About the Report
Base Year 2024Qatar Induced Pluripotent Stem Cells Production Market Overview
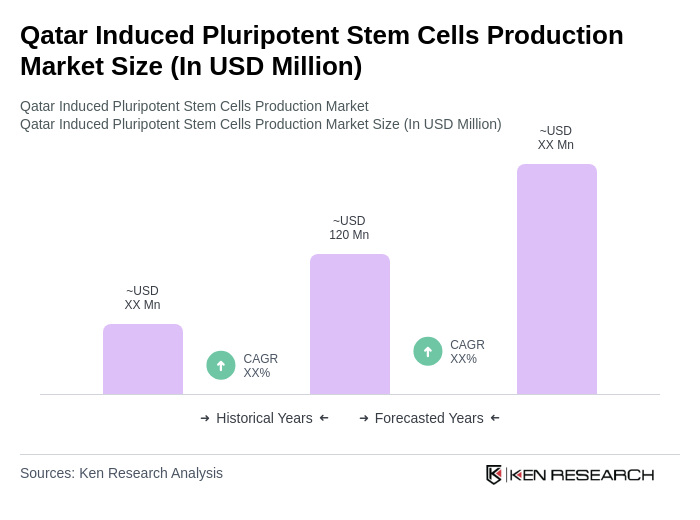
- The Qatar Induced Pluripotent Stem Cells Production Market is valued at USD 120 million, based on a five-year historical analysis and regional share of the Middle East stem cell market. Growth is primarily driven by advancements in regenerative medicine, increasing investments in biotechnology, and a rising demand for personalized medicine solutions. The market is further supported by a growing number of research initiatives and collaborations between academic institutions and private companies, as well as the expansion of clinical applications for iPSC-derived therapies .
- Key players in this market are concentrated in Doha, Al Rayyan, and Lusail, which lead due to robust healthcare infrastructure, advanced research facilities, and strong government support for biotechnology initiatives. These cities host major medical institutions and research centers that drive innovation and development in stem cell research and clinical applications .
- The Qatari government regulates stem cell research and clinical applications under the “Qatar National Research Ethics Committee Guidelines for Stem Cell Research, 2023” issued by the Ministry of Public Health. This framework establishes operational standards for the ethical use of induced pluripotent stem cells in research and therapy, mandates institutional review board approval, and requires compliance with international best practices for donor consent, cell line derivation, and clinical translation.
Qatar Induced Pluripotent Stem Cells Production Market Segmentation
By Type:The market is segmented into several stem cell types: Induced Pluripotent Stem Cells (iPSCs), Mesenchymal Stem Cells (MSCs), Hematopoietic Stem Cells (HSCs), and others. iPSCs lead the market due to their versatility in regenerative medicine, disease modeling, and drug discovery. The ability of iPSCs to differentiate into multiple cell types and their increasing use in personalized medicine and advanced therapeutic applications drive their dominance in the segment .

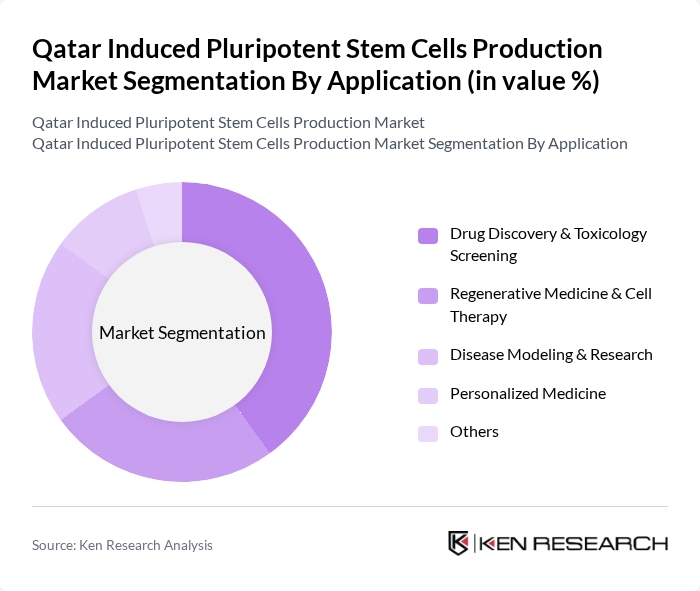
By Application:Applications of induced pluripotent stem cells include Drug Discovery & Toxicology Screening, Regenerative Medicine & Cell Therapy, Disease Modeling & Research, Personalized Medicine, and others. Drug Discovery & Toxicology Screening currently dominates due to the growing need for efficient drug development and the ability of iPSCs to provide physiologically relevant models for preclinical testing. Regenerative medicine and disease modeling are also rapidly expanding segments as clinical translation and disease-specific cell line development accelerate .
