market due to their application in vaccine development, gene silencing technologies, and increasing adoption in precision medicine. The demand for mRNA vaccines and siRNA therapies has notably accelerated post-pandemic, positioning RNA-based modalities as the dominant segment .
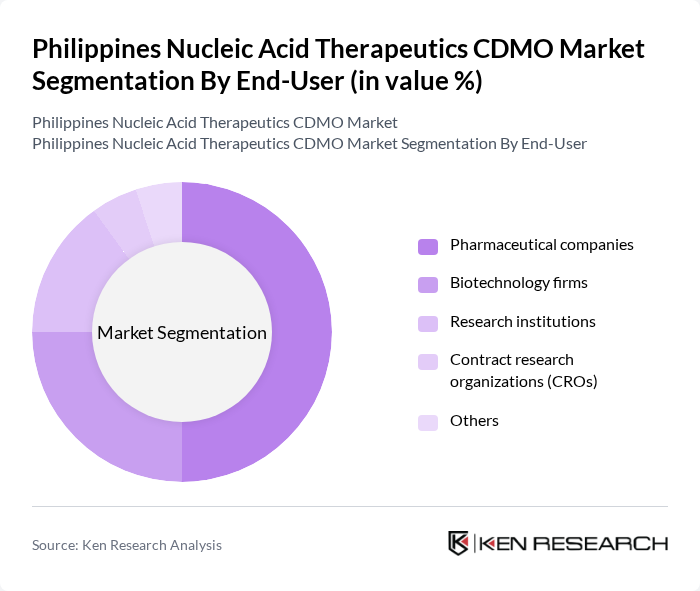
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, research institutions, contract research organizations (CROs), and others.Pharmaceutical companiesare the dominant end-users, driven by their need for innovative therapies and the increasing focus on personalized medicine, which requires advanced nucleic acid-based solutions. The adoption of nucleic acid therapeutics by biotechnology firms and research institutions is also rising, reflecting broader industry engagement in gene and RNA therapy development .
The Philippines Nucleic Acid Therapeutics CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Philippine Genome Center, Genetika+ Inc., Syneos Health Philippines, AGC Biologics, ST Pharm Co., Ltd., BioNTech SE, Novartis Philippines, Merck Sharp & Dohme (MSD) Philippines, Pfizer Philippines, Sanofi Philippines, Roche Philippines, GlaxoSmithKline (GSK) Philippines, AstraZeneca Philippines, Amgen Philippines, Takeda Pharmaceuticals, Thermo Fisher Scientific, Lonza Group, WuXi AppTec, Samsung Biologics, and Regeneron Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space .
The future of the Philippines nucleic acid therapeutics CDMO market appears promising, driven by technological advancements and increased collaboration between public and private sectors. As the demand for personalized medicine grows, CDMOs are likely to expand their capabilities to meet specific patient needs. Additionally, the integration of artificial intelligence in drug development processes is expected to enhance efficiency and reduce time-to-market, positioning the Philippines as a competitive player in the global biotechnology landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Oligonucleotides RNA-based therapeutics (e.g., siRNA, mRNA, antisense oligonucleotides, RNA aptamers) DNA-based therapeutics (e.g., plasmid DNA, gene therapy vectors, DNA vaccines) CRISPR/Cas and gene editing components Others (e.g., ribozymes, decoys, circular RNAs) |
| By End-User | Pharmaceutical companies Biotechnology firms Research institutions Contract research organizations (CROs) Others |
| By Application | Gene therapy Vaccines (including mRNA and DNA vaccines) Diagnostics (molecular diagnostics, companion diagnostics) Cell therapy support Others |
| By Delivery Method | Lipid nanoparticles (LNPs) Viral vectors (AAV, lentivirus, adenovirus, etc.) Electroporation and physical methods Polymer-based carriers Others |
| By Therapeutic Area | Oncology Infectious diseases Genetic disorders Rare diseases Others |
| By Region | Luzon Visayas Mindanao |
| By Funding Source | Government grants Private investments Venture capital International funding agencies Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Nucleic Acid Therapeutics Development | 60 | Biotech Researchers, Product Development Managers |
| Contract Manufacturing Organizations | 50 | Operations Managers, Quality Assurance Leads |
| Regulatory Affairs in Biotech | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Healthcare Providers' Perspectives | 45 | Healthcare Administrators, Clinical Researchers |
| Investment and Funding in Biotech | 40 | Venture Capitalists, Financial Analysts |
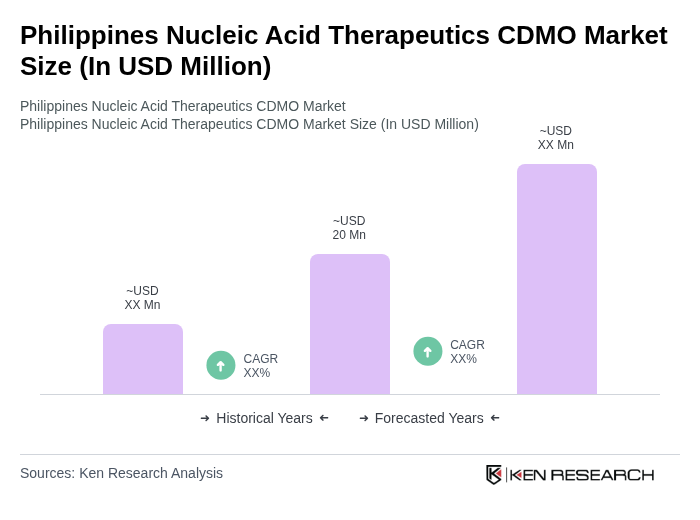
The Philippines Nucleic Acid Therapeutics CDMO Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This growth is driven by advancements in biotechnology and increasing demand for personalized medicine.