Qatar Clinical Trial Imaging Market Overview
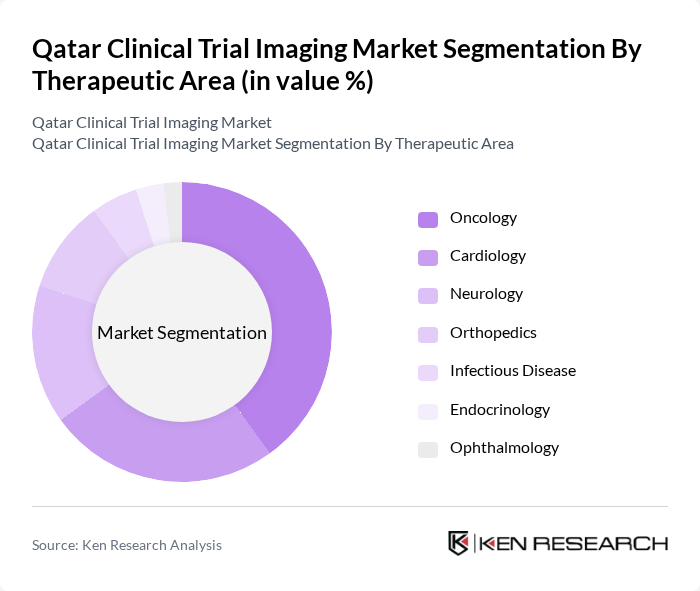
- The Qatar Clinical Trial Imaging Market is valued at USD 150 million, based on a five-year historical analysis. This growth is primarily driven by the increasing number of clinical trials, advancements in imaging technologies, and the rising demand for efficient diagnostic solutions in the healthcare sector. The integration of artificial intelligence and digital imaging technologies has further enhanced the capabilities of imaging modalities, making them indispensable in clinical research. The adoption of AI-powered imaging solutions is accelerating, with hospitals and research centers increasingly leveraging these tools for improved diagnostic accuracy and operational efficiency .
- Doha is the dominant city in the Qatar Clinical Trial Imaging Market due to its robust healthcare infrastructure, presence of leading hospitals, and research institutions. The city is home to several international healthcare organizations and has established itself as a hub for clinical research, attracting investments and fostering innovation in medical imaging technologies. Doha hosts major medical centers such as Hamad Medical Corporation and Sidra Medicine, which are actively involved in advanced imaging research and clinical trials .
- In 2023, the Qatari government implemented the Clinical Trials Regulations, 2023 issued by the Ministry of Public Health, to streamline the approval process for clinical trials, enhancing the efficiency of imaging services in research. This initiative aims to reduce the time required for regulatory approvals, thereby encouraging more pharmaceutical and biotechnology companies to conduct clinical trials in Qatar, ultimately boosting the demand for advanced imaging solutions. The regulations set clear timelines for review and approval, mandate standardized protocols, and require compliance with international ethical standards .
Qatar Clinical Trial Imaging Market Segmentation
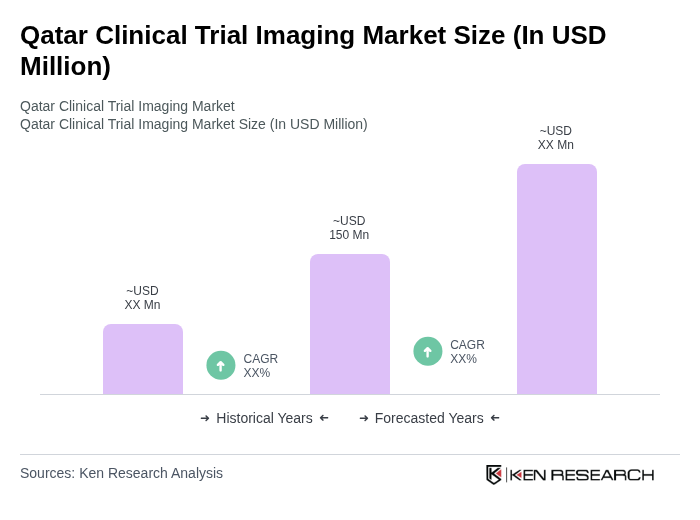
By Imaging Modality:The imaging modalities used in clinical trials are crucial for accurate diagnosis and monitoring of treatment efficacy. The market is segmented into various modalities, including MRI, CT Scans, PET Scans, Ultrasound, X-ray, Echocardiography, and Others. Each modality serves specific diagnostic purposes, with MRI and CT scans being the most widely utilized due to their advanced imaging capabilities and non-invasive nature. MRI remains the leading modality, valued for its detailed soft tissue imaging and broad application across therapeutic areas .