Qatar Induced Pluripotent Stem Cells Production Market Competitive Landscape
The Qatar Induced Pluripotent Stem Cells Production Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sidra Medicine, Hamad Medical Corporation (HMC), Qatar Biobank, Qatar Foundation, Qatar Science & Technology Park (QSTP), Raf Healthcare Consultancy, Thermo Fisher Scientific, Inc., STEMCELL Technologies Inc., Takara Bio Inc., Lonza Group AG, Merck KGaA, Cellular Dynamics International, Inc., Fate Therapeutics, Inc., ReNeuron Group plc, Bluebird Bio, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Qatar Induced Pluripotent Stem Cells Production Market Industry Analysis
Growth Drivers
- Increasing Research Funding:Qatar's investment in research and development has seen a significant increase, with funding reaching approximately QAR 1.5 billion in future. This financial support is crucial for advancing stem cell research, particularly in regenerative medicine. The Qatar National Research Fund (QNRF) has allocated substantial grants, fostering innovation and collaboration among local universities and research institutions. This influx of capital is expected to enhance the production capabilities of induced pluripotent stem cells (iPSCs), driving market growth.
- Advancements in Regenerative Medicine:The global regenerative medicine market is valued at over USD 24 billion according to the most recent World Health Organization and OECD data, with Qatar positioning itself as a key player. The country's focus on developing advanced therapies using iPSCs is supported by local universities and research centers. Notably, the establishment of specialized facilities for stem cell research has led to breakthroughs in treating chronic diseases, thereby increasing the demand for iPSCs. This trend is expected to significantly boost the market in the coming years.
- Supportive Government Initiatives:The Qatari government has implemented various initiatives to promote biotechnology and stem cell research. In future, the government launched a national strategy aimed at enhancing healthcare innovation, with a budget of QAR 2 billion. This strategy includes incentives for private sector investments in stem cell technologies and partnerships with international research organizations. Such initiatives are expected to create a conducive environment for the growth of the iPSC production market in Qatar.
Market Challenges
- High Production Costs:The production of induced pluripotent stem cells involves complex and costly processes, with estimates indicating that the average cost per cell line can exceed QAR 500,000. This high cost poses a significant barrier to entry for new players in the market and limits the scalability of existing operations. As a result, many research institutions may struggle to secure funding for large-scale production, hindering overall market growth.
- Ethical Concerns:Ethical issues surrounding stem cell research remain a significant challenge in Qatar. Public perception and ethical debates can influence funding and regulatory approvals. In future, approximately 60% of the population expressed concerns regarding the ethical implications of stem cell research, which may lead to stricter regulations. These concerns can slow down research initiatives and deter potential investors, impacting the growth of the iPSC production market.
Qatar Induced Pluripotent Stem Cells Production Market Future Outlook
The future of the Qatar induced pluripotent stem cells production market appears promising, driven by increasing investments in biotechnology and a growing emphasis on personalized medicine. As the government continues to support research initiatives, collaborations with international institutions are likely to expand. Furthermore, advancements in technology, such as CRISPR and AI integration, will enhance research capabilities. These factors combined suggest a robust growth trajectory for the market, positioning Qatar as a leader in stem cell research and applications in the region.
Market Opportunities
- Collaborations with Research Institutions:Establishing partnerships with global research institutions can enhance Qatar's capabilities in stem cell research. Such collaborations can lead to knowledge transfer, access to advanced technologies, and shared funding opportunities, ultimately boosting the local iPSC production market.
