About the Report
Base Year 2024Philippines Preclinical CRO Market Overview
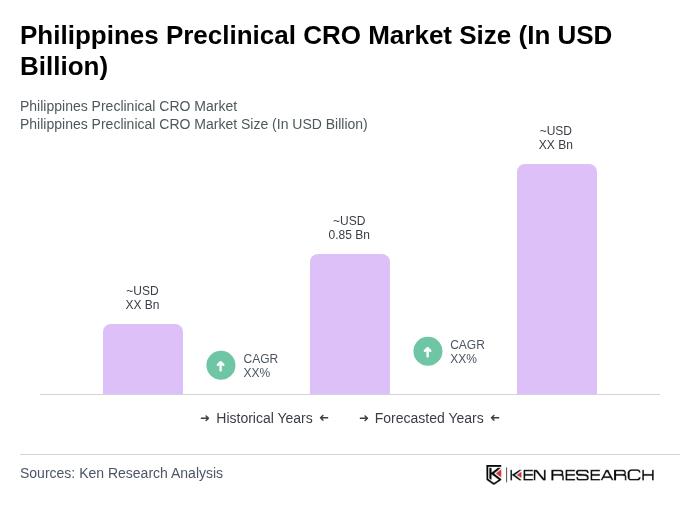
- The Philippines Preclinical CRO Market is valued at USD 0.85 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing outsourcing of preclinical testing by pharmaceutical and biotechnology companies, which seek cost efficiency and faster time-to-market. Additionally, rising investments in research and development, along with a skilled workforce and adherence to international regulatory standards, contribute to a favorable environment for contracted preclinical services.
- Key cities dominating the market include Metro Manila, which serves as the hub for pharmaceutical and biotechnology firms due to its advanced infrastructure and access to skilled professionals. Other regions like Luzon and Visayas also play significant roles, supported by local universities and research institutions that foster collaboration and innovation in preclinical research.
- The regulatory framework governing the preclinical CRO sector in the Philippines is shaped by the Philippine FDA mandates, including Administrative Order No. 2014-0034 and FDA Circular No. 2015-003. These regulations require CROs to secure a License to Operate (LTO) and adhere to updated compliance protocols outlined in AO No. 2024-0015, ensuring quality and alignment with international clinical trial standards.
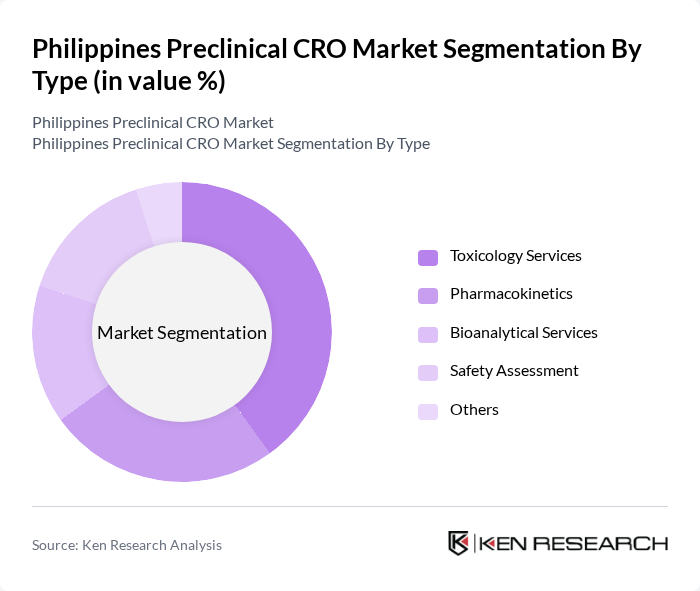
Philippines Preclinical CRO Market Segmentation
By Type:The preclinical CRO market is segmented into various types of services, including Toxicology Services, Pharmacokinetics, Bioanalytical Services, Safety Assessment, and Others. Among these, Toxicology Services dominate the market due to the increasing demand for safety assessments in drug development. The need for comprehensive toxicological evaluations is critical for regulatory approvals, making this segment essential for pharmaceutical companies.
