Region:Middle East
Author(s):Rebecca
Product Code:KRAC4640
Pages:86
Published On:October 2025
 Solutions Market.png)
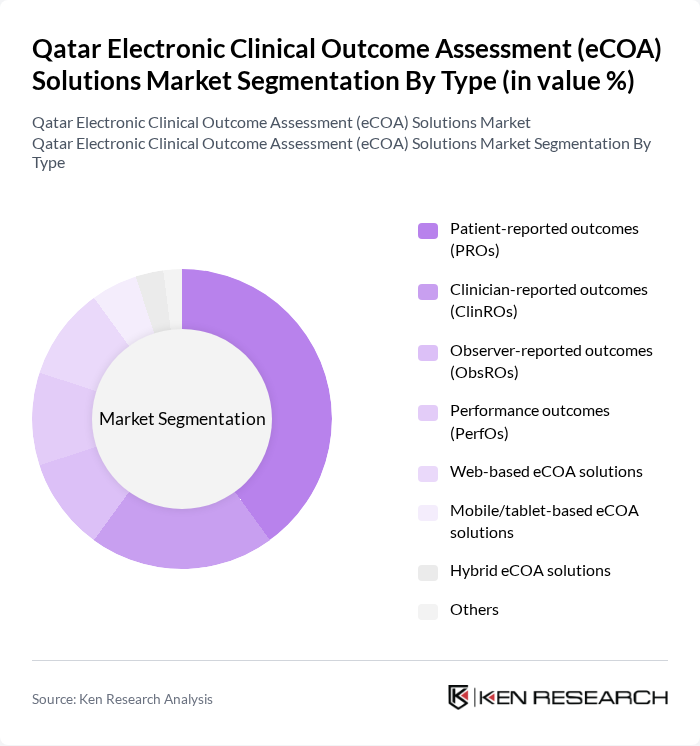
By Type:The eCOA solutions market can be segmented into various types, including Patient-reported outcomes (PROs), Clinician-reported outcomes (ClinROs), Observer-reported outcomes (ObsROs), Performance outcomes (PerfOs), Web-based eCOA solutions, Mobile/tablet-based eCOA solutions, Hybrid eCOA solutions, and Others. Among these, Patient-reported outcomes (PROs) are currently dominating the market due to their ability to capture the patient's perspective on health status and treatment effects, which is increasingly recognized as vital in clinical research and healthcare delivery.
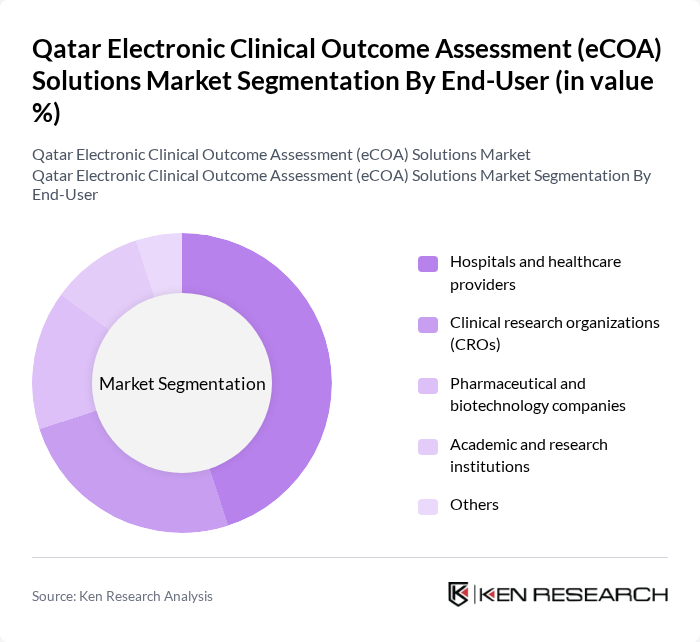
By End-User:The eCOA solutions market is segmented by end-users, including Hospitals and healthcare providers, Clinical research organizations (CROs), Pharmaceutical and biotechnology companies, Academic and research institutions, and Others. Hospitals and healthcare providers are leading this segment as they increasingly adopt eCOA solutions to enhance patient engagement, streamline clinical workflows, and improve data collection processes in both clinical practice and research settings.
The Qatar Electronic Clinical Outcome Assessment (eCOA) Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Inc. (Dassault Systèmes), Oracle Corporation (Oracle Health Sciences), Veeva Systems Inc., Signant Health, Clario (formerly ERT and Bioclinica), Parexel International Corporation, YPrime, LLC, Clinical Ink, Inc., Medable Inc., ArisGlobal LLC, Suvoda LLC, Castor EDC, eClinical Solutions LLC, IQVIA Inc., ICON plc contribute to innovation, geographic expansion, and service delivery in this space.
The future of the eCOA solutions market in Qatar appears promising, driven by technological advancements and increasing healthcare digitization. The integration of artificial intelligence and machine learning into eCOA systems is expected to enhance data analysis and patient engagement. Additionally, the growing emphasis on personalized medicine will likely lead to more tailored eCOA solutions, improving patient outcomes. As regulatory frameworks evolve, the market is poised for significant growth, fostering innovation and collaboration among stakeholders.
| Segment | Sub-Segments |
|---|---|
| By Type | Patient-reported outcomes (PROs) Clinician-reported outcomes (ClinROs) Observer-reported outcomes (ObsROs) Performance outcomes (PerfOs) Web-based eCOA solutions Mobile/tablet-based eCOA solutions Hybrid eCOA solutions Others |
| By End-User | Hospitals and healthcare providers Clinical research organizations (CROs) Pharmaceutical and biotechnology companies Academic and research institutions Others |
| By Application | Oncology Cardiovascular diseases Neurological disorders Diabetes management Rare diseases and orphan indications Others |
| By Deployment Mode | Cloud-based solutions On-premise solutions Hybrid solutions |
| By Sales Channel | Direct sales Distributors Online platforms |
| By Region | Doha Al Rayyan Al Wakrah Others |
| By Pricing Model | Subscription-based Pay-per-use One-time license fee Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 120 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Medical Device Studies | 90 | Product Development Managers, Clinical Research Coordinators |
| Patient-Reported Outcomes Research | 60 | Healthcare Providers, Patient Advocacy Representatives |
| Digital Health Initiatives | 70 | IT Managers, Digital Transformation Officers |
| Regulatory Compliance in eCOA | 50 | Compliance Officers, Quality Assurance Managers |
The Qatar Electronic Clinical Outcome Assessment (eCOA) Solutions Market is valued at approximately USD 20 million, reflecting a significant growth trend driven by the adoption of digital health technologies and the demand for real-time patient data collection.