Philippines Parp Inhibitor Biomarkers Market Overview
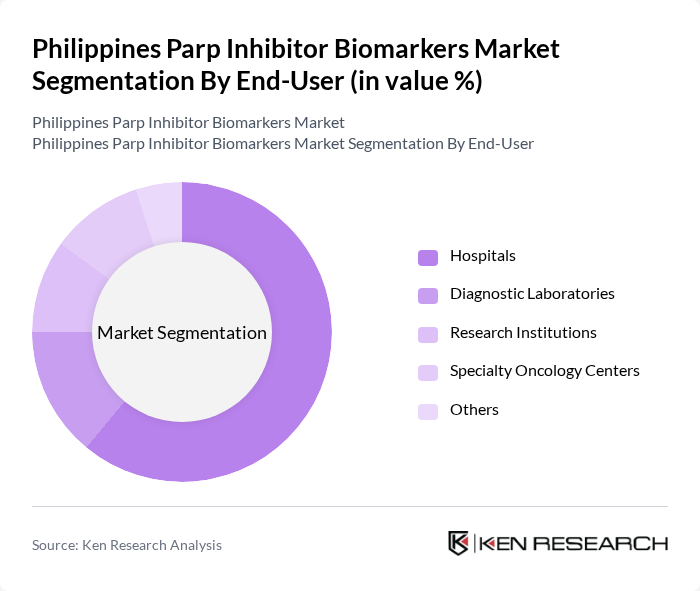
- The Philippines Parp Inhibitor Biomarkers Market is valued at USD 12 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cancer, advancements in personalized medicine, and the rising adoption of targeted therapies. The demand for PARP inhibitors, particularly in oncology, has surged as healthcare providers focus on precision medicine to improve patient outcomes. Recent trends highlight the integration of biomarker-driven therapies, expansion of companion diagnostics, and greater access to genomic testing as key growth drivers in the Philippines market .
- Key cities such asManila, Cebu, and Davaodominate the market due to their advanced healthcare infrastructure, presence of leading hospitals, and research institutions. Manila, being the capital, serves as a hub for medical research and clinical trials, while Cebu and Davao are emerging centers for healthcare services, contributing to the overall growth of the market. The concentration of cancer specialty centers and tertiary hospitals in these cities further accelerates the adoption of PARP inhibitor biomarker testing .
- The Universal Health Care Act (Republic Act No. 11223), issued by the Department of Health in 2019, aims to provide equitable access to healthcare services, including cancer treatments. This regulation mandates the progressive inclusion of innovative therapies, such as PARP inhibitors, in the national health insurance program (PhilHealth), thereby enhancing patient access and driving market growth. The Act requires coverage of essential cancer medicines and diagnostics, and supports the integration of precision oncology into standard care .
Philippines Parp Inhibitor Biomarkers Market Segmentation
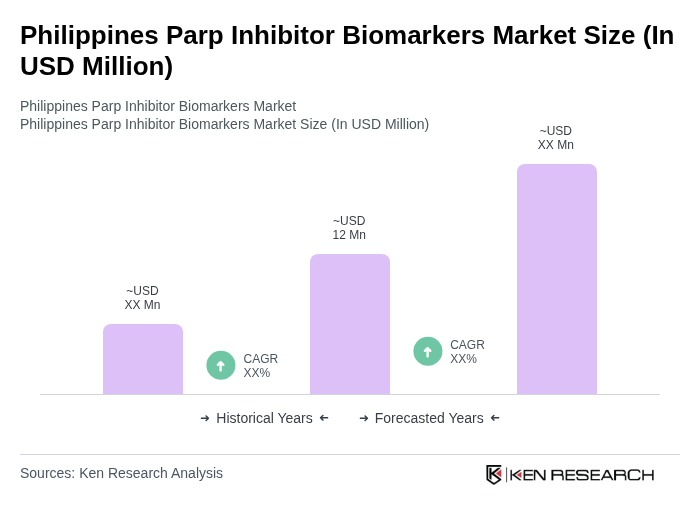
By Type:The market is segmented into various types of PARP inhibitors, including Olaparib, Niraparib, Rucaparib, Talazoparib, and others. Among these,Olaparibhas emerged as the leading sub-segment due to its extensive clinical validation and approval for multiple cancer types, particularly breast and ovarian cancers. The increasing awareness and acceptance of targeted therapies, along with its earlier regulatory approvals and broad clinical use, have further propelled the demand for Olaparib, making it a preferred choice among healthcare providers .