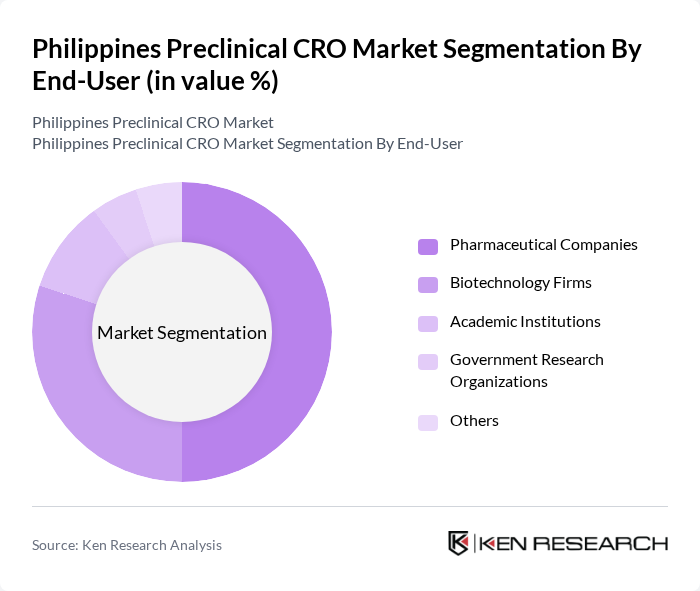
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, Government Research Organizations, and Others. Pharmaceutical Companies are the leading end-users, driven by their need for extensive preclinical testing to ensure drug safety and efficacy. The collaboration between these companies and CROs enhances the overall drug development process, making this segment pivotal in the market.
Philippines Preclinical CRO Market Competitive Landscape
The Philippines Preclinical CRO Market is characterized by a dynamic mix of regional and international players. Leading participants such as PPD, Charles River Laboratories, Covance, WuXi AppTec, Eurofins Scientific, Medpace, Syneos Health, ICON plc, PRA Health Sciences, Labcorp Drug Development, KCR, BioClinica, Clinipace, Celerion, Medpace contribute to innovation, geographic expansion, and service delivery in this space.
Philippines Preclinical CRO Market Industry Analysis
Growth Drivers
- Robust National GDP and Rising Per-Capita Income:The Philippines' nominal GDP in the future is projected at USD 471.5 billion, with a GDP per capita of USD 4,130. This economic foundation supports increased investment in specialized sectors, including preclinical contract research organizations (CROs). The growing economy enhances the capacity for funding drug development and research initiatives, which are crucial for the expansion of preclinical services in the country. Source: Republic-level macroeconomic figures.
- Expanding Inward Foreign Direct Investment (FDI):Inward FDI in the Philippines reached USD 125.53 billion in the future. This influx of capital is vital for developing healthcare infrastructure and research capabilities, enabling local CROs to enhance their service offerings. Increased foreign investment often correlates with improved research facilities and technology adoption, which are essential for competitive preclinical services. Source: Philippines macroeconomic data.
- Asia-Pacific as a High-Growth Region for Preclinical CRO Market:The Asia-Pacific preclinical CRO market is valued at USD 968.4 million in the future. As the region experiences rapid growth, the Philippines stands to benefit from increased outsourcing and the establishment of preclinical research facilities. This trend positions the country as an emerging hub for biopharmaceutical research, attracting both local and international clients seeking cost-effective solutions. Source: Asia-Pacific CRO market figures.
Market Challenges
- Economic and Infrastructure Constraints:Despite a growing economy, the Philippines has a modest GDP per capita of USD 4,130 in the future, which limits the availability of capital for high-cost infrastructure necessary for advanced preclinical CRO services. This economic constraint can hinder the establishment and expansion of specialized research facilities, impacting the overall growth of the sector. Source: Philippines GDP per capita.