- Investment in Biobanking:The establishment of biobanks in Qatar presents a significant opportunity for the iPSC market. By investing in biobanking, researchers can ensure a steady supply of high-quality stem cells for research and therapeutic applications, facilitating advancements in regenerative medicine and personalized therapies.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Induced Pluripotent Stem Cells (iPSCs) Mesenchymal Stem Cells (MSCs) Hematopoietic Stem Cells (HSCs) Others |
| By Application | Drug Discovery & Toxicology Screening Regenerative Medicine & Cell Therapy Disease Modeling & Research Personalized Medicine Others |
| By End-User | Academic & Research Institutions Hospitals & Specialty Clinics Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
| By Source | Human Somatic Cells Cord Blood & Perinatal Sources Others |
| By Delivery Method | Direct Injection Infusion Scaffold-based Delivery Others |
| By Research Phase | Preclinical Research Clinical Trials Commercial Production Post-Market Surveillance |
| By Funding Source | Government Grants & Public Funding Private Investments & Venture Capital Institutional & Corporate Funding International Collaborations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Qatar Ministry of Public Health, Qatar Biomedical Research Institute)
Biotechnology and Pharmaceutical Companies
Healthcare Providers and Hospitals
Research and Development Organizations
Biobanks and Cell Culture Facilities
Clinical Research Organizations
Healthcare Technology Companies
Players Mentioned in the Report:
Sidra Medicine
Hamad Medical Corporation (HMC)
Qatar Biobank
Qatar Foundation
Qatar Science & Technology Park (QSTP)
Raf Healthcare Consultancy
Thermo Fisher Scientific, Inc.
STEMCELL Technologies Inc.
Takara Bio Inc.
Lonza Group AG
Merck KGaA
Cellular Dynamics International, Inc.
Fate Therapeutics, Inc.
ReNeuron Group plc
Bluebird Bio, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Qatar Induced Pluripotent Stem Cells Production Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Qatar Induced Pluripotent Stem Cells Production Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Qatar Induced Pluripotent Stem Cells Production Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Research Funding
3.1.2 Advancements in Regenerative Medicine
3.1.3 Rising Demand for Personalized Medicine
3.1.4 Supportive Government Initiatives
3.2 Market Challenges
3.2.1 High Production Costs
3.2.2 Ethical Concerns
3.2.3 Regulatory Hurdles
3.2.4 Limited Awareness Among Healthcare Providers
3.3 Market Opportunities
3.3.1 Collaborations with Research Institutions
3.3.2 Expansion into Emerging Markets
3.3.3 Development of Novel Therapies
3.3.4 Investment in Biobanking
3.4 Market Trends
3.4.1 Growing Use of CRISPR Technology
3.4.2 Increasing Focus on Cell-Based Therapies
3.4.3 Rise of Biopharmaceutical Companies
3.4.4 Integration of AI in Stem Cell Research
3.5 Government Regulation
3.5.1 Guidelines for Stem Cell Research
3.5.2 Approval Processes for Clinical Trials
3.5.3 Funding Programs for Stem Cell Research
3.5.4 Ethical Review Board Requirements
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Qatar Induced Pluripotent Stem Cells Production Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Qatar Induced Pluripotent Stem Cells Production Market Segmentation
8.1 By Type
8.1.1 Induced Pluripotent Stem Cells (iPSCs)
8.1.2 Mesenchymal Stem Cells (MSCs)
8.1.3 Hematopoietic Stem Cells (HSCs)
8.1.4 Others
8.2 By Application
8.2.1 Drug Discovery & Toxicology Screening
8.2.2 Regenerative Medicine & Cell Therapy
8.2.3 Disease Modeling & Research
8.2.4 Personalized Medicine
8.2.5 Others
8.3 By End-User
8.3.1 Academic & Research Institutions
8.3.2 Hospitals & Specialty Clinics
8.3.3 Pharmaceutical & Biotechnology Companies
8.3.4 Contract Research Organizations (CROs)
8.3.5 Others
8.4 By Source
8.4.1 Human Somatic Cells
8.4.2 Cord Blood & Perinatal Sources
8.4.3 Others
8.5 By Delivery Method
8.5.1 Direct Injection
8.5.2 Infusion
8.5.3 Scaffold-based Delivery
8.5.4 Others
8.6 By Research Phase
8.6.1 Preclinical Research
8.6.2 Clinical Trials
8.6.3 Commercial Production
8.6.4 Post-Market Surveillance
8.7 By Funding Source
8.7.1 Government Grants & Public Funding
8.7.2 Private Investments & Venture Capital
8.7.3 Institutional & Corporate Funding
8.7.4 International Collaborations
8.7.5 Others
9. Qatar Induced Pluripotent Stem Cells Production Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Qatar/MENA iPSC segment)