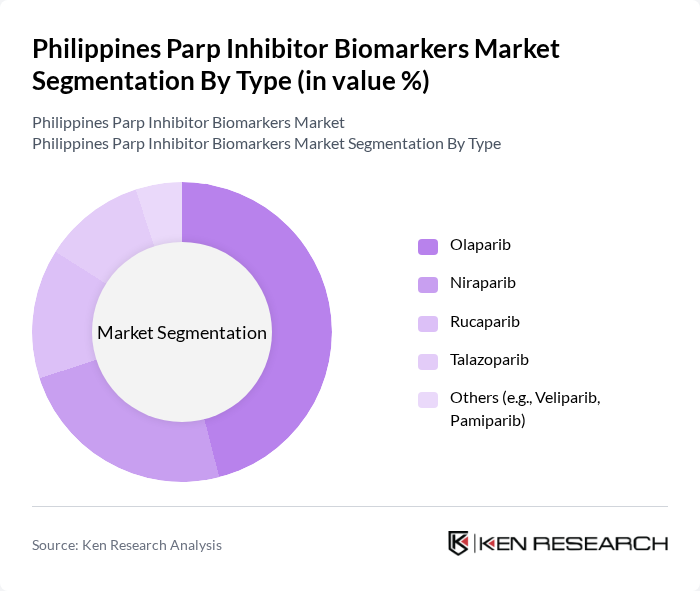
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Specialty Oncology Centers, and others.Hospitalsare the dominant end-user segment, primarily due to their capacity to provide comprehensive cancer care and access to advanced diagnostic tools. The increasing number of cancer patients seeking treatment in hospitals has significantly contributed to the growth of this segment, as they are equipped with the necessary infrastructure and expertise to administer PARP inhibitors effectively. The growing role of multidisciplinary cancer centers and the expansion of molecular pathology services in hospitals further reinforce this trend .
Philippines Parp Inhibitor Biomarkers Market Competitive Landscape
The Philippines Parp Inhibitor Biomarkers Market is characterized by a dynamic mix of regional and international players. Leading participants such as AstraZeneca, Merck & Co., Pfizer, F. Hoffmann-La Roche AG, Clovis Oncology, Tesaro (GSK), Myriad Genetics, Inc., Invitae Corporation, NeoGenomics Laboratories, Inc., Thermo Fisher Scientific Inc., Qiagen N.V., Beckman Coulter, Inc., Euro Diagnostica AB, Svar Life Science AB, BPS Bioscience, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Philippines Parp Inhibitor Biomarkers Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer:The Philippines has seen a significant rise in cancer cases, with the Department of Health reporting approximately 200,000 new cancer cases annually. This alarming statistic underscores the urgent need for effective treatment options, including PARP inhibitors. The growing patient population is driving demand for innovative therapies, as healthcare providers seek to address the increasing burden of cancer, which is projected to become the leading cause of death in the country in future.
- Advancements in Biomarker Research:The Philippines is experiencing rapid advancements in biomarker research, with over 50 ongoing clinical trials focused on cancer biomarkers. These developments are crucial for identifying patients who would benefit from PARP inhibitors, enhancing treatment efficacy. The collaboration between local universities and international research institutions has led to breakthroughs in understanding genetic mutations, which are pivotal in personalizing cancer therapies, thereby fostering a more targeted approach to treatment.
- Rising Demand for Personalized Medicine:The shift towards personalized medicine is evident in the Philippines, where the market for targeted therapies is expected to reach PHP 10 billion in future. Patients and healthcare providers are increasingly recognizing the benefits of tailored treatments, which improve outcomes and reduce side effects. This trend is further supported by the growing availability of genetic testing, enabling healthcare professionals to make informed decisions regarding the use of PARP inhibitors in specific patient populations.
Market Challenges
- High Cost of PARP Inhibitors:The high cost of PARP inhibitors remains a significant barrier to access in the Philippines, with prices ranging from PHP 50,000 to PHP 100,000 per month. This financial burden limits patient access to these critical therapies, particularly in a country where the average monthly income is approximately PHP 25,000. Consequently, many patients may forgo treatment, leading to poorer health outcomes and increased mortality rates among cancer patients.
- Limited Awareness Among Healthcare Professionals:Despite advancements in cancer treatment, there is a notable lack of awareness regarding PARP inhibitors among healthcare professionals in the Philippines. A recent survey indicated that only 30% of oncologists are familiar with the latest PARP inhibitor therapies. This knowledge gap can hinder timely diagnosis and treatment, ultimately affecting patient outcomes and the overall effectiveness of cancer care in the region.
Philippines Parp Inhibitor Biomarkers Market Future Outlook
The future of the Philippines PARP inhibitor biomarkers market appears promising, driven by ongoing research and development efforts. As the government increases funding for cancer research, the landscape for innovative therapies is expected to evolve. Additionally, the integration of advanced technologies, such as artificial intelligence in biomarker discovery, will likely enhance the precision of cancer treatments. These trends indicate a shift towards more effective and personalized cancer care, ultimately improving patient outcomes and survival rates in the coming years.
Market Opportunities
- Expansion of Diagnostic Testing:The growing emphasis on early cancer detection presents a significant opportunity for the expansion of diagnostic testing services. With an estimated 60% of cancer cases diagnosed at advanced stages, enhancing diagnostic capabilities can lead to earlier interventions and improved patient outcomes, thereby increasing the demand for PARP inhibitors.
- Collaborations with Research Institutions:Collaborating with local and international research institutions can accelerate the development of new PARP inhibitors. Such partnerships can leverage shared resources and expertise, fostering innovation in treatment options. This collaborative approach is essential for addressing the unique cancer challenges faced in the Philippines, ultimately benefiting patients through improved therapeutic options.