- Regulatory Complexity and Institutional Gaps:The global CRO market faces significant regulatory hurdles and compliance issues, which are echoed in the Philippines. Without streamlined local regulatory frameworks and Good Laboratory Practice (GLP)-compliant infrastructure, Philippine CROs may struggle to gain the trust of international clients, limiting their competitiveness in the global market. Source: Global CRO market challenges.
Philippines Preclinical CRO Market Future Outlook
The future of the Philippines preclinical CRO market appears promising, driven by technological advancements and increasing global demand for outsourcing. As the industry shifts towards AI-enabled drug discovery and specialized services like biologics and bioanalysis, local CROs that invest in these areas are likely to capture significant market share. Additionally, fostering collaborations with academic institutions and government agencies can enhance research capabilities and attract further investment, positioning the Philippines as a competitive player in the regional CRO landscape.
Market Opportunities
- Targeting Untapped Domestic Pharma and Biotech Segments:The Philippines' pharmaceutical sector is emerging, supported by GDP growth and increasing healthcare demand. Local CROs can capitalize on this by providing preclinical services tailored to domestic biotech firms, reducing reliance on international contracts and fostering local innovation. Source: Philippines economy sectors.
- Export Potential via Regional Outsourcing:With the Asia-Pacific preclinical CRO market projected at USD 968.4 million in the future, Philippine CROs can position themselves as cost-effective alternatives to established markets like China and India. This regional demand presents a significant opportunity for local firms to expand their service offerings and attract international clients seeking GLP-compliant preclinical testing. Source: Asia-Pacific market data.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Toxicology Services Pharmacokinetics Bioanalytical Services Safety Assessment Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Government Research Organizations Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Service Model | Full-Service CROs Functional Service Providers Hybrid Models Others |
| By Study Phase | Preclinical Studies Phase I Trials Phase II Trials Others |
| By Geographic Focus | Metro Manila Luzon Visayas Mindanao Others |
| By Client Type | Large Enterprises SMEs Startups Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Department of Health)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations (CROs)
Contract Manufacturing Organizations (CMOs)
Pharmaceutical Supply Chain Partners
Healthcare Technology Companies
Pharmaceutical Industry Associations
Players Mentioned in the Report:
PPD
Charles River Laboratories
Covance
WuXi AppTec
Eurofins Scientific
Medpace
Syneos Health
ICON plc
PRA Health Sciences
Labcorp Drug Development
KCR
BioClinica
Clinipace
Celerion
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Philippines Preclinical CRO Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Philippines Preclinical CRO Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Philippines Preclinical CRO Market Analysis
3.1 Growth Drivers
3.1.1 Increasing investment in drug development
3.1.2 Rising demand for outsourcing preclinical services
3.1.3 Advancements in biotechnology and pharmaceuticals
3.1.4 Supportive government policies for research and development
3.2 Market Challenges
3.2.1 Limited funding for early-stage research
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Competition from established global CROs
3.2.4 Shortage of skilled professionals in the field
3.3 Market Opportunities
3.3.1 Expansion of biopharmaceutical companies in the region
3.3.2 Collaborations with academic institutions
3.3.3 Growth in personalized medicine and genomics
3.3.4 Increasing focus on animal welfare in research
3.4 Market Trends
3.4.1 Shift towards integrated CRO services
3.4.2 Adoption of digital technologies in research
3.4.3 Emphasis on data analytics and AI in preclinical studies
3.4.4 Rising importance of regulatory compliance and quality assurance
3.5 Government Regulation
3.5.1 Implementation of stricter animal testing regulations
3.5.2 Guidelines for Good Laboratory Practice (GLP)
3.5.3 Policies promoting ethical research practices
3.5.4 Incentives for local CROs to enhance capabilities