9.2.4 Number of iPSC Lines Produced Annually
9.2.5 Number of Active Clinical Trials (iPSC-based)
9.2.6 Market Penetration Rate (Qatar iPSC market share %)
9.2.7 R&D Investment as % of Revenue
9.2.8 Time-to-Approval for New iPSC Therapies (months)
9.2.9 Strategic Partnerships/Collaborations (count)
9.2.10 Regulatory Compliance Score (MoPH/International)
9.2.11 Product Portfolio Breadth (number of iPSC applications)
9.2.12 Manufacturing Capacity Utilization (%)
9.2.13 Local Workforce Development Initiatives
9.2.14 Intellectual Property (IP) Portfolio Size
9.2.15 Customer Retention Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sidra Medicine
9.5.2 Hamad Medical Corporation (HMC)
9.5.3 Qatar Biobank
9.5.4 Qatar Foundation
9.5.5 Qatar Science & Technology Park (QSTP)
9.5.6 Raf Healthcare Consultancy
9.5.7 Thermo Fisher Scientific, Inc.
9.5.8 STEMCELL Technologies Inc.
9.5.9 Takara Bio Inc.
9.5.10 Lonza Group AG
9.5.11 Merck KGaA
9.5.12 Cellular Dynamics International, Inc.
9.5.13 Fate Therapeutics, Inc.
9.5.14 ReNeuron Group plc
9.5.15 Bluebird Bio, Inc.
10. Qatar Induced Pluripotent Stem Cells Production Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Research Facilities
10.2.2 Funding for Clinical Trials
10.2.3 Expenditure on Technology Upgrades
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Quality Stem Cells
10.3.2 Regulatory Compliance Challenges
10.3.3 Cost Management Issues
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Infrastructure Readiness
10.4.3 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Long-term Benefits Realization
10.5.3 Opportunities for Scaling
11. Qatar Induced Pluripotent Stem Cells Production Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from Qatar's Ministry of Public Health and relevant health authorities
- Review of scientific literature on induced pluripotent stem cells (iPSCs) from academic journals and databases
- Examination of market reports and white papers from biotechnology and pharmaceutical industry associations
Primary Research
- Interviews with leading researchers and scientists in the field of stem cell research in Qatar
- Surveys with biotechnology firms involved in iPSC production and applications
- Focus group discussions with healthcare professionals and regulatory bodies regarding iPSC applications
Validation & Triangulation
- Cross-validation of findings through multiple data sources including academic, industry, and government reports
- Triangulation of market insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel reviews comprising industry veterans and academic leaders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market for iPSCs based on healthcare expenditure in Qatar
- Segmentation of the market by application areas such as regenerative medicine, drug discovery, and disease modeling
- Incorporation of government initiatives and funding for stem cell research and biotechnology
Bottom-up Modeling
- Collection of data on production capacities and operational metrics from local biotechnology firms
- Cost analysis based on laboratory setup, maintenance, and operational expenses for iPSC production
- Volume estimates derived from projected demand in clinical applications and research sectors
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population growth, healthcare advancements, and regulatory changes
- Scenario modeling based on potential shifts in funding, research breakthroughs, and market adoption rates
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotechnology Firms in Qatar | 100 | CEOs, R&D Directors, Product Managers |
| Healthcare Professionals in Regenerative Medicine | 80 | Surgeons, Oncologists, Clinical Researchers |
| Regulatory Bodies and Policy Makers | 50 | Health Policy Advisors, Regulatory Affairs Managers |
| Academic Institutions and Research Centers | 70 | Professors, Research Scientists, Lab Managers |
| Investors and Venture Capitalists in Biotech | 40 | Investment Analysts, Fund Managers, Business Development Executives |
Frequently Asked Questions
What is the current value of the Qatar Induced Pluripotent Stem Cells Production Market?
The Qatar Induced Pluripotent Stem Cells Production Market is valued at approximately USD 120 million, reflecting significant growth driven by advancements in regenerative medicine, increased investments in biotechnology, and a rising demand for personalized medicine solutions.