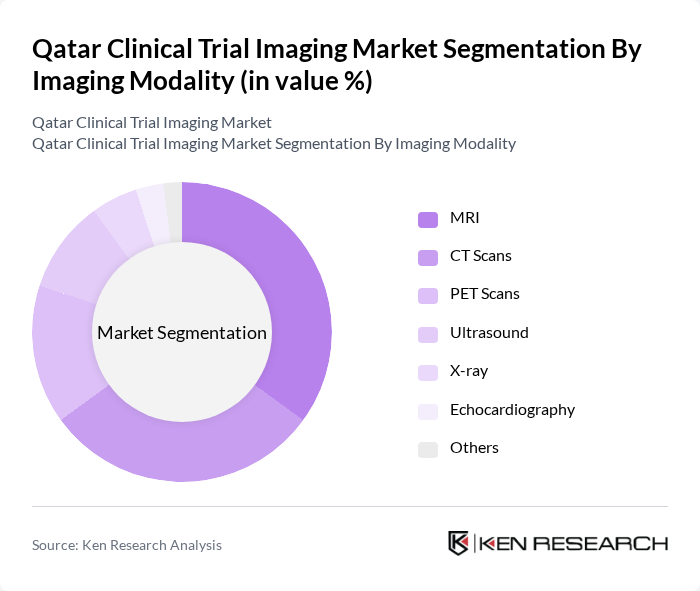
By Therapeutic Area:The therapeutic areas in which imaging modalities are applied include Oncology, Cardiology, Neurology, Orthopedics, Infectious Disease, Endocrinology, Ophthalmology, and Others. Oncology is the leading therapeutic area, driven by the increasing prevalence of cancer and the need for precise imaging techniques to guide treatment decisions and monitor patient outcomes. Imaging technologies play a vital role in diagnosing, staging, and monitoring cancer, making them essential in oncology-focused clinical trials .
Qatar Clinical Trial Imaging Market Competitive Landscape
The Qatar Clinical Trial Imaging Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, GE Healthcare, Philips Healthcare, Canon Medical Systems, Fujifilm Healthcare, Agfa HealthCare, Hitachi Healthcare, Carestream Health, Mindray Medical International, Varian Medical Systems, EIZO Corporation, Hologic, Inc., Bracco Imaging, Medica Group PLC, ICON plc, Parexel International, Syneos Health, QSTP (Qatar Science & Technology Park), Hamad Medical Corporation, Sidra Medicine contribute to innovation, geographic expansion, and service delivery in this space.
Qatar Clinical Trial Imaging Market Industry Analysis
Growth Drivers
- Increasing Demand for Advanced Imaging Technologies:The Qatar clinical trial imaging market is experiencing a surge in demand for advanced imaging technologies, driven by the need for precise diagnostics. In future, the healthcare expenditure in Qatar is projected to reach approximately $6.2 billion, reflecting a steady increase from previous years. This growth is fueled by the rising prevalence of chronic diseases, necessitating advanced imaging solutions for effective clinical trials. Enhanced imaging technologies are essential for improving patient outcomes and accelerating drug development processes.
- Rising Number of Clinical Trials in Qatar:Qatar has seen a significant increase in clinical trials, with fewer than 100 active trials reported recently, a notable rise from the previous year. This trend is supported by the Qatar National Research Strategy, which aims to enhance research capabilities. The growing number of clinical trials necessitates advanced imaging solutions to ensure accurate data collection and analysis, thereby driving the demand for imaging services in the clinical trial sector.
- Government Support for Healthcare Innovation:The Qatari government is actively promoting healthcare innovation through initiatives like the Qatar National Vision 2030, which allocates substantial funding for healthcare advancements. In future, the government is expected to invest around $1.5 billion in healthcare infrastructure, including imaging technologies. This support fosters an environment conducive to clinical trial advancements, encouraging local and international companies to invest in imaging solutions tailored for clinical research.
Market Challenges
- High Costs Associated with Advanced Imaging Equipment:The high costs of advanced imaging equipment pose a significant challenge for clinical trial imaging in Qatar. For instance, the price of MRI machines can exceed $1 million, making it difficult for smaller research facilities to acquire necessary technology. This financial barrier limits access to cutting-edge imaging solutions, potentially hindering the quality and efficiency of clinical trials conducted in the region.
- Regulatory Hurdles in Clinical Trial Approvals:Navigating the regulatory landscape for clinical trials in Qatar can be complex and time-consuming. The approval process for new imaging technologies often involves multiple regulatory bodies, leading to delays. The average time for obtaining clinical trial approval was approximately 6 months, which can impede timely access to innovative imaging solutions. These regulatory challenges can deter investment and slow down the overall growth of the clinical trial imaging market.
Qatar Clinical Trial Imaging Market Future Outlook
The future of the Qatar clinical trial imaging market appears promising, driven by technological advancements and increased collaboration among stakeholders. The integration of artificial intelligence in imaging solutions is expected to enhance diagnostic accuracy and efficiency. Additionally, the expansion of telemedicine services will facilitate remote imaging capabilities, making clinical trials more accessible. As the government continues to invest in healthcare infrastructure, the market is likely to witness significant growth, fostering innovation and improving patient outcomes in clinical research.
Market Opportunities
- Expansion of Telemedicine and Remote Imaging Services:The rise of telemedicine presents a unique opportunity for the clinical trial imaging market. In future, it is estimated that telemedicine services in Qatar will grow to encompass over 30% of healthcare consultations, allowing for remote imaging services that can streamline clinical trial processes and improve patient participation rates.
- Collaborations with International Research Organizations:Collaborating with international research organizations can enhance the capabilities of Qatar's clinical trial imaging market. Such partnerships can facilitate knowledge transfer and access to advanced imaging technologies, potentially increasing the number of clinical trials conducted in the region and improving the overall quality of research outcomes.