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Philippines Preclinical CRO Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Philippines Preclinical CRO Market Segmentation
8.1 By Type
8.1.1 Toxicology Services
8.1.2 Pharmacokinetics
8.1.3 Bioanalytical Services
8.1.4 Safety Assessment
8.1.5 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Academic Institutions
8.2.4 Government Research Organizations
8.2.5 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Cardiovascular
8.3.3 Neurology
8.3.4 Infectious Diseases
8.3.5 Others
8.4 By Service Model
8.4.1 Full-Service CROs
8.4.2 Functional Service Providers
8.4.3 Hybrid Models
8.4.4 Others
8.5 By Study Phase
8.5.1 Preclinical Studies
8.5.2 Phase I Trials
8.5.3 Phase II Trials
8.5.4 Others
8.6 By Geographic Focus
8.6.1 Metro Manila
8.6.2 Luzon
8.6.3 Visayas
8.6.4 Mindanao
8.6.5 Others
8.7 By Client Type
8.7.1 Large Enterprises
8.7.2 SMEs
8.7.3 Startups
8.7.4 Others
9. Philippines Preclinical CRO Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Client Retention Rate
9.2.5 Market Penetration Rate
9.2.6 Pricing Strategy
9.2.7 Service Diversification Index
9.2.8 Customer Satisfaction Score
9.2.9 Operational Efficiency Ratio
9.2.10 Innovation Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PPD
9.5.2 Charles River Laboratories
9.5.3 Covance
9.5.4 WuXi AppTec
9.5.5 Eurofins Scientific
9.5.6 Medpace
9.5.7 Syneos Health
9.5.8 ICON plc
9.5.9 PRA Health Sciences
9.5.10 Labcorp Drug Development
9.5.11 KCR
9.5.12 BioClinica
9.5.13 Clinipace
9.5.14 Celerion
9.5.15 Medpace
10. Philippines Preclinical CRO Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Department of Science and Technology
10.1.3 Department of Trade and Industry
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Sector Investments
10.2.2 Biotechnology Sector Investments
10.2.3 Government Research Funding
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotechnology Firms
10.3.3 Academic Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of CRO Services
10.4.2 Willingness to Outsource
10.4.3 Budget Allocations
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Deployments
10.5.3 Future Expansion Plans
10.5.4 Others
11. Philippines Preclinical CRO Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from local and international health organizations
- Review of government publications and regulatory frameworks related to preclinical research
- Examination of academic journals and publications focusing on CRO activities in the Philippines
Primary Research
- Interviews with executives from leading preclinical CROs operating in the Philippines
- Surveys targeting pharmaceutical companies and biotech firms utilizing CRO services
- Field interviews with researchers and scientists involved in preclinical studies
Validation & Triangulation
- Cross-validation of data through multiple sources including industry reports and expert opinions
- Triangulation of findings from primary interviews and secondary data sources
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure
- Segmentation of the market by therapeutic areas and types of preclinical services offered
- Incorporation of growth trends in the pharmaceutical and biotechnology sectors
Bottom-up Modeling
- Collection of data on service pricing and volume from leading CROs
- Estimation of market share based on the number of active preclinical studies
- Calculation of revenue projections based on service demand and pricing strategies
Forecasting & Scenario Analysis
- Utilization of historical growth rates to project future market trends
- Scenario analysis based on potential regulatory changes and funding availability
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Preclinical Studies | 100 | Clinical Research Directors, Project Managers |
| Biotechnology CRO Services | 80 | Biotech Researchers, Regulatory Affairs Specialists |
| Contract Research for Medical Devices | 60 | Product Development Managers, Quality Assurance Leads |
| Academic Collaborations in Preclinical Research | 70 | University Researchers, Lab Managers |
| Preclinical Toxicology Services | 90 | Toxicologists, Safety Assessment Managers |
Frequently Asked Questions
What is the current value of the Philippines Preclinical CRO Market?
The Philippines Preclinical CRO Market is valued at approximately USD 0.85 billion, reflecting a significant growth trend driven by increased outsourcing of preclinical testing by pharmaceutical and biotechnology companies seeking cost efficiency and faster time-to-market.